Conjugate Acid Base Pairs Worksheet Answers

So, you’ve been staring at a worksheet. A conjugate acid-base pairs worksheet. Sounds a bit… intense, right? Like it’s gonna zap your brain with lightning bolts of pure chemistry knowledge. But guess what? It’s actually kinda cool. And the answers? Even cooler.
Let’s ditch the textbook vibe for a sec. Think of conjugate pairs like chemistry besties. They’re always hanging out together, just one tiny proton (that’s H+ for the uninitiated) away from being each other. Seriously, it’s a drama-free relationship.
When you're tackling those worksheet questions, you're basically playing detective. You're looking for that specific relationship. A molecule walks into a reaction. Boom. It either gives away a proton or it grabs one. That’s it. The thing left behind, or the thing that just made a new friend? That’s its conjugate.
Must Read
Imagine a super generous friend, let’s call him Acidy. He’s got an extra H+ and he’s like, “Here, take it!” The person who takes it? They’re the Base-y, and after they grab that proton, they become Acidy’s conjugate acid. It’s like getting a new superhero cape.
On the flip side, you have Base-y, who is totally ready to snatch a proton. When they do, the molecule that lost the proton? That’s the Acidy, and it becomes Base-y’s conjugate base. It’s like losing your favorite toy, but then getting an even cooler one.
The worksheet is just asking you to spot these proton handoffs. It's a game of "who gave what and who got what." Simple, right? Don't let the fancy terms scare you. Conjugate acid, conjugate base. It's just a fancy way of saying "the thing with the proton" and "the thing without the proton" in a pair.

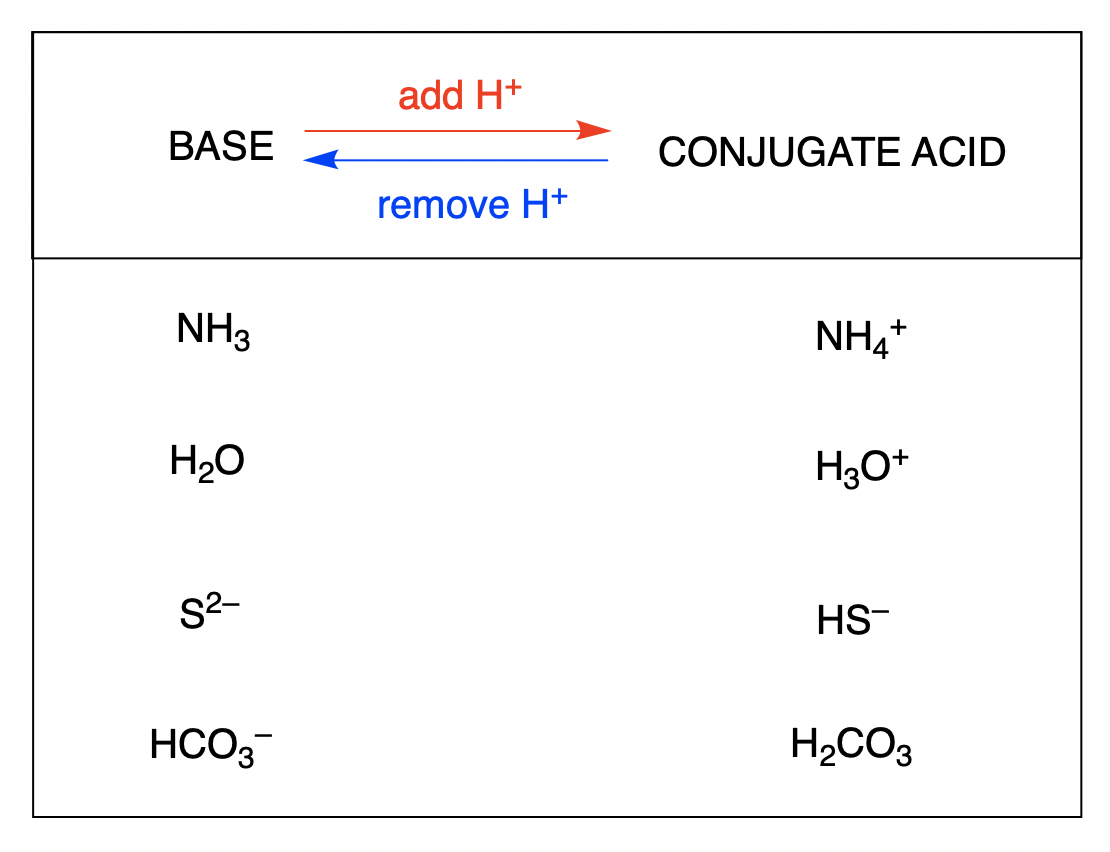
Let’s look at some classic examples. Water (H2O) is like the ultimate chameleon of chemistry. It can be an acid, or it can be a base. Wild, right? When water acts as an acid, it gives away a proton. What’s left? OH-. That’s the hydroxide ion. So, H2O and OH- are a conjugate pair. H2O is the acid, OH- is its conjugate base. Easy peasy.
But wait! There’s more! When water acts as a base, it grabs a proton. From what? Let’s say from a super strong acid. What does water become? H3O+. That’s the hydronium ion. So, H2O and H3O+ are another conjugate pair. This time, H3O+ is the conjugate acid, and H2O is the base. See? Water's got layers.
Think about it like this: a conjugate acid is what you get when a base picks up a proton. A conjugate base is what you get when an acid drops off a proton. It’s all about that proton’s journey. Is it being a giver or a taker? That’s the million-dollar question.
So, when you see a problem that looks like: HCl + H2O -> H3O+ + Cl-, you need to find the pairs. HCl gives a proton to H2O. So, HCl is the acid. H2O is the base. After the transfer, H3O+ is formed. That's the conjugate acid of H2O. And Cl- is left behind. That's the conjugate base of HCl.

See? You're not just filling in blanks; you're understanding how molecules interact. It’s like watching a tiny dance of atoms. One moves, another reacts. It's all connected.
Sometimes the worksheet might throw in something a little more complex, like NH3. Ammonia. This little guy is also a fantastic proton-grabber. If NH3 grabs a proton, it becomes NH4+. So, NH3 and NH4+ are a conjugate pair. NH4+ is the conjugate acid, and NH3 is the conjugate base.
Why is this stuff important? Well, it's the backbone of understanding acids and bases in solution. It helps us predict how reactions will go. It's fundamental for things like how your stomach acid works, or how buffers keep your blood pH stable. Pretty crucial stuff, even if it sounds a bit nerdy.
And let's be honest, the names are a little funny. Conjugate acid. Conjugate base. It sounds like something out of a sci-fi novel. "Prepare for the arrival of the Conjugate Acid, defender of protons!" Okay, maybe not quite. But you get the idea.
The fun part is realizing that these aren't just abstract concepts. They're happening all around you, all the time. In your coffee, in the rain, even in your own body. It’s a little secret world of chemical relationships.
When you’re checking your answers, don’t just look if you got it right. Ask yourself why it’s right. Did you correctly identify the proton donor and the proton acceptor? Did you correctly identify what's left over or what's newly formed?
Let’s say your worksheet has a question like: Identify the conjugate acid-base pairs in the following reaction: HSO4- + NH3 <=> SO4(2-) + NH4+
Okay, deep breaths. Look at HSO4-. It looks like it’s giving away a proton to NH3. So, HSO4- is the acid. What’s left after it loses that proton? SO4(2-). That’s its conjugate base. Your first pair: HSO4- / SO4(2-).

Now look at NH3. It’s accepting that proton. So, NH3 is the base. What does it become after it gets the proton? NH4+. That’s its conjugate acid. Your second pair: NH3 / NH4+.
Ta-da! You’ve just decoded a chemical interaction. It’s like being a translator for the molecular world. You’re making sense of their proton-trading habits.
The key takeaway from those worksheet answers is this: a conjugate acid-base pair differs by only one proton (H+). That’s your magic wand for solving these problems. One proton. That’s the secret handshake.
So, next time you see a worksheet on conjugate acid-base pairs, don’t groan. Smile. It’s a chance to be a chemistry detective, to understand the secret lives of molecules, and to realize that even complex science can have a playfully simple core. You're not just getting answers; you're getting a little peek behind the curtain of how the world works. And that, my friend, is pretty awesome.





