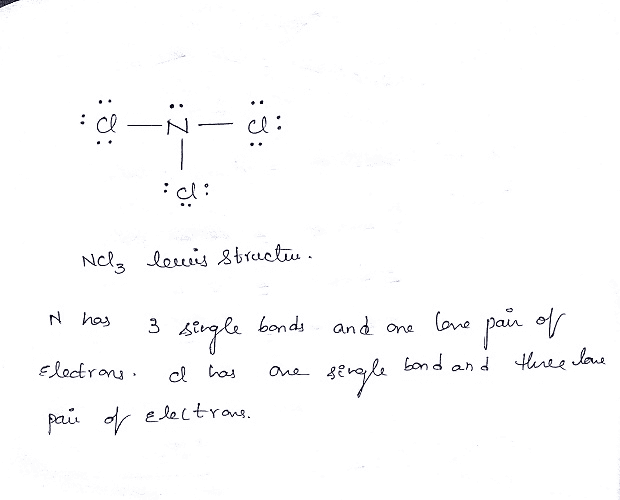Lewis Dot Structure For Nitrogen Trichloride
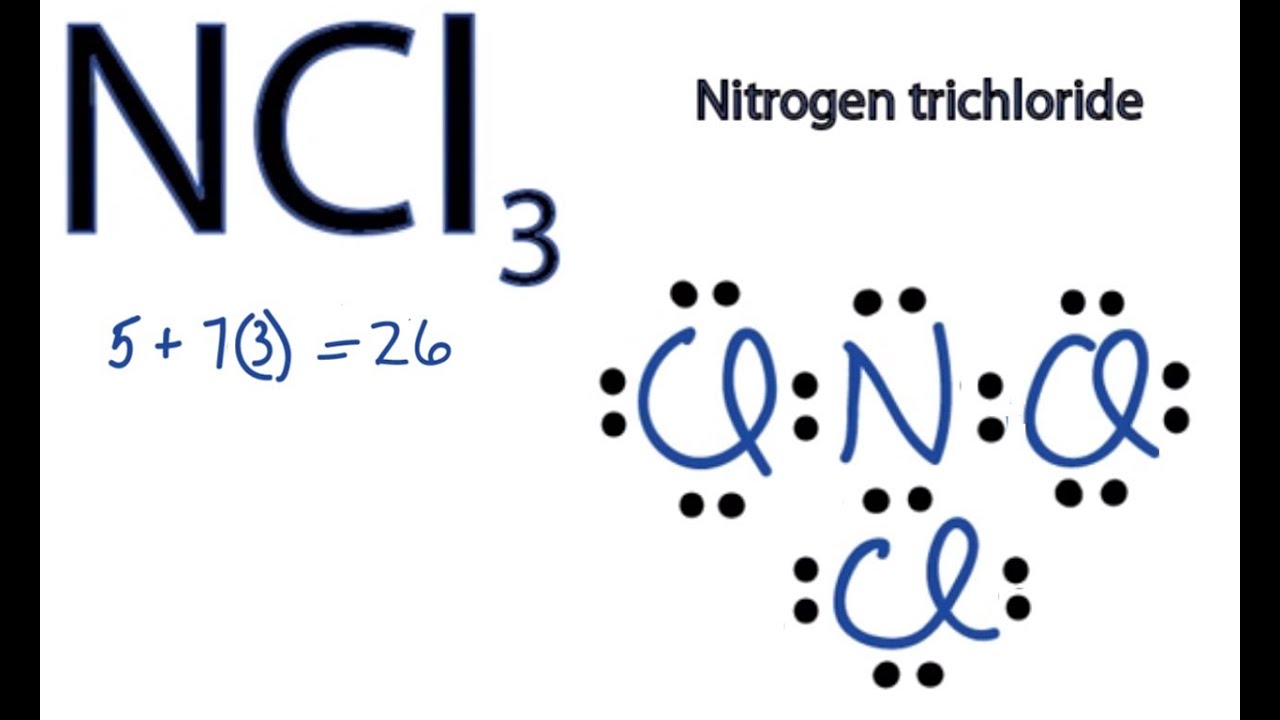
Hey there, science buddy! Ever looked at a chemical formula and thought, "What in the wobbly world of atoms is going on here?" Well, you're not alone! Today, we're diving into the wonderfully weird world of Lewis dot structures, and our star of the show is none other than Nitrogen Trichloride. Don't let the fancy name fool you; it's actually pretty cool once you break it down.
Think of Lewis dot structures as the super-simplified LEGOs of chemistry. Instead of weirdly shaped bricks, we've got atomic symbols, and instead of connecting pegs, we have tiny dots representing electrons. These dots are the real MVPs, the tiny dancers that make all the chemical magic happen. They're basically the atoms' way of holding hands and forming friendships, or in this case, super-strong bonds.
So, what's the deal with Nitrogen Trichloride? It's a molecule made up of one nitrogen atom and three chlorine atoms. Sounds straightforward, right? Well, the way they hang out together is where the Lewis dot structure comes in. It shows us exactly how many of these electron dots each atom has and how they share them to feel all cozy and complete.
Must Read
Let's Get Our Dotty On!
Before we start building our Nitrogen Trichloride masterpiece, we need to know a few things about our atomic buddies. First off, we need to figure out the total number of valence electrons. "Valence electrons," you ask? Think of them as the outermost electrons, the ones that are ready to mingle and form bonds. They're the party animals of the electron world!
Nitrogen (N) is in Group 15 of the periodic table, which means it's got five valence electrons. It's like it’s bringing five snacks to the electron party. Chlorine (Cl), on the other hand, is in Group 17. That means each chlorine atom is rocking seven valence electrons. They're the guests who really know how to bring the goods, with seven snacks each!
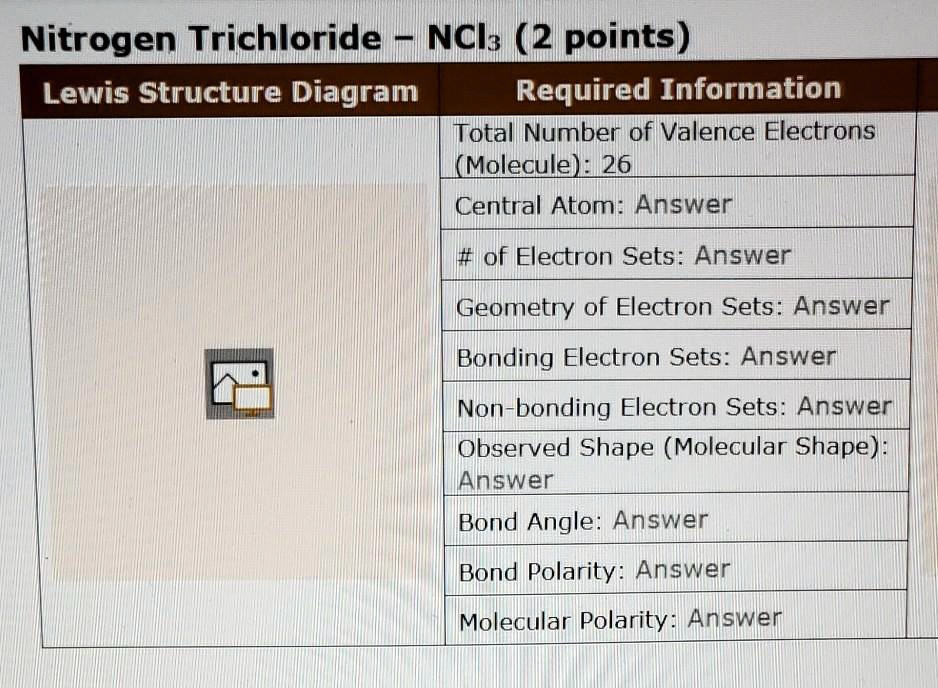
So, for Nitrogen Trichloride (NCl₃), we've got one nitrogen atom (5 valence electrons) and three chlorine atoms (7 valence electrons each). Let's do some quick math, no calculators needed, just your awesome brain power! 5 + (3 * 7) = 5 + 21 = 26 valence electrons in total. Ta-da! That's the grand total of dots we'll be playing with.
The Grand Unveiling: Building the Structure
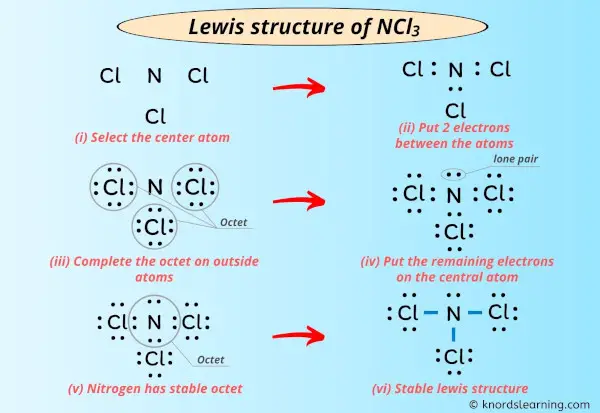
Now for the fun part: putting it all together! We need to decide which atom is going to be the central hub. Generally, the atom that's least electronegative or present in the smallest number usually takes the central stage. In our NCl₃ case, nitrogen is less electronegative than chlorine, and there's only one of it. So, nitrogen gets the VIP treatment and becomes our central atom. It's like the popular kid in school, everyone wants to be near them!
We place our nitrogen atom in the middle. Now, we have our three chlorine atoms hanging around it. Let's imagine them forming a little triangle around our nitrogen friend. It's starting to look like a molecular family portrait already, isn't it?
Next, we start connecting these atoms with single bonds. A single bond is formed when two atoms share a pair of electrons. In our Lewis dot structure, we represent this sharing with a line between the atoms. So, we draw a line from the nitrogen atom to each of the three chlorine atoms. Each line represents two electrons being shared.
Let’s count how many electrons we've used up so far. We have three single bonds, and each bond uses two electrons. That's 3 * 2 = 6 electrons accounted for. We started with 26, so we have 26 - 6 = 20 electrons left to distribute. Phew, still plenty of dots to go around!
Completing the Octet: The Quest for Stability
Atoms, especially in molecules, love to feel "stable." And in chemistry, stability often means having a full outer shell of electrons, typically eight electrons. This is known as the octet rule. It's like they want to have their own little electron clique, all feeling satisfied.
Our chlorine atoms are on the outside, and they've each shared one electron to form a bond with nitrogen. So, they each have 7 electrons in total (6 lone electrons + 2 shared electrons in the bond = 8 electrons). They're already feeling pretty good! But, we need to give them the rest of their lone electrons to complete their octets.
Remember, we had 20 electrons left? We’ll put six lone electrons (represented by dots) around each of the chlorine atoms. Six dots for the first chlorine, six dots for the second, and six dots for the third. That's a total of 3 * 6 = 18 electrons used for the lone pairs on the chlorine atoms. Clever, right?
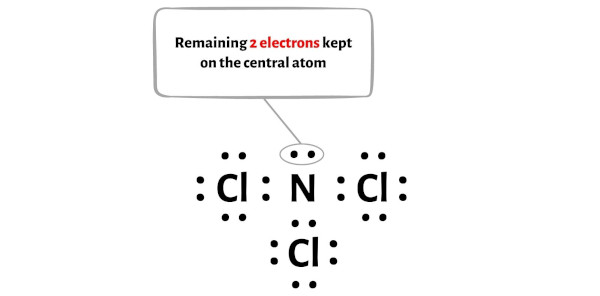
Now, let's see where we stand. We've used 6 electrons for the bonds and 18 electrons for the lone pairs on the chlorine atoms. That's 6 + 18 = 24 electrons used in total. We started with 26, so we have 26 - 24 = 2 electrons remaining. Where do these last two little guys go?
They go on our central nitrogen atom! Nitrogen has already formed three bonds, contributing one electron to each bond. So, it currently "sees" 6 electrons (two from each bond). To complete its octet, it needs two more. These remaining two electrons form a lone pair on the nitrogen atom. We draw them as a pair of dots right on the nitrogen symbol.
The Grand Finale: The Lewis Dot Structure of NCl₃
So, let's put it all together and visualize our masterpiece! In the center, we have our Nitrogen (N) atom.
Branching out from the nitrogen, we have three Chlorine (Cl) atoms.
Between the nitrogen and each chlorine, there's a single bond (represented by a line). Each of these lines signifies two shared electrons.

Each of the three chlorine atoms has three lone pairs of electrons around it (six dots in total). These are the electrons they aren't sharing.
And finally, the nitrogen atom has one lone pair of electrons on it (two dots). This is our leftover pair!
If you count all the electrons now, each chlorine atom has 6 lone electrons + 2 shared electrons in its bond = 8 electrons. They're happy campers! Our nitrogen atom has 6 electrons from the three bonds + 2 electrons from its lone pair = 8 electrons. It's also feeling super stable and content. Mission accomplished! We’ve successfully drawn the Lewis dot structure for Nitrogen Trichloride!
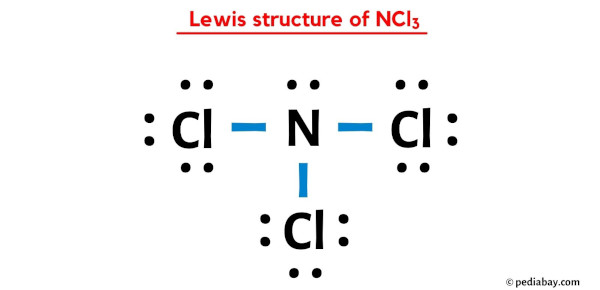
It looks something like this (imagine the dots and lines!):
:Cl:
|
:Cl - N - Cl:

:
Or, if you prefer to see the electrons more explicitly around the central atom:
..
:N:
/ | \
:Cl: :Cl: :Cl:
.. .. ..
The dots you see around the chlorine atoms are their lone pairs. The lines are the shared electron pairs forming the covalent bonds. And the two dots on the nitrogen are its lone pair. Pretty neat, huh?
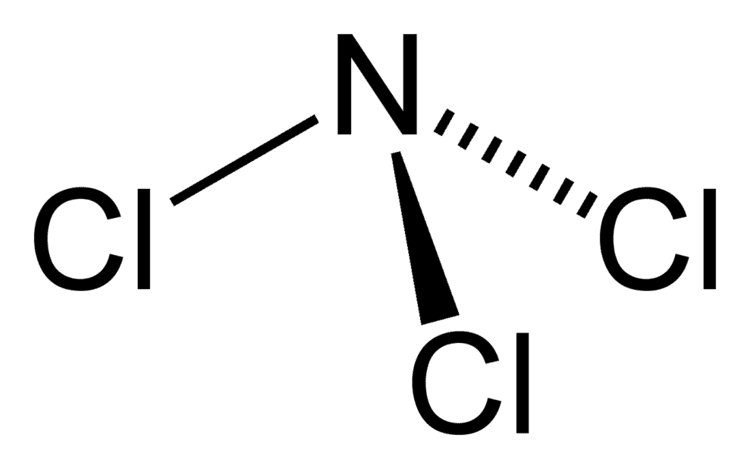
Why is this important, you ask? Well, this little diagram tells us a lot about how Nitrogen Trichloride behaves. The lone pair on the nitrogen atom, for instance, can influence its reactivity and how it interacts with other molecules. It's like the little quirks that make each of us unique and interesting!
A Little Side Quest: Formal Charge (Optional, but Cool!)
Sometimes, to be extra sure we've drawn the best structure, chemists use something called formal charge. It's like a way of assigning a "score" to each atom to see how "happy" it is in the current arrangement. The goal is to have formal charges as close to zero as possible.
For an atom, formal charge = (number of valence electrons) - (number of non-bonding electrons) - (1/2 * number of bonding electrons).
Let's try it for nitrogen in NCl₃: Valence electrons = 5. Non-bonding electrons (lone pair) = 2. Bonding electrons (3 bonds * 2 electrons/bond) = 6. Formal charge for N = 5 - 2 - (1/2 * 6) = 5 - 2 - 3 = 0. Nice! Zero is a happy number for formal charge!
Now for chlorine: Valence electrons = 7. Non-bonding electrons (3 lone pairs * 2 electrons/pair) = 6. Bonding electrons (1 bond * 2 electrons/bond) = 2. Formal charge for Cl = 7 - 6 - (1/2 * 2) = 7 - 6 - 1 = 0. Again, zero! This tells us our Lewis structure is a pretty good representation of how the electrons are distributed.
If you ended up with significant positive or negative formal charges on your atoms, it might be a sign that you need to rearrange some electrons, maybe even try forming a double bond if possible. But for NCl₃, it all works out beautifully!
You've Got This!
See? Lewis dot structures aren't so scary after all! They're like little puzzles that help us understand the hidden world of atoms and how they connect. Nitrogen Trichloride, with its one central nitrogen and three surrounding chlorines, is a classic example, and you've just conquered it!
Every time you see a chemical formula, remember these little dots and lines. They're the secret language of molecules, and you're well on your way to becoming fluent. So go forth, my science adventurer, and draw more Lewis structures! The universe of molecules is waiting for you, and you're definitely equipped to explore it with a smile.