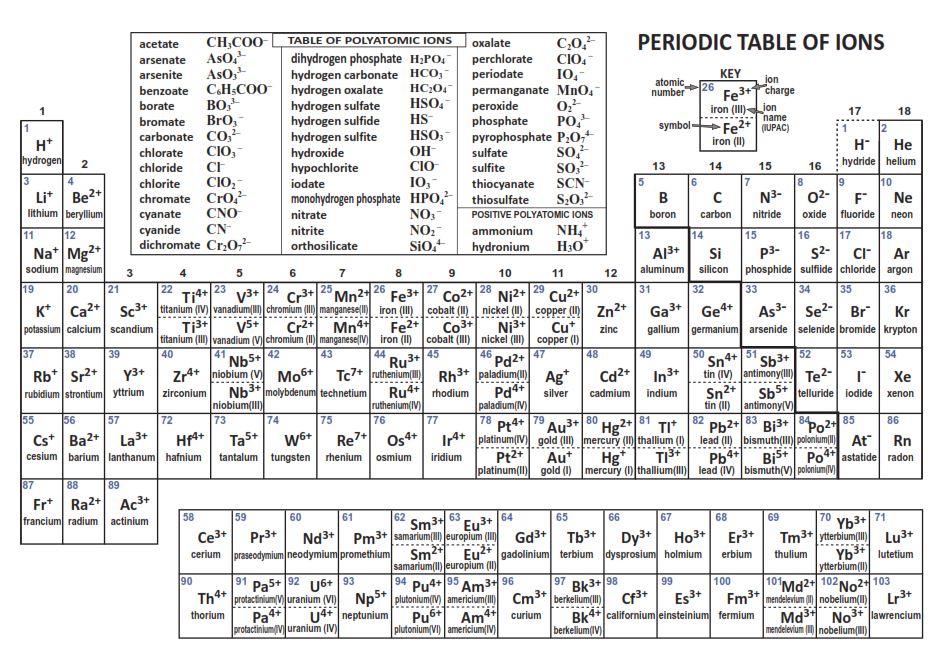
Click On The Ions Whose Names End In -ate.

Hey there, chemistry whiz-in-training! Or maybe you're just staring at your screen, wondering what all those squiggly letters mean. No worries, we've all been there! Today, we're diving into a super fun, super easy little game that’s actually a sneak attack on your chemical knowledge. Get ready to play: Click On The Ions Whose Names End In -ate!
Now, I know what you're thinking. "Ions? -ate? Is this some kind of secret code for baking a cake?" Well, sort of! Think of it like this: in the wild, wonderful world of chemistry, ions are like the tiny building blocks that make up everything. And some of these building blocks have names that end in "-ate." It's like a secret handshake for these particular ions. Once you know the handshake, you can spot them from a mile away (or, you know, across your computer screen).
So, what exactly are these "-ate" ions? They’re usually what we call polyatomic ions. Don't let the fancy name scare you! Polyatomic just means "many atoms." So, instead of a single atom chilling solo, these guys are a little crew, a team of atoms stuck together, all carrying a combined electrical charge. Think of them like a little chemical family, all holding hands and sharing a vibe (the vibe being that electrical charge, of course!).
Must Read
The really cool thing is that these "-ate" ions are everywhere. They’re in your food (hello, nitrates in leafy greens!), they’re in your cleaning supplies (ever heard of phosphates?), and they’re definitely going to pop up in any chemistry textbook you ever crack open. So, learning to recognize them isn't just for game-playing glory; it's like unlocking a secret level in real-life science!
Alright, let's get down to the nitty-gritty. How do we actually play this game? It's as simple as it sounds! You'll be presented with a list, or maybe a grid, of various ions. Your mission, should you choose to accept it (and you totally should, it's fun!), is to identify and click on only the ones whose names have that sweet, sweet "-ate" ending.
Imagine you're a detective, and the "-ate" ending is your magnifying glass. You’re looking for clues. Is it a sulfate? A carbonate? A nitrate? Bam! Found one! Move along, nothing to see here with the other ions. It’s a race against the clock, or a test of your keen observation skills. Either way, it's a brain-tickler without being a brain-melter. We want smiles, not strained expressions!
Let's talk about some common offenders, shall we? These are the "-ate" ions that you'll probably encounter the most. It’s like meeting the popular kids in the chemistry hallway. You gotta know their names!

The Usual Suspects: Your "-ate" Ion Squad
First up, we have sulfate. Its name sounds a bit… regal, doesn't it? Like it wears a tiny crown. The chemical formula for sulfate is SO42-. See? Four oxygen atoms clinging to a sulfur atom, and they’ve got a charge of negative two. They're a pretty popular bunch and show up in lots of places, from fertilizers to certain types of rocks.
Then there's carbonate. Think of all those fizzy drinks and seashells! Carbonate is CO32-. One carbon, three oxygens, and a negative two charge. It’s a fundamental ion in geology and biology, so definitely worth remembering its "-ate" status.
Next, let's shine a spotlight on nitrate. This little guy is NO3-. One nitrogen, three oxygens, and a negative one charge. Nitrates are super important for plants, which is why they’re often found in fertilizers. Just try not to confuse it with nitrite (NO2-), which has one less oxygen and doesn't end in "-ate." Sneaky, right?
What about phosphate? This one is PO43-. A phosphorus atom surrounded by four oxygen atoms, and a negative three charge. Phosphates are essential for life, forming the backbone of DNA and ATP (that's the energy currency of your cells, in case you were wondering!). They’re also used in detergents and food additives. So, yeah, important stuff!

And let’s not forget chlorate (ClO3-), bromate (BrO3-), and iodate (IO3-). These are part of the halogen family, just with more oxygen buddies. They all follow the same "-ate" naming convention. It's like they're all part of the same "-ate" club.
There are also some slightly less common but still important ones, like perchlorate (ClO4-) and permanganate (MnO4-). These have even more oxygen atoms than their "-ate" cousins. Think of them as the "extra-oxygen" versions. It’s like they went back for seconds of oxygen and still got charged!
Now, here’s a little trick for your brain. Many of these "-ate" ions are derived from what we call oxyacids. Remember those from chemistry class? Acids with oxygen in them? If you can remember the name of the acid, you can usually figure out the "-ate" ion. For example, sulfuric acid (H2SO4) gives you sulfate (SO42-). Carbonic acid (H2CO3) gives you carbonate (CO32-). Nitric acid (HNO3) gives you nitrate (NO3-). See the pattern? It's like a chemical family tree!
The key takeaway here is to really focus on that "-ate" ending. It's your golden ticket. When you see an ion name, just scan the end. Does it have the "-ate" sound? If yes, click it! If no, leave it alone. Don't get distracted by the first part of the name. It's all about the grand finale of the word!

What about the ions that don't end in "-ate"? We have ions like chloride (Cl-), bromide (Br-), iodide (I-), oxide (O2-), sulfide (S2-), and nitride (N3-). These are typically simple ions, meaning they consist of only one atom. They’re the solo artists of the ionic world. And then there are the polyatomic ions that end in "-ite," like sulfite (SO32-) and nitrite (NO2-). These are often the "less oxygen" cousins of the "-ate" ions. So, if you see "-ite," it’s a no-go for this particular game. It’s like trying to get into an exclusive club, and "-ate" is the password.
Think of it like a taste test. You’re sampling different flavors. Some are sweet "-ate" flavors, and others are… well, not. You only want the "-ate" ones! It's about being selective, about having a discerning chemical palate.
This game is fantastic for building up your ion recognition skills. The more you play, the more familiar these names will become. You'll start to see "sulfate" and just know it's an "-ate" ion without even thinking. It’s like learning to ride a bike; at first, it’s wobbly, but soon it’s second nature!
Here’s a pro-tip for you: sometimes, the ions might be presented with their charges, and sometimes they won't. Don't let the presence or absence of the charge throw you off. The game is specifically about the name. The charge is secondary information for this particular challenge. So, whether it’s SO42- or just SO4, if the name is "sulfate," you’re golden!

Let's imagine a scenario. You're presented with: * Chloride (Cl-) * Sulfate (SO42-) * Potassium (K+) * Nitrate (NO3-) * Oxide (O2-) * Carbonate (CO32-) What do you do? You scan the names! * "Chloride" - Nope, ends in "-ide." * "Sulfate" - Bingo! Ends in "-ate." Click! * "Potassium" - That's a metal ion, doesn't end in "-ate." Move on. * "Nitrate" - Yep! Ends in "-ate." Click! * "Oxide" - Another "-ide." Nope. * "Carbonate" - You got it! Ends in "-ate." Click! See? It’s like a treasure hunt, and the "-ate" ending is your treasure map. And the treasure? It's knowledge, my friend! The best kind of treasure!
Sometimes, the game might get a little tricky. You might see ions with similar-sounding names. For instance, you might see "sulfate" and "sulfite." Remember our rule: "-ate" is the magic word. "Sulfite" has one less oxygen and ends in "-ite." So, only "sulfate" gets the click in this scenario.
It's also a great way to practice distinguishing between different types of ions. You have simple cations (like Na+, K+), simple anions (like Cl-, O2-), and these polyatomic ions. The "-ate" ending is a strong indicator that you're dealing with a polyatomic ion, and a specific type of polyatomic ion at that.
Think of this game as a little mental workout. It’s low-impact, high-reward. You’re not breaking a sweat, but you’re definitely flexing those brain muscles. And the more you play, the stronger those muscles get. Soon, you’ll be able to spot these "-ate" ions with the speed and precision of a laser beam!
So, go forth and click those "-ate" ions with confidence! Embrace the challenge, enjoy the process, and remember that every click is a step towards becoming a chemistry superstar. You're not just playing a game; you're building a foundational skill that will serve you well in all your scientific adventures. Keep clicking, keep learning, and keep that wonderful, curious smile on your face. You’ve got this!