Chapter 6 Review Chemical Bonding Section 4

Hey there, awesome explorers of the universe! Ever looked at a shiny piece of metal, a fluffy cloud, or even a delicious cookie and wondered, "What's the secret sauce that holds all this amazing stuff together?" Well, buckle up, buttercups, because we're diving headfirst into the magical world of chemical bonding, specifically Chapter 6, Section 4 of your chemistry adventures! Don't worry, this isn't going to be your grandma's dusty textbook lecture. We're making this fun, we're making this inspiring, and we're going to see how understanding these tiny, invisible connections can actually make your life a whole lot more interesting!
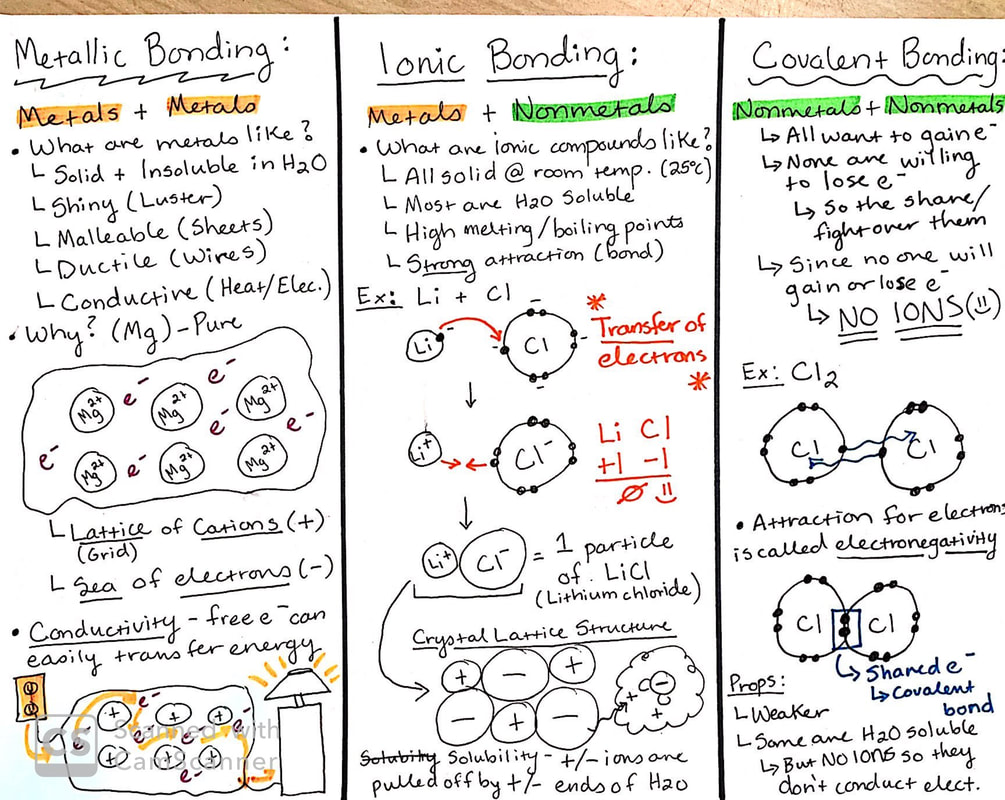
So, we've been chatting about how atoms, those tiny building blocks of everything, like to play nice (or sometimes, not so nice!) with each other. Remember how they're all about achieving that perfect, stable electron configuration? Think of it like atoms trying to find their perfect dance partners. And in our section today, we're zeroing in on a particularly groovy dance move called metallic bonding.
Now, you might be thinking, "Metallic bonding? Sounds a bit... dull. Like a robot convention." But hold your horses! This is where things get seriously cool. Imagine a bunch of metal atoms all hanging out together. Instead of one atom hogging all the electrons or reluctantly sharing them, in metallic bonding, it's like a giant, communal party! All the outer electrons from these metal atoms decide to ditch their original owners and become delocalized. What does that even mean, you ask? It means they're free agents, floating around like tiny, energetic dancers all over the place. Pretty wild, right?
Must Read
Think of it like a bustling city. The metal atoms are like the buildings, standing strong and tall. And the delocalized electrons? They're the citizens, zipping around, making connections, and keeping the whole place humming. This "sea of electrons," as chemists fondly call it, is the absolute key to why metals behave the way they do. It's the superpower behind their incredible properties!
The Superpowers of Metallic Bonding
Let's talk superpowers. What makes that shiny spoon in your kitchen so, well, spoon-like? Why can you bend that paperclip without it snapping? It all comes down to this amazing electron party! First off, metals are generally fantastic conductors of electricity. Why? Because those free-roaming electrons can easily hop from one atom to another when you apply a voltage. It's like opening a highway for electricity to zoom through! Isn't that neat? You're literally using a chemical bond to power your phone!

And what about thermal conductivity? Yep, that's another metallic superpower. Heat is basically just energy, and those energetic electrons are super efficient at transferring that energy throughout the metal. That's why your pot handle can get hot so quickly when you're cooking up a storm (sometimes a bit too quickly, am I right?). It’s the electrons doing their relay race!
But wait, there's more! Metals are also known for being malleable and ductile. Malleable means you can hammer them into thin sheets without them breaking. Think of those gold leaf decorations on fancy cakes – pure metallic magic! Ductile means you can pull them into wires. That's how we get all those essential electrical wires running through our homes and cities. How do the electrons make this possible? When you apply force, the metal atoms can actually slide past each other. The "sea of electrons" is so flexible that it accommodates this movement without the whole structure falling apart. It's like the electrons are saying, "No worries, friends, we'll just rearrange our dance floor!"
Imagine trying to bend a sugar cube or stretch a salt crystal. Snap! They'd crumble. That's because their bonding is much more rigid. Metallic bonding, on the other hand, is like a well-oiled machine, flexible and incredibly strong.
Making Life More Fun (Yes, Really!)
Okay, so how does this make your life more fun? Think about it! From the sleek aluminum foil that keeps your lunch fresh to the robust steel beams holding up skyscrapers, metallic bonds are everywhere, making our modern world possible. That bike you ride? Metallic bonds. The tools you use? Metallic bonds. The coins you use to buy that yummy treat? Metallic bonds!
Even in your favorite video games, the intricate circuitry inside your console relies on the conductivity of metals, thanks to metallic bonding. It’s the silent, unsung hero of so many things we take for granted. Understanding this allows you to appreciate the engineering marvels around you. You can look at a bridge and think, "Wow, those metallic bonds are really doing some heavy lifting!" It adds a whole new layer of wonder to the everyday.
And it's not just about the big stuff. Think about the joy of creating! If you're into art or crafts, working with metal – even simple things like wire sculpting – becomes more intuitive when you understand its properties. You can bend, shape, and connect it with a newfound appreciation for its inherent nature.

Furthermore, this knowledge sparks curiosity. Once you understand metallic bonding, you might start wondering about other types of bonds, like ionic and covalent. Each one has its own unique "dance," leading to an astonishing variety of materials with different properties. The world becomes a giant chemistry experiment waiting to be explored!
So, as you wrap up Chapter 6, Section 4, take a moment to celebrate. You've just unlocked a fundamental secret of the universe: how metals hold together and why they're so darn useful and awesome. It’s not just about memorizing facts; it’s about understanding the invisible forces that shape our reality.
Don't stop here! This is just the beginning of your chemical adventure. Keep asking questions, keep exploring, and keep marveling at the incredible complexity and beauty of the world around you. The more you learn, the more you'll see the magic woven into every atom, every bond, and every single thing you encounter. Go forth and be curious, my friends. The universe is waiting for you to discover its secrets!