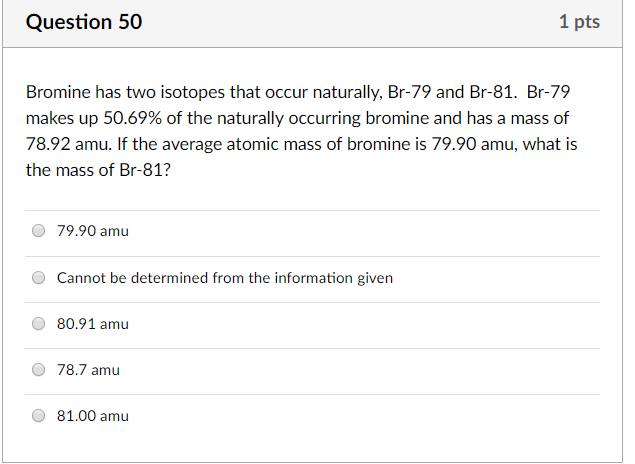
Bromine Has Two Naturally Occurring Isotopes

Imagine your favorite tasty treat, maybe a perfectly grilled burger or a warm, gooey chocolate chip cookie. Now, picture that treat having a little twin sibling, almost identical but with a tiny, almost invisible difference. That's a bit like what's going on with a fascinating element called bromine. It’s a bit like a celebrity with two well-loved, slightly different versions of themselves walking around.
This element, bromine, is pretty cool. It’s a reddish-brown liquid at room temperature, which is already a bit unusual for an element. Think of it as the element that likes to stay a little bit fluid, like a personality that's always on the move. It’s used in lots of things, from helping to make your medications work better to keeping swimming pools sparkling clean.
But the real story, the one that’s like finding out your favorite actor has a super-secret, equally awesome twin, is about bromine's isotopes. These aren’t just random variations; they are two naturally occurring versions, like two siblings born from the same family tree. Scientists have given them special names to tell them apart, even though they look and act pretty much the same.
Must Read
The two stars of our show are Bromine-79 and Bromine-81. You can think of the numbers, 79 and 81, as their birth order or maybe just a way to remember which one is which. It’s like having two best friends, one named Alex and the other named Al, and you just know who’s who after a while. They are the two natural faces of bromine, and they’ve been around since the universe decided to get creative.
So, what’s the big deal about these numbers, 79 and 81? It all comes down to something called neutrons. Inside every atom of bromine, there’s a central part called the nucleus. This nucleus is like a tiny playground with two kinds of characters: protons and neutrons.
All bromine atoms have the same number of protons. That’s what makes them bromine in the first place, kind of like having the same last name. But the number of neutrons can be a little different. It’s like having the same family features but with slightly different hair colors or heights.

Bromine-79 has 34 protons (that's the bromine part!) and 44 neutrons. Add them up, and you get 79. Simple, right? Then there’s Bromine-81. It also has those 34 protons, keeping it firmly in the bromine family. But this sibling comes with 47 neutrons. Add those up, and voila, you get 81!
This small difference in neutrons is the only thing that truly sets Bromine-79 and Bromine-81 apart in terms of their atomic weight. But don't let that fool you! For all intents and purposes, in the chemical world, they are practically identical twins. They behave the same way when they meet other elements, forming bonds and going on chemical adventures.
It’s like having identical twins who are both amazing at playing the piano. You might be able to tell them apart by a tiny mole or a slightly different smile, but when it comes to their musical talent, they are equally brilliant. Bromine-79 and Bromine-81 are like that in the world of chemistry. They both participate in the same reactions, they both enjoy the same chemical company.
The universe, in its infinite wisdom and perhaps a touch of playful mischief, decided to give us these two forms of bromine. They are found together in nature, mixed in a way that’s been happening for billions of years. It’s not like one is rare and the other is common; they are both present in significant amounts, each contributing to the overall presence of bromine on Earth.

Think about it: when you encounter bromine in a product, whether it's in your photo developing chemicals (yes, old-school photography used a lot of bromine!) or in flame retardants that help keep us safe, you're interacting with a blend of these two natural siblings. It’s a team effort, a dynamic duo that works together.
Scientists love to study these isotopes. They are like tiny clues to understanding the history of the universe and how elements are formed. The precise ratio of Bromine-79 to Bromine-81 found in different rocks or meteorites can tell us fascinating stories about where they came from and the conditions they experienced. It's like finding two different versions of the same family photo from different eras, each telling a part of the story.
For instance, by analyzing the relative amounts of Bromine-79 and Bromine-81 in ancient samples, geologists can learn about the conditions in the early solar system. It's a bit like forensic science, but on a cosmic scale! These isotopes are silent witnesses to billions of years of cosmic evolution.

And here’s a fun thought: sometimes, elements can even have versions that aren’t stable. These are called radioactive isotopes. But our Bromine-79 and Bromine-81 are the lucky ones! They are perfectly stable, meaning they don’t break down and release energy over time. They are in it for the long haul, content to just be bromine.
So, the next time you hear about bromine, remember its dual nature. It’s not just a single entity; it’s a partnership, a duo of Bromine-79 and Bromine-81, working together. They are the unsung heroes in many of the materials we use, living their stable, quiet lives, contributing to the world without much fanfare.
It’s a beautiful reminder that even in the seemingly simple world of elements, there are layers of complexity and fascinating stories waiting to be discovered. The fact that bromine has these two natural, stable isotopes is a little marvel of the natural world. It’s like a constant, subtle harmony that underlies its presence everywhere.
So, whether you’re thinking about science, nature, or just the idea of having a favorite twin, bromine offers a fun little glimpse into the world of isotopes. It’s a reminder that even the things we take for granted have their own unique and sometimes surprisingly interesting stories to tell. These two siblings, Bromine-79 and Bromine-81, are a testament to the elegant variety that exists all around us.

They are the quiet backbone of many applications. They are the dependable duo that helps make our lives a little bit safer and our world a little bit more functional. And the best part? They are just naturally, happily themselves, existing side-by-side, two versions of a truly remarkable element.
Think of them as the element equivalent of a classic, beloved movie that has both a cherished original and an equally enjoyable remake. Both are fantastic, both contribute to the legend, and both are simply part of the joy. That’s the magic of bromine’s isotopes.
So, let’s raise a metaphorical glass to Bromine-79 and Bromine-81! They’ve been around the block, seen it all, and continue to be an essential part of our world, all while keeping their very minor, yet significant, differences. They are the perfect example of how nature loves to have a little bit of variety, even within something as fundamental as an element.
And isn’t that just wonderful? That something as seemingly simple as bromine has this subtle, enduring story of duality, a quiet testament to the endless creativity of the universe? It’s these little discoveries that make learning about science so enjoyable and, dare we say, heartwarming.