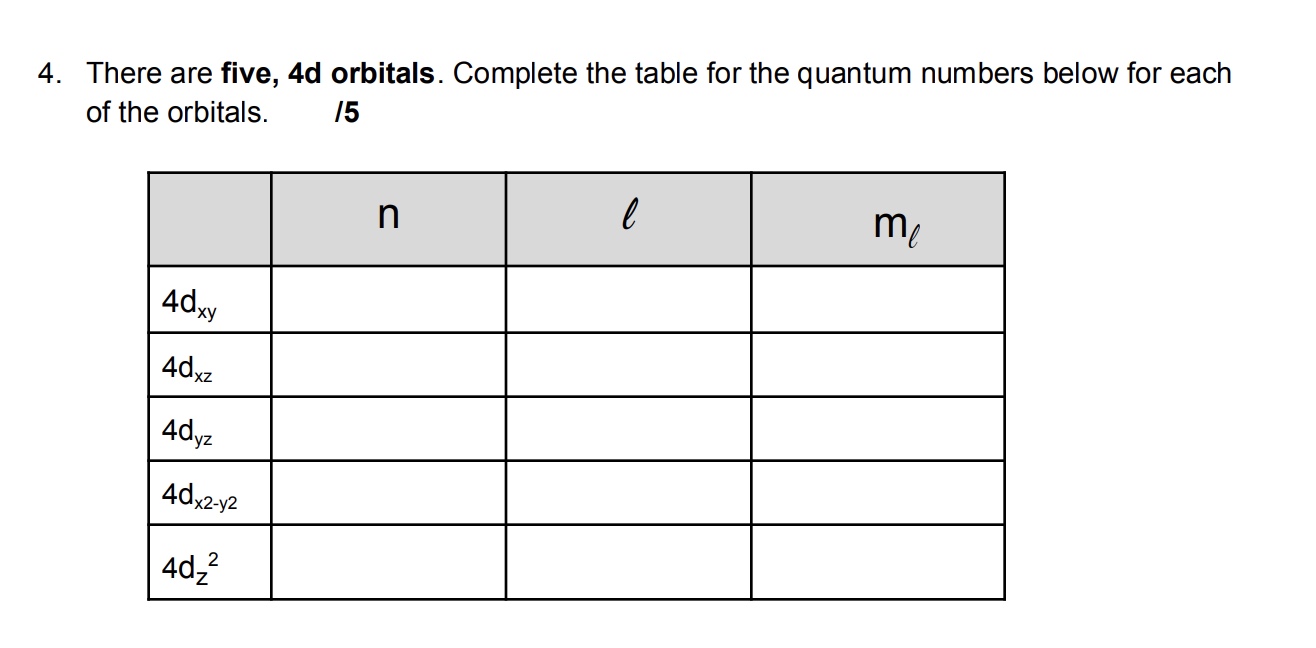
How Many 5s Orbitals Are There In An Atom

Hey there! So, you're wondering about these… 5s orbitals in an atom, huh? Like, how many of those little things are even rattling around in there? It's a good question, honestly. Sometimes these atomic concepts can feel like trying to count grains of sand at the beach, can't they? But don't sweat it, we'll get to the bottom of this, no problem. Grab your coffee, settle in, and let's chat about it.
First off, let's talk about what an orbital even is. It's not like a tiny planet orbiting a sun, okay? Forget that mental image. Think of it more like a probability cloud. It's where you're most likely to find an electron chilling out. So, when we talk about a 5s orbital, we're talking about a specific region in space around the atom's nucleus where electrons with a particular energy level hang out. Kinda like their own little designated hangout zone, you know?
And that '5' in '5s'? That's actually a pretty big deal. It tells us the energy level of the electron. The higher the number, the further away from the nucleus these electrons are, and the more energy they've got. So, a 5s electron is definitely not as close to the action as, say, a 1s electron. It's like being in the nosebleed seats versus the front row at a concert. Big difference in distance and energy!
Must Read
Now, about the 's' part. This tells us the shape of the orbital. Think of it like a style. 'S' orbitals are the simplest. They're basically spherical. Yep, just a big, round ball. Imagine a perfect little bubble. Easy enough, right? So, a 5s orbital is a spherical cloud of probability, just like a 1s or 2s or 3s orbital. The only difference is its size and its energy level. The 5s is just a much, much bigger bubble than the others.
So, here's the big reveal, the moment you've been waiting for: How many 5s orbitals are there? Drumroll, please…
There is only one 5s orbital.
Yep, that's it! Just one. It's like there's only one King of the hill for that particular energy level and shape. One spherical region where those 5s electrons love to be. It's not like there are a bunch of them scattered around. It’s a specific, single entity.
Think about it this way: If you're looking for a specific type of restaurant, say, a really good pizza place, you're probably looking for that one specific place that everyone raves about, right? You're not typically looking for multiple identical, amazing pizza places all over town. There's usually one standout. The 5s orbital is kind of like that standout pizza place for electrons at that energy level and shape.
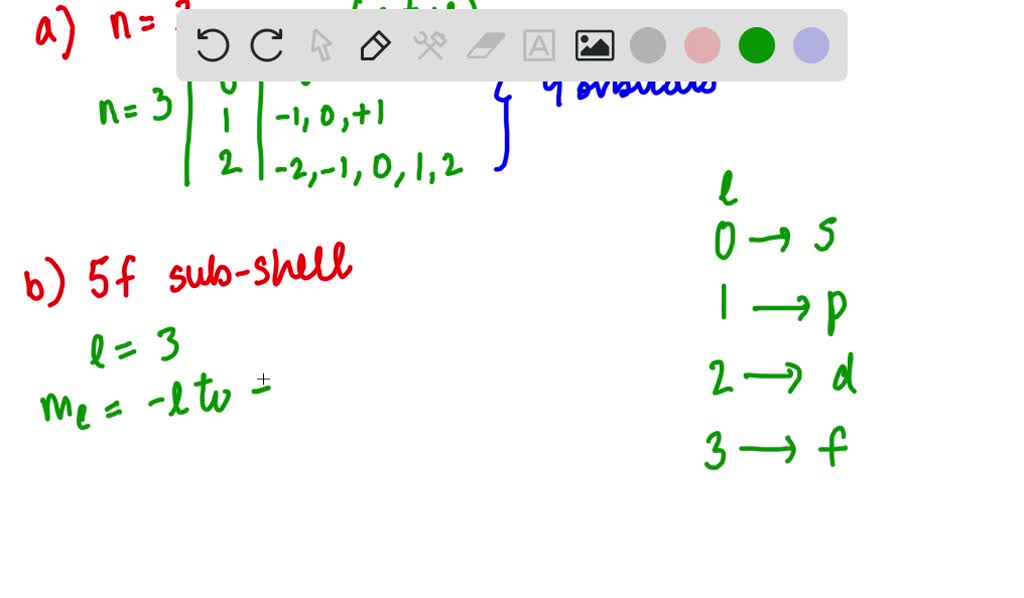
It's a common misconception, though! People often get confused because orbitals come in different types or subshells. You've got your s orbitals, your p orbitals, your d orbitals, and your f orbitals. And those come in different numbers. For example, you have one s orbital, but you have three p orbitals, five d orbitals, and seven f orbitals in higher energy levels. It’s like having different kinds of rooms in a hotel. You might have a lot of standard rooms (p orbitals, perhaps?), but only one presidential suite (the s orbital!).
So, when we’re talking about the 5s orbital, we're specifying both the energy level (the '5') and the shape (the 's'). And for that combination, there’s only one shape of that size. It’s a very precise description. Imagine you're ordering a very specific coffee. You don't just ask for "a coffee," right? You ask for a "large, soy, extra-shot, no-foam latte." You're being super specific, and you expect one of those. The 5s orbital is like that hyper-specific coffee order for electrons.

Let’s break down those other orbital shapes just to clear the air a bit. The 'p' orbitals? They're shaped like dumbbells. Think of two balloons tied together at the middle. And there are three of them, all pointing in different directions (like on the x, y, and z axes). So, if you have a 5p subshell, you'd have three 5p orbitals. The 'd' orbitals? They get a bit fancier. They have shapes that are kind of like clover leaves. And there are five of them! And the 'f' orbitals? Those are even more complex and there are seven of them. See? The number of orbitals definitely changes depending on the type (s, p, d, f).
But the 's' orbital is always the simplest. Always spherical. Always just one. It's the foundational orbital, really. The OG of electron habitats. It’s the most basic shape for an electron cloud at any given energy level. So, for the 5s, it’s just that one, big, spherical home for those energetic electrons.
Why is this important, you ask? Well, it helps scientists understand how atoms bond together. The arrangement and energy of electrons dictate all the chemistry we see. It's like knowing where all the furniture is in a house – it helps you understand how people move around and interact in it. And knowing there’s only one 5s orbital is a crucial piece of that furniture-placement puzzle.
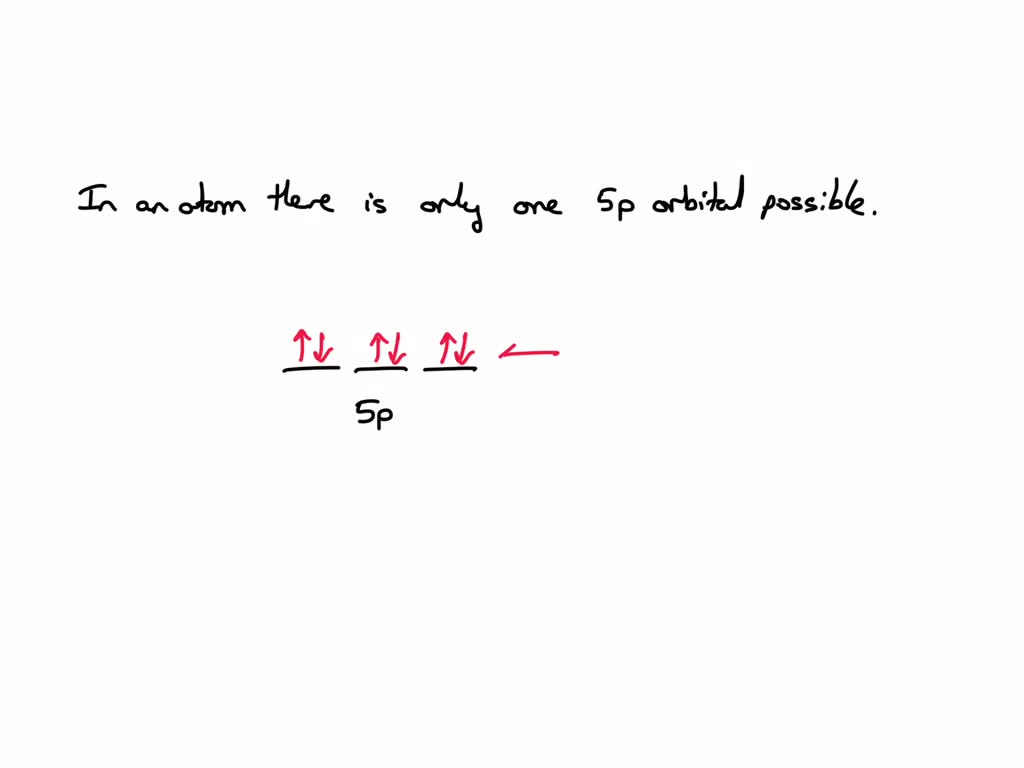
It’s also related to quantum mechanics, the super weird and wonderful science of the very, very small. These orbitals are a direct result of the solutions to the Schrödinger equation for a hydrogen atom, which, while simplified, gives us the fundamental understanding of electron behavior. It's where all those complex mathematical equations boil down to these simple shapes and energy levels. Pretty neat, huh?
So, to recap: the '5' tells you how much energy the electrons have and how far they are from the nucleus. The 's' tells you the shape, which is a sphere. And for that specific combination of energy and shape, there is always, without fail, exactly one 5s orbital. It’s a singular entity, a unique electron dwelling.
It’s not like there are a bunch of 5s orbitals floating around, waiting to be filled. It’s the 5s orbital. Think of it as a special seat. There's only one "premium view" seat at the very front row for the 5s level. All the 5s electrons that exist in an atom will reside within this single orbital. They might be spinning in different directions (that's the 'spin' quantum number, a whole other story!), but they're all within that same 5s spatial region.

It's kind of comforting, in a way, isn't it? To know that some things in the universe are so clearly defined. No ambiguity here. One 5s orbital. Period. It's not like, "Oh, maybe there are two, or three if you catch it on a good day." Nope. Always one.
So, next time you hear about a 5s orbital, you can confidently say, "Ah, yes! The solitary sphere of the fifth energy level! I know exactly how many of those there are." You'll be the atomic orbital expert at your next coffee chat. You can impress your friends with your newfound knowledge. Imagine their faces!
And the electron capacity for this single 5s orbital? It can hold a maximum of two electrons. Remember, electrons have opposite spins to occupy the same orbital. So, one spins up, the other spins down, and they're perfectly happy coexisting in their cozy 5s abode. It’s like a tiny, two-person apartment. Cozy, but functional.
So, there you have it. The mystery of the 5s orbital, solved! It’s not a trick question, no hidden numbers. Just a straightforward answer: one. Because in the world of atoms, sometimes the simplest things are the most fundamental. And the 5s orbital is definitely one of those fundamental, spherical things. Pretty cool when you think about it, right? Keep those questions coming!