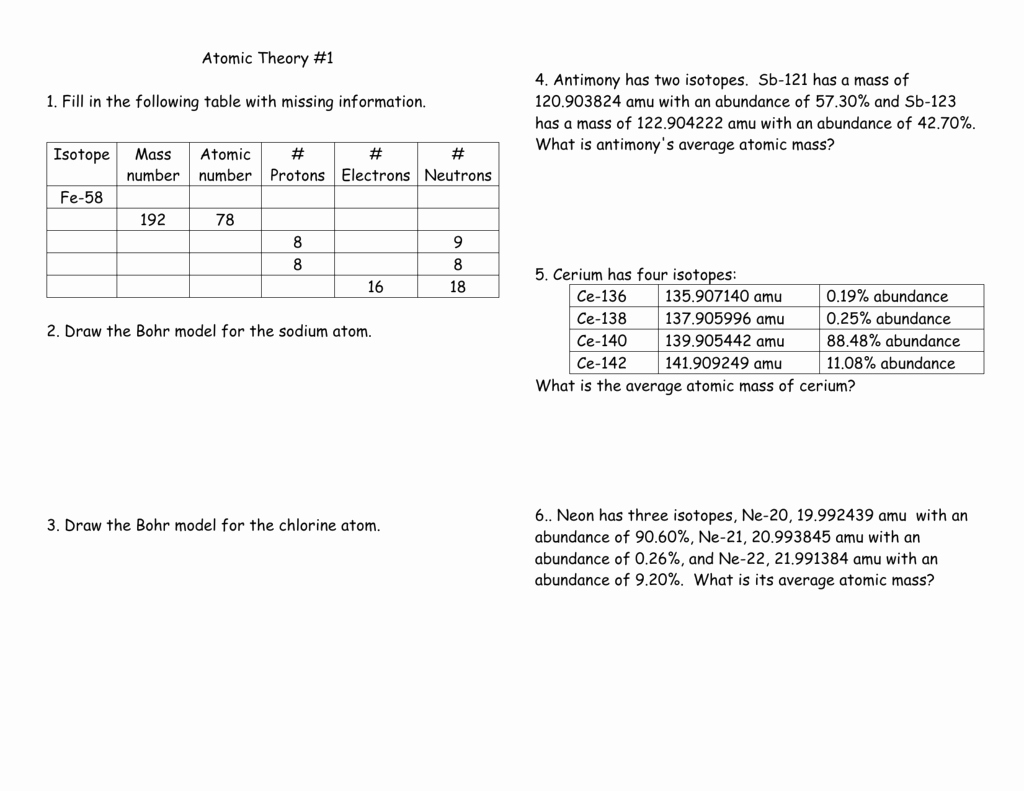
Average Atomic Mass Worksheet Show All Work

Hey there, fellow humans! Ever stare at a periodic table and feel a little… mystified? Like, what's with all those numbers floating around those cool elemental symbols? Today, we're going to demystify one of those numbers in particular: the average atomic mass. And don't worry, we're not going to break out any super complicated calculus or anything. Think of this more like figuring out the average age of your favorite band members to get a general vibe of their experience level. It's about making sense of things that seem a bit abstract!
So, what exactly is average atomic mass? Imagine you're baking a batch of your famous cookies. You've got a bunch of chocolate chips, right? Some are milk chocolate, some are dark chocolate. Now, if you were to just grab a handful and try to guess the average sweetness of those chips, you'd probably think about how many of each kind you have and how sweet they are individually. That's kind of what scientists do with atoms, but instead of sweetness, they're looking at mass.
Unpacking the "Average" Part
The "average" part is key here. Atoms of the same element aren't always exactly the same. They can have slightly different numbers of neutrons, which affects their mass. These variations are called isotopes. Think of it like siblings. They're all part of the same family (the same element), but they might have different heights or hair colors (different masses due to varying neutron counts).
Must Read
For example, let's take carbon. The most common type of carbon we encounter is carbon-12, which has 6 protons and 6 neutrons. But there's also carbon-13, with 6 protons and 7 neutrons. Carbon-14, the one used for radiocarbon dating, has 6 protons and 8 neutrons. They're all still carbon, but they weigh a little differently. If you just said "carbon's mass is 12," you'd be missing out on the whole picture!
So, scientists don't just pick one type of atom to represent the element. Instead, they look at all the naturally occurring isotopes and how abundant they are. They then calculate a weighted average. It's like if you have 10 friends, and 8 of them are 20 years old, and 2 are 30 years old. The average age isn't just (20+30)/2 = 25. It's more like (8 * 20 + 2 * 30) / 10, which is (160 + 60) / 10 = 22. The older friends contribute more to the "average" because there are more of them.
Why Should We Care About This Slightly Heavier Average?
Okay, so why bother with this average atomic mass thing? It might seem like a nerdy detail, but it's actually super important! Think about it like this: if you're building something, you need to know the average weight of your materials to make sure everything is stable, right? Scientists do the same thing when they're working with elements.
For Chemists: The Foundation of Calculations

For chemists, average atomic mass is like the LEGO brick of the periodic table. It's the fundamental number that helps them figure out how much of something they have, how much of it they need, and what will happen when different elements combine. When you see a chemical formula like H₂O (water), the numbers associated with hydrogen (H) and oxygen (O) are their average atomic masses. So, if a chemist needs to make a certain amount of water, they'll use these masses to calculate exactly how much hydrogen and oxygen they need to combine. It's like following a recipe where you need precise measurements!
Imagine you're a baker and you're making a huge wedding cake. You need to know the exact weight of your flour, sugar, and eggs to get the proportions just right. If you only guessed the weight of your ingredients, your cake might turn out too dense, too crumbly, or just plain weird. Average atomic mass gives scientists that precise information for the atomic-scale ingredients.
For Everyone Else: Understanding the World Around Us
Even if you're not a chemist, understanding average atomic mass helps you appreciate the world in a cooler way. It's part of the reason why different materials have different properties. Why is gold so dense and shiny? Why is aluminum so light and bendy? These properties are linked to the atoms that make them up, and their average masses play a role in how those atoms behave and interact.
Think about your phone. It's made of all sorts of elements, each with its own average atomic mass. These masses influence how the metals conduct electricity, how the silicon chips process information, and how the glass screen stays intact (mostly!). So, the next time you're scrolling through your feed, you can secretly nod to yourself, thinking, "Yup, that's all thanks to some nicely averaged atomic masses!"

Showing Your Work: The "How-To"
Now, let's get to the nitty-gritty of how you'd actually figure out this average atomic mass, especially if you're given a worksheet. This is where the "show all work" instruction comes in, and it's all about proving you understand the process. It's like showing your steps when you're trying to solve a math problem – it helps you and anyone looking at your work understand your thinking.
Typically, you'll be given information about the different isotopes of an element and their relative abundance (how common they are). You might see something like:
- Isotope A: Mass = X, Abundance = Y%
- Isotope B: Mass = Z, Abundance = W%
To calculate the average atomic mass, you'll do the following:
Step 1: Convert Percentages to Decimals. Since abundance is usually given as a percentage, you'll need to divide each percentage by 100 to turn it into a decimal. So, 50% becomes 0.50, 25% becomes 0.25, and so on. This is like converting a price in dollars and cents to a decimal number for easier multiplication.

Step 2: Multiply Mass by Abundance. For each isotope, you'll multiply its mass by its decimal abundance. This gives you the "contribution" of that specific isotope to the overall average. Think of it as weighing each type of chocolate chip by how many you have of it.
Step 3: Add Them All Up. Finally, you'll add up the results from Step 2 for all the isotopes. This sum is your average atomic mass!
Let's do a quick, pretend example. Let's say we have a made-up element called "Glowium."
- Glowium-10: Mass = 10.0 amu, Abundance = 70%
- Glowium-11: Mass = 11.0 amu, Abundance = 30%
Show your work!
Step 1: Convert abundances to decimals.

- Glowium-10 abundance: 70% / 100 = 0.70
- Glowium-11 abundance: 30% / 100 = 0.30
Step 2: Multiply mass by abundance.
- Glowium-10 contribution: 10.0 amu * 0.70 = 7.0 amu
- Glowium-11 contribution: 11.0 amu * 0.30 = 3.3 amu
Step 3: Add the contributions.
- Average Atomic Mass of Glowium = 7.0 amu + 3.3 amu = 10.3 amu
See? Not so scary! The "show all work" part just makes sure you're not just pulling a number out of thin air. It's like showing your friend how you figured out the average score of your video game matches – it makes the result more trustworthy and understandable.
The Takeaway: It's All About Balance
So, the next time you see that number next to an element on the periodic table, remember it's not just a random digit. It's a carefully calculated average atomic mass, a weighted blend of all the different forms that atom can take. It's a testament to how scientists observe, measure, and then make sense of the complex world around us.
It’s this kind of understanding that allows us to create new medicines, build incredible technologies, and even understand the age of ancient artifacts. It’s all about balance, about understanding the whole picture by considering all the parts. So, go forth and tackle those average atomic mass worksheets with a smile! You're basically becoming a master of atomic-level cookie mixing!