Lewis Dot Structure For Hydrogen Carbonate
Ever wonder what makes the fizz in your soda? Or how your body keeps everything balanced? It all comes down to tiny, invisible structures. And one of the coolest, most essential players is the hydrogen carbonate ion.
Think of it like a microscopic construction crew. These little guys are busy building and holding things together. They’re found everywhere, from the oceans to your own bloodstream. It's a real team effort down at the molecular level.
So, what exactly is this hydrogen carbonate? It’s also known by its fancy name, the bicarbonate ion. You might have heard that one before! It's a super important part of what makes life possible.
Must Read
The way these atoms connect is pretty neat. They arrange themselves in a special pattern. This pattern is what we call a Lewis Dot Structure. It's like a blueprint for how they behave.
Imagine the atoms are people holding hands. The dots in a Lewis Dot Structure represent the tiny bits that allow them to hold hands. These are called valence electrons. They are the outermost party-goers of the atom.
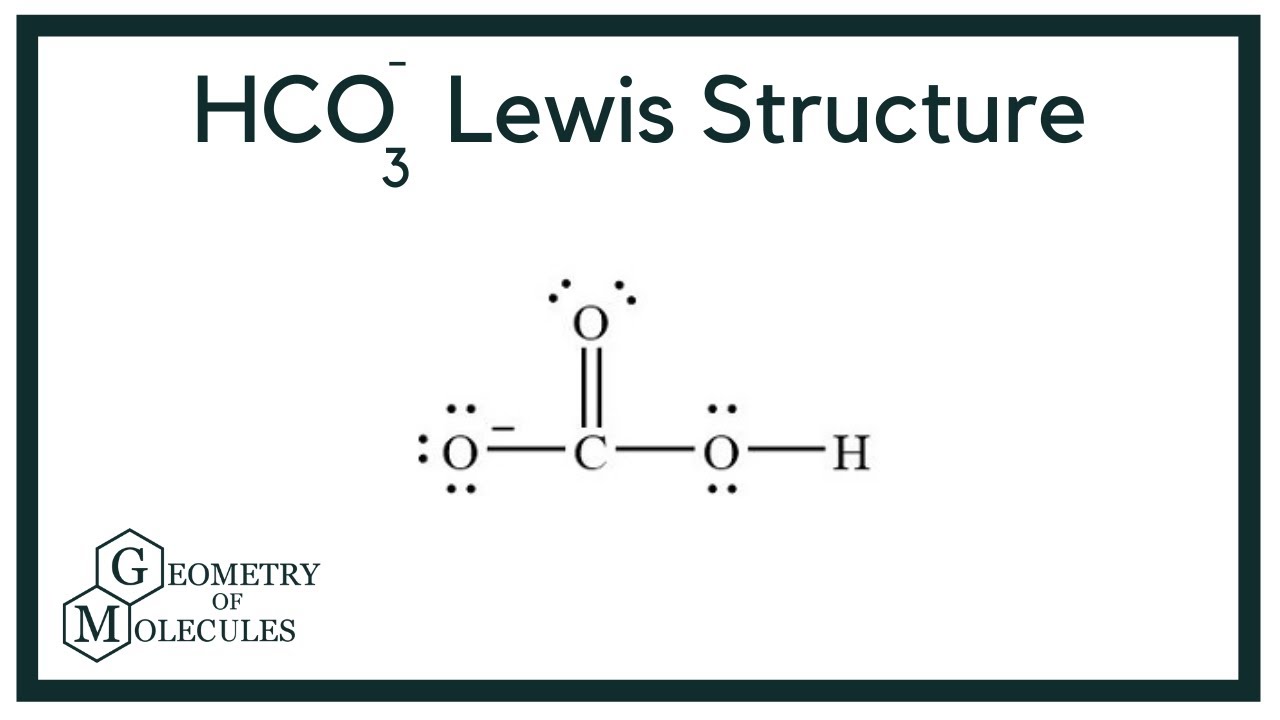
For hydrogen carbonate, we’re looking at a few key atoms. We have one carbon atom, three oxygen atoms, and one hydrogen atom. That’s our little molecular squad!
Now, let’s talk about the star of the show: carbon. Carbon is like the master organizer. It loves to make friends and connect with lots of other atoms. It’s the backbone of so many important molecules.
Oxygen atoms are also very social. They have a strong desire to complete their outer shell. They’re always looking for a few more electrons to feel perfectly balanced.
And then there’s hydrogen. It’s the smallest atom, but it plays a vital role. It’s happy with just one electron, and it readily shares to make connections.

When you put them all together to form hydrogen carbonate, something magical happens. They arrange themselves in a specific, stable way. This arrangement is what we visualize with the Lewis Dot Structure.
The carbon atom usually sits in the center. It's like the hub of a tiny wheel. It then reaches out to connect with the other atoms.
Two of the oxygen atoms form a pretty standard bond with carbon. They share electrons, creating a strong connection. This is like a firm handshake.
The third oxygen atom is where things get a bit more interesting. This one often has a double bond with the carbon. It's sharing two pairs of electrons, making a super strong grip.
And then there's the hydrogen atom. It latches onto one of the oxygen atoms. This is typically the one that's not involved in the double bond. It forms a single, loving bond.
But wait, there’s a twist! To make the whole structure stable and happy, there's a bit of juggling of electrons. This results in the entire hydrogen carbonate ion carrying a negative charge. It’s like the group has an extra electron they're all sharing.
This negative charge is key to its function. It means the hydrogen carbonate ion is attracted to positive things. It's a bit like a tiny magnet!

When we draw the Lewis Dot Structure, we show these shared electrons as dots or lines between the atoms. Lines usually represent a pair of shared electrons, forming a bond. Single lines are single bonds, and double lines are double bonds.
We also show the non-bonding electrons. These are the dots that belong to an atom but aren't being shared. They float around the atom like little companions.
And don't forget the brackets! Because it's an ion, the whole structure is enclosed in brackets. This signifies that it has a charge. The negative sign is proudly displayed outside.
So, the Lewis Dot Structure for hydrogen carbonate is a visual masterpiece. It shows how carbon, oxygen, and hydrogen cooperate. It reveals their electron dance to achieve stability.
It’s fascinating to see how these simple atoms create such a complex and vital molecule. The way they share and arrange their electrons dictates their behavior. It's like a secret handshake that makes them work.
This structure explains why hydrogen carbonate is such a good buffer. It can accept or donate a hydrogen ion. This helps maintain a stable pH. Think of it as a chemical referee, keeping things fair.
This buffering ability is crucial for life. In our blood, it prevents our pH from swinging wildly. This keeps our cells happy and functioning. It’s a silent guardian protecting us.
And in the oceans, hydrogen carbonate plays a huge role. It helps regulate the pH of the water. This is essential for marine life, from tiny plankton to giant whales. It’s the ocean’s balance keeper.
The Lewis Dot Structure is our window into this amazing world. It allows us to visualize these interactions. It’s like looking at a tiny, intricate puzzle.
Seeing the dots and lines makes abstract chemistry tangible. You can almost feel the electron sharing. It’s a simple way to understand complex processes.
What makes it so entertaining is the elegance of it all. Just a few atoms, a few electrons, and you have something so powerful and necessary. It’s nature’s ingenuity on full display.
It’s like discovering a secret code. Once you understand the language of dots and lines, you unlock a new level of understanding. You can see the hidden connections everywhere.
Consider the resonance structures of hydrogen carbonate. This is where it gets even more intriguing! The electrons in the double bond can actually “move.” They can shift around the oxygen atoms.

This means the actual structure is a blend of these possibilities. It's not just one fixed drawing. It's like a dynamic, flexible arrangement. This explains its remarkable stability.
The Lewis Dot Structure helps us appreciate this dynamism. We see how the electrons can be delocalized. They aren’t strictly tied to just two atoms.
This electron mobility is what gives hydrogen carbonate its robust nature. It can withstand various chemical environments. It’s a survivor in the molecular world.
It's a testament to how even the simplest drawings can reveal profound truths. The Lewis Dot Structure is not just a diagram; it’s a story. It’s a story of atoms working together.
It’s a story of balance, stability, and the very essence of life. From the fizz in your drink to the chemistry of your body, hydrogen carbonate is there. And its Lewis Dot Structure is its captivating portrait.
So next time you take a sip of something bubbly, or think about how your body works, remember the humble hydrogen carbonate ion. And perhaps, take a peek at its Lewis Dot Structure. You might find yourself surprisingly entertained.
It's a fun and easy way to peek behind the curtain. You'll see the fundamental building blocks of our world. And it all starts with understanding how atoms draw their little dot families.