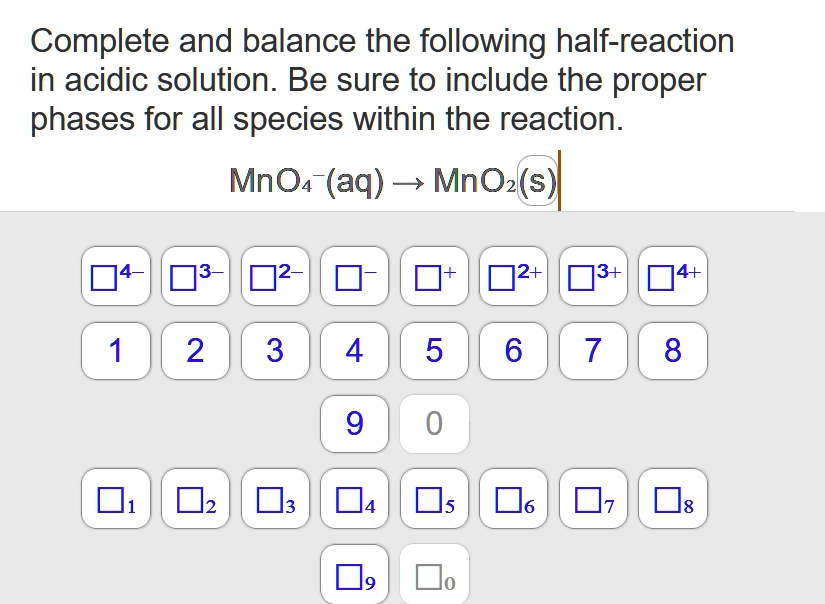
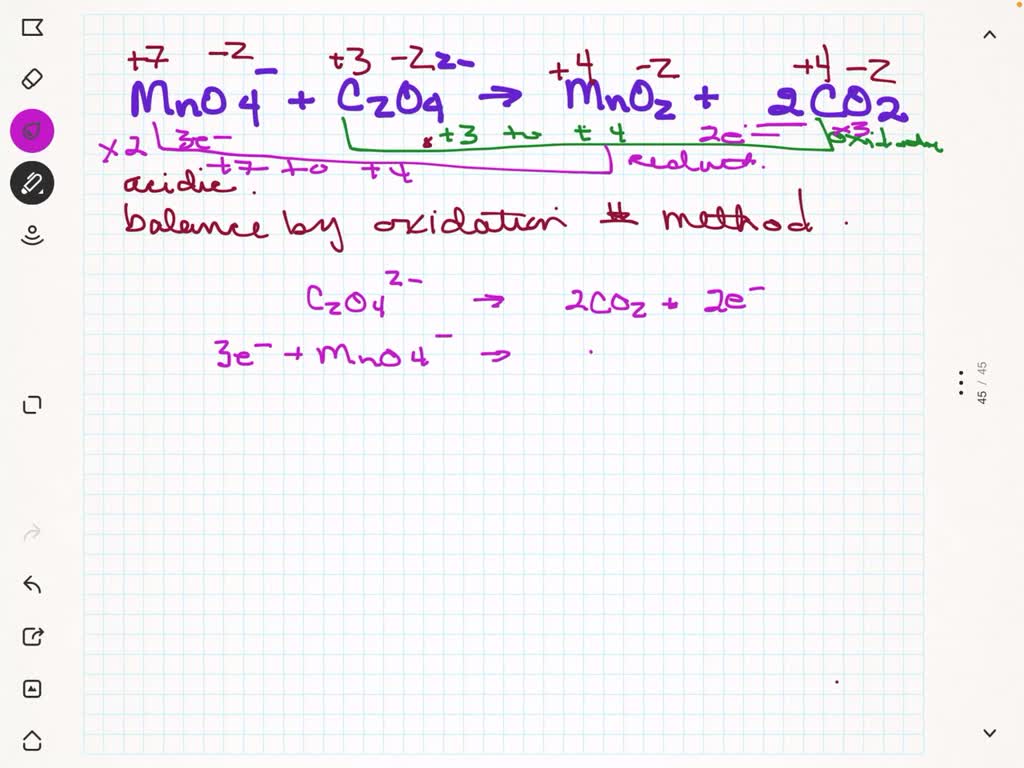
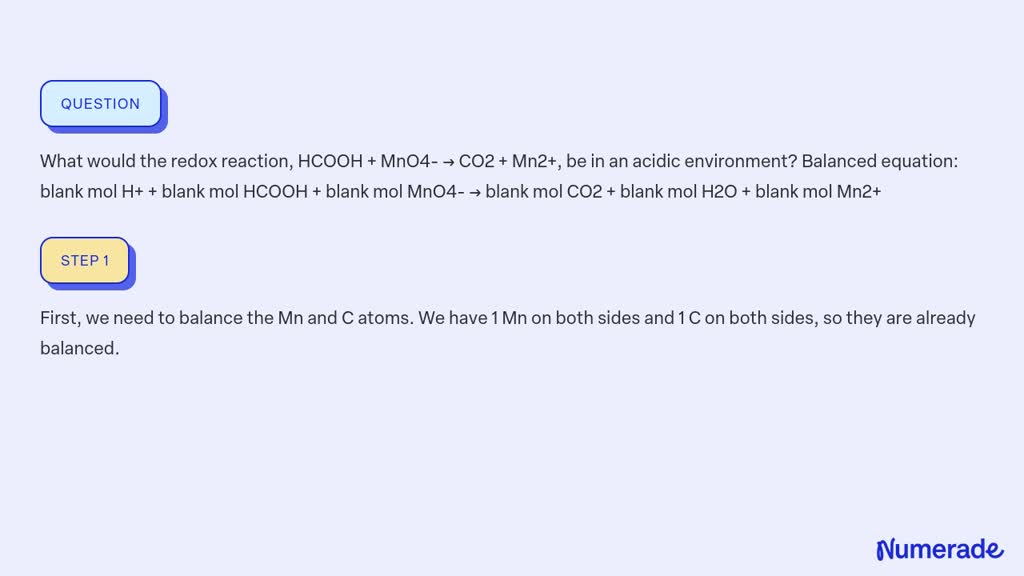
Hcooh + Mno4- Co2 + Mn2+ In Acidic Solution

Okay, let's dive into a little chemical drama. We're talking about a reaction that's got some serious personality. Think of it as a tiny, energetic party happening in a beaker.
We've got two main characters here. First up is Formic Acid, or Hcooh. It’s kind of the humble, everyday hero of the acid world.
Then we have Permanganate Ion, or Mno4-. This one’s the showstopper, the diva, the one with all the flair. It's got a deep, rich purple color that just screams "look at me!"
Must Read
When these two meet in an acidic solution, things get interesting. It's like they've been waiting for this moment. The environment is set, the stage is lit.
So, what happens? Well, Hcooh, our mild-mannered friend, decides to get a bit adventurous. It's going to shed its hydrogen atoms and transform.
And what does it turn into? Drumroll please… Carbon Dioxide, or Co2! Yes, that fizzy stuff you find in soda. It’s a bit of a glow-up for formic acid, going from a simple liquid to a gas.
Meanwhile, Mno4-, our glamorous permanganate, takes center stage. It’s been the oxidizer, the power player. It’s been giving electrons left and right.
But after all that effort, it needs a change of pace. It’s tired of being that intense purple. It’s ready for something more… mellow.
And what does Mno4- become? It transforms into Manganese(II) Ion, or Mn2+. This guy is pretty colorless. It’s the quiet one in the corner, after the party.
The whole process happens in an acidic solution. Think of the acid as the party planner, making sure everything is just right. It keeps the vibe upbeat and energetic.

It’s a redox reaction, fancy folks call it. Basically, one thing gives electrons, and another thing takes them. It’s a chemical trade, but way more dramatic.
I’ve always thought Hcooh + Mno4- in acidic solution is an underrated reaction. It doesn't get enough credit. It’s not as flashy as some others, maybe.
But there’s a certain elegance to it, don’t you think? The transformation of something simple into something gaseous. The reduction of a vibrant color into something almost invisible.
It's like a Cinderella story, but with molecules. Formic Acid goes from a humble liquid to a free-spirited gas. The Permanganate Ion sheds its dramatic persona for a more understated role.
And the whole thing is facilitated by the acidic solution. It’s the supportive friend, the encouraging coach. Without it, the magic wouldn’t happen.
Sometimes I feel like these reactions are like people. Some are loud and boisterous, demanding attention. Others are quiet but incredibly effective.
Mno4-, in its initial form, is definitely the attention-grabber. That purple is intense! It’s like it’s wearing a ballgown to a casual get-together.

But then it does its work. It oxidizes things, it gets things done. It’s a powerful agent.
And Hcooh? It's the unassuming one. You might overlook it. It’s just there, doing its thing.
But when the Mno4- comes around, it gets a chance to shine. It gets to release its inner gas. It becomes Co2.
It’s a beautiful partnership, really. A testament to how different substances can interact and change each other.
I know, I know. You’re probably thinking, "Who gets this excited about chemical reactions?" Well, I do. And I suspect, deep down, you might too.
Think about the sheer transformation. It’s not just a little tweak. It’s a complete makeover for both Hcooh and Mno4-.
The creation of Co2 is particularly satisfying. It's a gas that is so fundamental to our planet. It's in the air we breathe, it's in the bubbles of our drinks.

And the reduction of Mno4- to Mn2+ is equally fascinating. It’s a visual cue that the reaction has taken place. The purple fades, and a subtle change occurs.
It’s like watching a magic trick. Except, of course, it’s science. And science, in my humble, perhaps unpopular opinion, is the best kind of magic.
The role of the acidic solution is often overlooked. It’s the silent partner, the backbone of the operation. Without its acidic nature, the reaction might not proceed as smoothly, or at all.
It’s like the conductor of an orchestra. It ensures all the instruments are playing in harmony. It sets the tempo and the mood for the entire performance.
And when the reaction is complete, you're left with Co2 and Mn2+. Two distinct products, each with its own story.
The Co2 can bubble away, contributing to the atmosphere. The Mn2+, now a more stable ion, can go on to participate in other chemical processes.
It’s a cycle, a continuous flow of change. And it all starts with the simple, yet profound, interaction of Hcooh and Mno4-.
So next time you see a purple solution in a lab, or perhaps you're just contemplating the wonders of chemistry, remember this little duo. Remember the drama, the transformation, the sheer fun of it all.
It’s not just about balancing equations. It’s about understanding the personalities of these molecules. It’s about appreciating the hidden performances happening all around us.
The transformation from Hcooh to Co2 is a gas-tastic achievement. And the fading of the mighty Mno4- to the quiet Mn2+ is a testament to the power of reduction.
It’s a chemical ballet, a tiny dance of electrons and atoms. And it all happens in the invigorating embrace of an acidic solution.
So let’s give a round of applause, shall we? For Hcooh + Mno4- Co2 + Mn2+ in acidic solution. It’s a classic, a crowd-pleaser, and frankly, a bit of a genius.
Don't you agree? It’s like the quiet kid in class who turns out to be a brilliant artist or a witty comedian. You just have to look a little closer.
And that, my friends, is the entertaining, albeit slightly biased, take on this particular chemical spectacle. Now go forth and appreciate the drama!