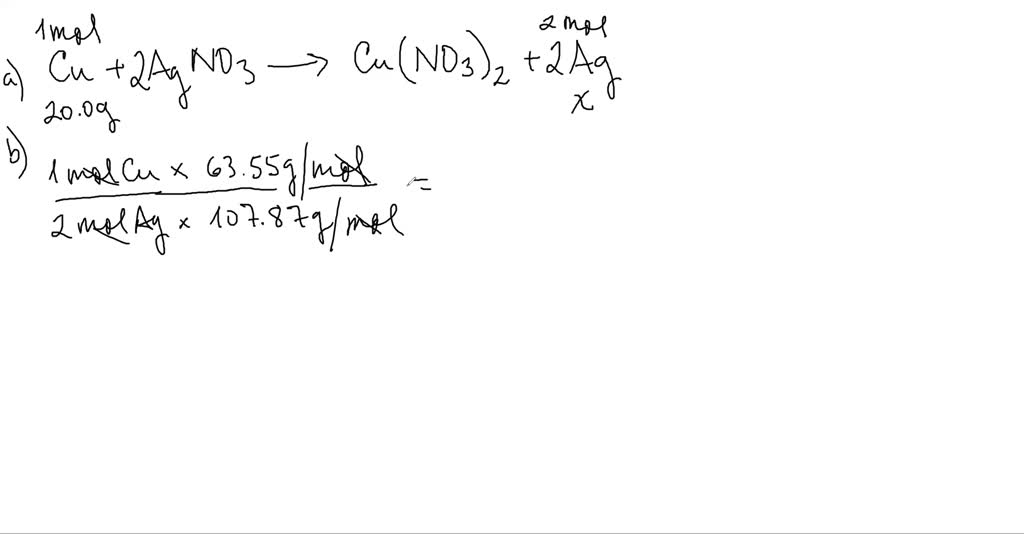Copper + Silver Nitrate Balanced Equation
Have you ever wondered what happens when you mix certain common household items and watch a cool reaction unfold? Chemistry, at its heart, is like a fascinating puzzle, and today, we're going to tackle a particularly neat piece of it: the balanced chemical equation for copper reacting with silver nitrate. Don't let the fancy name scare you! This is a wonderfully accessible topic that can be surprisingly fun and even a little bit magical to observe, and understanding it is easier than you think.
So, why bother with a balanced equation? Think of it as the recipe for a chemical reaction. Just like you need the right amounts of ingredients to bake a perfect cake, chemists need to know the exact proportions of substances to make a reaction happen predictably and safely. For beginners, learning about this equation is a fantastic introduction to the fundamental principles of chemistry. It teaches you about how atoms and molecules interact. For families looking for engaging science activities, observing this reaction (with proper supervision, of course!) can be a memorable and educational experience. Imagine seeing shiny new silver crystals "grow" right before your eyes! Hobbyists, especially those interested in electrochemistry or creating interesting visual effects, might find this reaction a great starting point for more complex projects.
Let's break down what's happening. When you place a piece of copper metal into a solution of silver nitrate, something visually stunning occurs. The copper atoms decide to team up with the nitrate part of the silver nitrate, leaving the silver atoms to fend for themselves. These freed-up silver atoms then start to clump together, forming beautiful, shimmering silver crystals. The copper, meanwhile, dissolves into the solution as copper ions. The balanced equation that describes this dance of atoms is:
Must Read
Cu(s) + 2AgNO₃(aq) → Cu(NO₃)₂(aq) + 2Ag(s)

Let's decode this simplified version: 'Cu' is our copper, 'AgNO₃' is our silver nitrate, 'Cu(NO₃)₂' is copper nitrate, and 'Ag' is our newly formed silver. The 's' means solid, and the 'aq' means dissolved in water. See how the numbers in front (the '2') ensure that we have the same number of atoms on both sides? That's the balancing act that makes the equation correct!
Want to see a variation? While this is the most common example, you could explore what happens if you used a different metal, like zinc, instead of copper. The fundamental principle of one metal displacing another from a salt solution remains, but the specific products and the speed of the reaction might change. Or, consider increasing the concentration of the silver nitrate – you'll likely see the silver crystal formation happen more rapidly!

Getting started with this is surprisingly easy. You can often find copper wire (like from an old electrical cord, stripped clean) and silver nitrate solution from science supply stores or even online. Make sure to wear safety goggles and gloves, and conduct the experiment in a well-ventilated area. You'll need a glass container to hold the silver nitrate solution and the copper piece. Observe the changes over time – it’s a slow but captivating process!
Ultimately, understanding the balanced equation for copper and silver nitrate isn't just about memorizing symbols and numbers. It's about appreciating the elegant order of the universe at a microscopic level. It’s a glimpse into how atoms rearrange themselves, creating something new and beautiful. It’s a small, accessible window into the vast and exciting world of chemistry, proving that even complex science can be both enjoyable and incredibly rewarding.