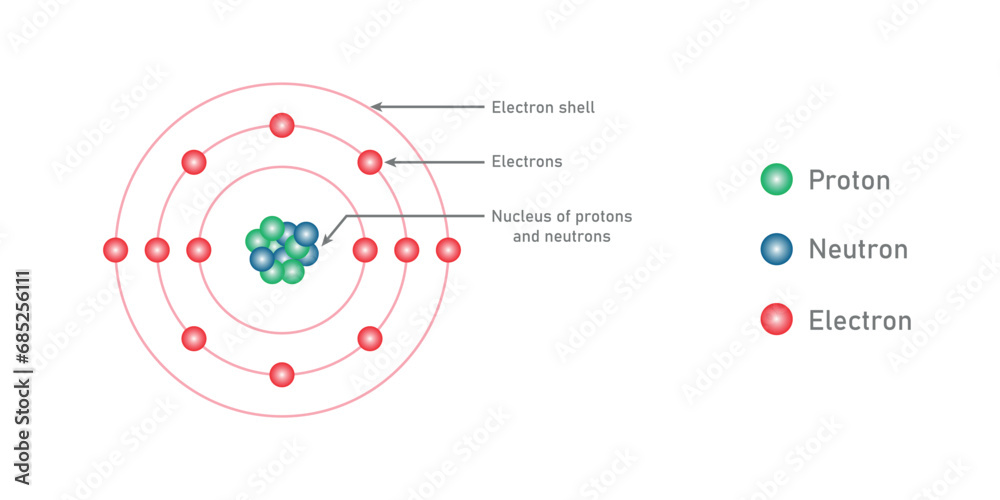
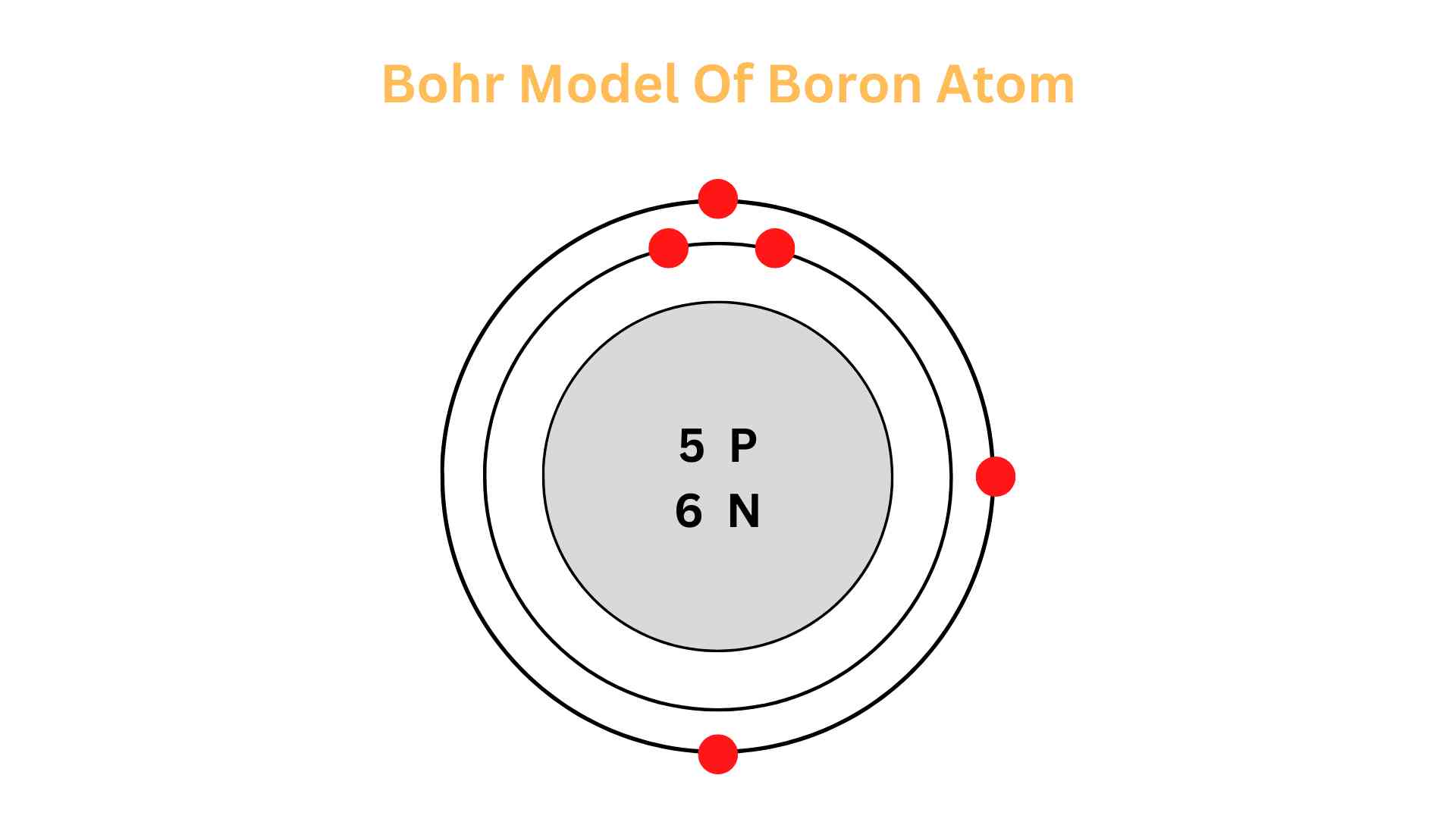
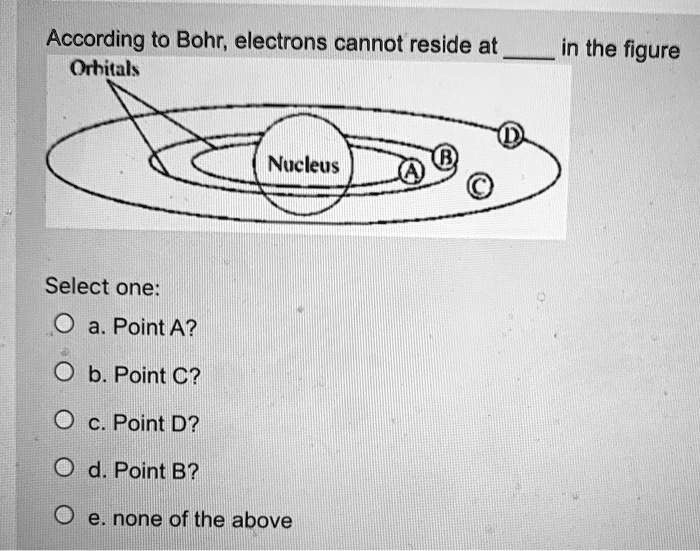
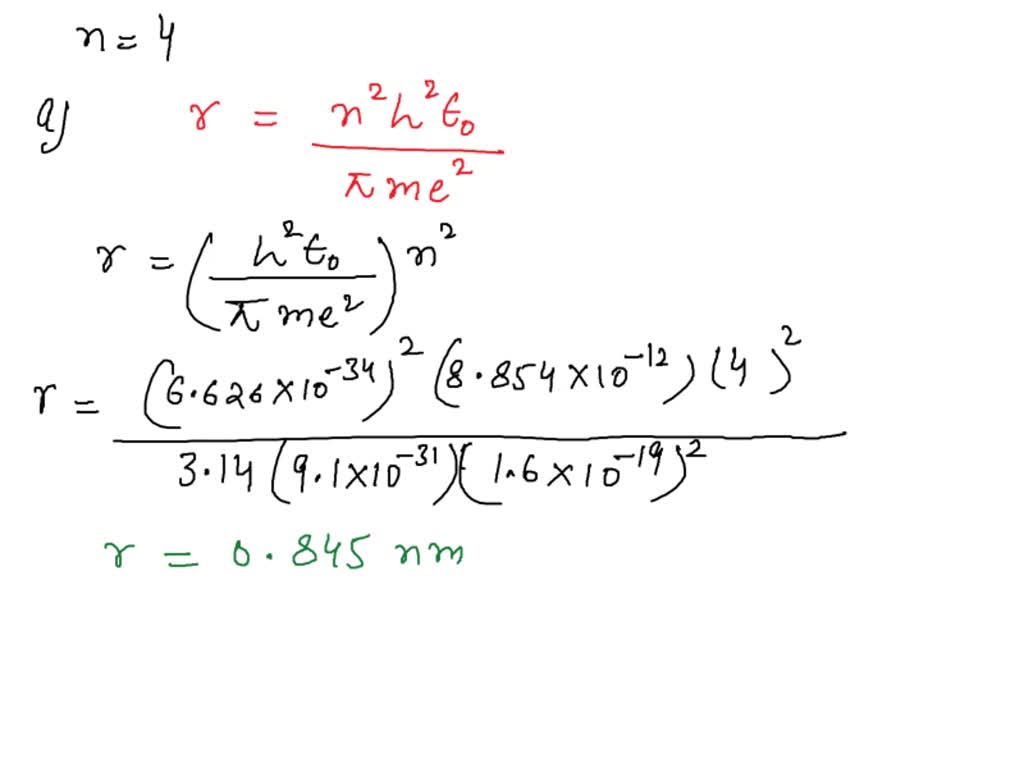According To Bohr Electrons Cannot Reside At
You know how sometimes you feel like you're just meant to be in a certain place? Like, that's your spot, your comfy chair, your favorite park bench? Well, it turns out that for tiny, energetic little things called electrons, there are some spots they absolutely cannot hang out in. It's like they have their own secret, off-limits zones in the world of atoms! And the brilliant mind who figured this out was a super cool scientist named Niels Bohr.
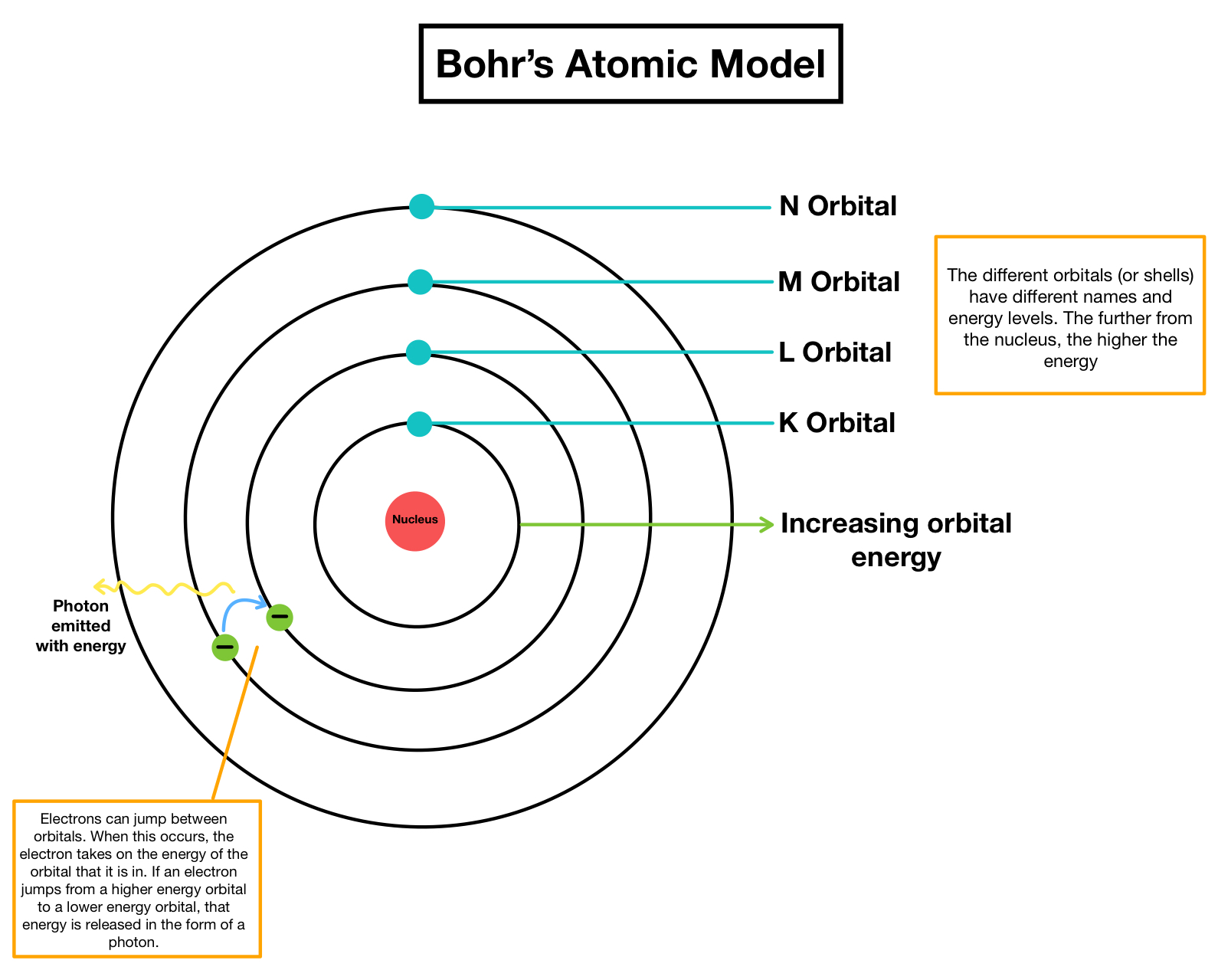
Think about it like this: imagine atoms are like a mini-city. In this city, there are buildings, but they're not just any old buildings. They're like special levels or floors where the electrons can live. Bohr's big idea was that electrons can only be on these specific floors. They can't just float around anywhere they please. It's a bit like having elevators that only stop at certain floors. You can't just dial a floor that doesn't exist!
So, what does this mean for us regular folks trying to wrap our heads around it? It means that the universe, at its very smallest level, is incredibly organized and has some super strict rules. It's not some chaotic free-for-all. These electrons, these little bundles of energy, have their designated "neighborhoods." They can jump from one floor to another, but only by taking a specific kind of leap, and they have to land on one of the allowed floors. They can't just decide to chill in the hallway between floors, or in the stairwell. Nope!
Must Read
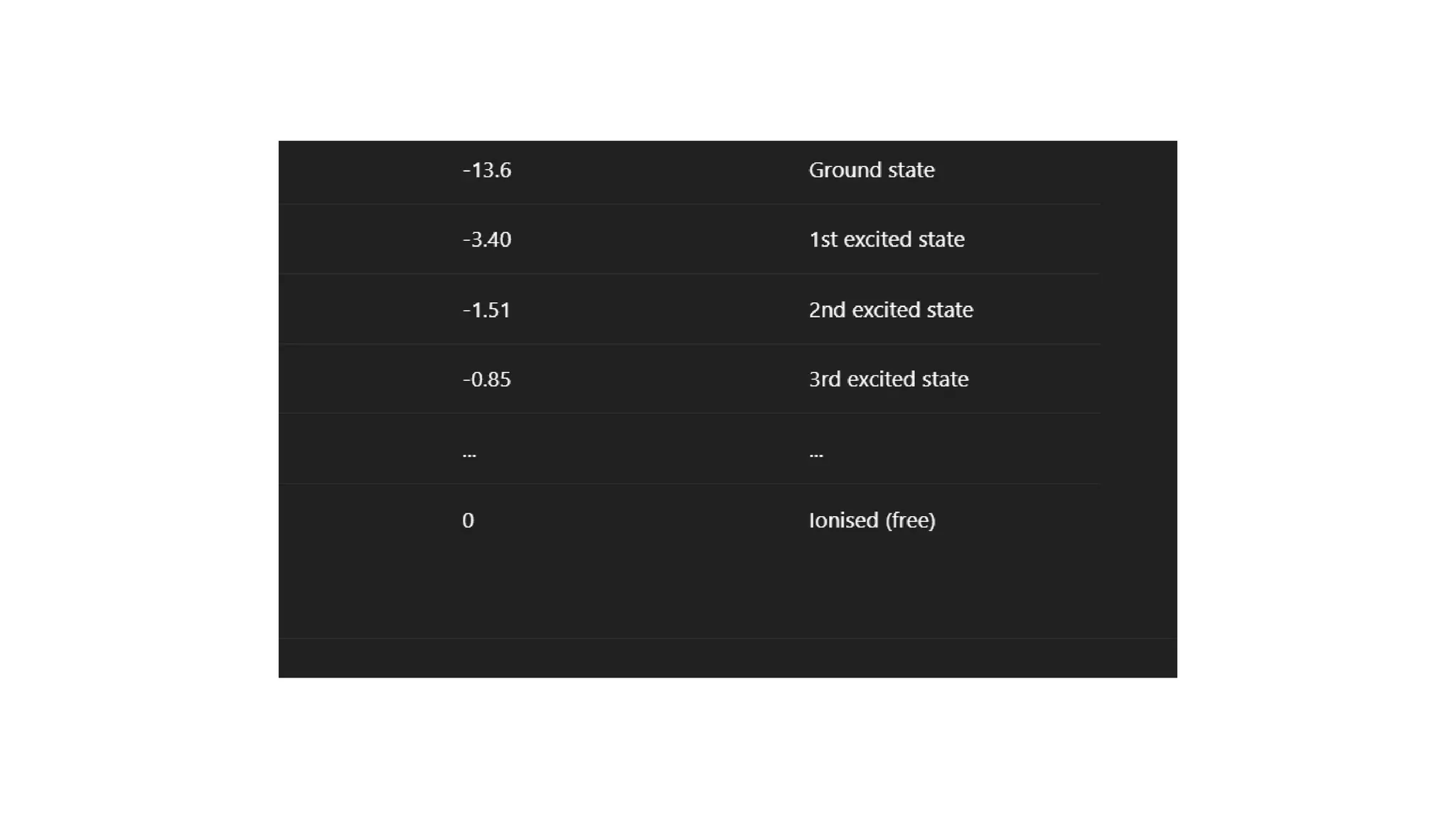
This whole concept is seriously mind-blowing when you stop and think about it. It's like discovering that your house only has rooms on specific levels, and you can't just create a new room in the middle of the wall. You have to be on the first floor, or the second floor, or maybe the attic. You can't be, say, halfway between the first and second floor. That's the "off-limits" territory for our electron friends.
And the reason this is so fascinating is because it's so different from how we experience the world every day. We can walk anywhere in our homes, we can stand on any part of the ground. But the quantum world, the world of atoms and electrons, plays by a completely different set of rules. It's a hidden universe with its own quirky physics, and Bohr's insight helped us peek into it.
So, why is it entertaining? Because it’s like a secret code! It’s like learning that there’s a hidden level in your favorite video game that you never knew existed. It sparks your imagination and makes you wonder about all the other hidden rules and patterns in the universe. It’s not a dry textbook explanation; it’s a glimpse into a bizarre and wonderful reality. It’s like saying, "Hey, guess what? Electrons can't stand here!" and your brain immediately goes, "Wait, where can they stand? And why not there?"
What makes it special is that Niels Bohr didn't just pull this out of thin air. He was looking at experiments, at the way atoms behaved, and he noticed something strange. When atoms got excited, they would give off light, but only specific colors of light. It was like they were singing songs, but only with certain notes. Bohr realized that these specific notes, these specific colors, were directly related to the electrons jumping between their allowed floors. Each jump corresponded to a specific packet of energy, which in turn, created a specific color of light.
It's like a cosmic concert where the electrons are the musicians, and they can only play certain notes on their atomic instruments. They can't hit a random note; it has to be one of the allowed ones!
This idea, that energy comes in little packets, or "quanta," was revolutionary. Before Bohr, scientists thought energy was like a smooth, continuous flow, like water from a faucet. But Bohr showed that it's more like a dispenser that only gives out drops of water. And the electrons? They're the ones who decide which size of drop they're going to get or give away when they move between their special energy levels.
The fact that electrons cannot reside at certain places is a fundamental building block of everything we see. It's why chemistry works the way it does. It's why stars shine. It's why your phone screen lights up. All of these incredible phenomena are rooted in these simple, yet profound, rules about where electrons can and cannot be.

It's a bit like knowing that a chef can't just throw any random ingredients together and expect a Michelin-star meal. There are rules of flavor, of combination, of technique. Similarly, in the atomic kitchen, electrons have their designated spots, and their movements between these spots dictate the "flavors" of energy and light that atoms produce.
So, next time you hear about atoms or electrons, remember Niels Bohr and his brilliant idea. Remember that these tiny particles have their own "no-fly zones." It’s a concept that might seem a little abstract, but it’s the foundation of so much of our modern world. It’s a peek into the elegant, rule-bound, and surprisingly entertaining universe of the very small. It’s a reminder that even at the most fundamental levels, there’s a fascinating order to things, and that order can be downright delightful to explore. It makes you want to learn more, doesn't it? To dig a little deeper and uncover more of these incredible secrets that the universe is quietly keeping.