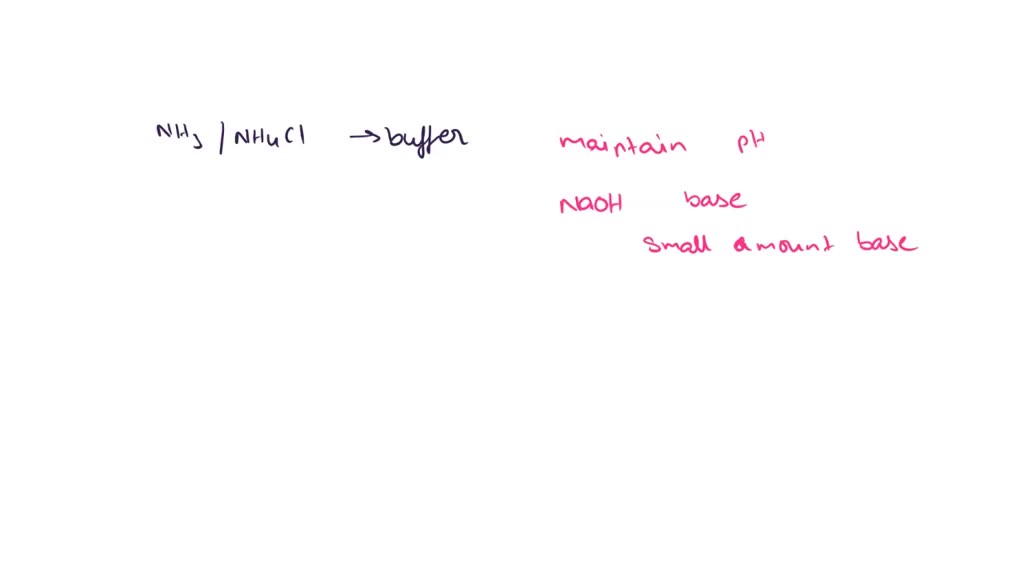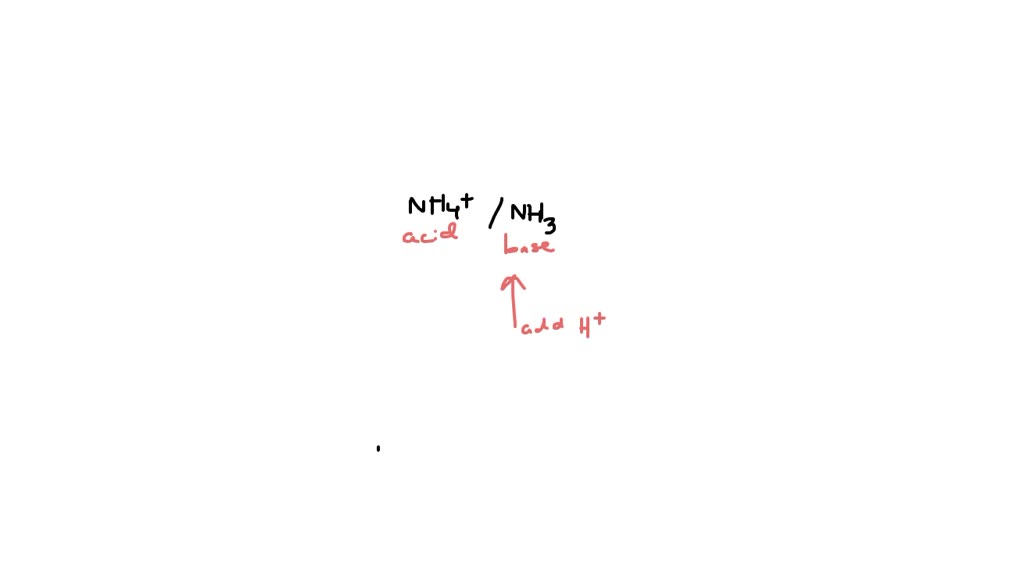You Have A Buffer Composed Of Nh3 And Nh4cl.

Hey there, science buddy! Ever heard of a buffer? Nope, not the kind you spill on your keyboard. We're talking about a chemical kind. And guess what? You've got one sitting pretty in your life, made of ammonia (NH3) and ammonium chloride (NH4Cl). Sounds fancy, right? But it's actually super cool.
Think of a buffer like a tiny, chemical bodyguard. It’s there to keep things stable. Imagine you're trying to balance a wobbly table. You add a little something here, adjust a bit there. That’s what a buffer does for a solution. It stops it from going totally bonkers when you add acids or bases.
So, why ammonia and ammonium chloride specifically? Because they're a dynamic duo. NH3 is a base. It's a bit of a lone ranger, happy to pick up extra protons. NH4Cl, on the other hand, is kind of like the responsible friend. It's the salt of a strong acid (HCl) and a weak base (NH3), and it readily gives up a proton (H+) to become NH3. See the connection? They're basically two sides of the same coin, ready to jump in and save the day.
Must Read
This NH3/NH4Cl system is a classic. It’s like the peanut butter and jelly of buffer solutions. Easy to find. Easy to understand. And surprisingly important.
Why Buffers Matter (Even When You Don't Know It)
You know how your body has to keep its pH just right? Too acidic, and you're in trouble. Too alkaline, and, well, also trouble. Your blood is a super-sophisticated buffer system. And wouldn't you know it, ammonia and ammonium ions play a huge role in your kidneys' ability to manage that pH balance. How wild is that? Your insides are like a tiny chemical lab, and these guys are part of the crew.
It’s not just you. Lots of biological processes rely on stable pH. Enzymes, those tiny workhorses in your cells, are super picky. They only function optimally within a narrow pH range. If the pH swings too far, they just… stop working. Imagine your favorite video game character suddenly losing all their powers because the game's console had a "pH glitch." Not fun.

And it’s not just living things! Think about chemical manufacturing. Many reactions need a very specific pH to happen correctly. If you're making pharmaceuticals, or dyes, or even just cleaning products, you need to control that acidity. Using a buffer like NH3/NH4Cl is a common way to do it. It's the unsung hero of many a bubbly concoction.
The Quirky Chemistry of NH3 and NH4Cl
Let's dive a little deeper into our favorite dynamic duo. Ammonia (NH3) is a gas. It's got that distinctive smell. You know the one. Sometimes it’s the smell of a clean barn, sometimes it’s… well, let's just say it’s memorable. It's a weak base because it doesn't hog protons like a strong base would. It's more of a gentle persuader.
Ammonium chloride (NH4Cl) is a salt. When you dissolve it in water, it acts like it’s got a secret agenda. It dissociates into NH4+ ions and Cl- ions. The Cl- ions are pretty chill, they just hang out. But the NH4+ ion? That's our star player. It’s the proton donor. It's like, "Here, take this H+ if you need it!"
So, here’s the magic. If you add acid (which is full of H+ ions) to your buffer solution, the NH3 is like, "Ooh, extra H+? I'll take that!" It snatches up the extra protons, forming NH4+. The solution’s pH barely budges. It’s like adding a few extra people to a party that’s already got plenty of room. No biggie.

Now, what if you add a base (which wants to take H+ ions)? The NH4+ ion is like, "Hold up! You're trying to steal my friend's protons? Not on my watch!" It readily gives up its proton to the base, turning back into NH3. Again, the pH stays remarkably steady. It’s like the bouncer at the party, preventing any unwanted chaos.
It's this delicate dance between NH3 accepting protons and NH4+ donating them that makes this buffer system so effective. They’re constantly looking out for each other, keeping the solution in a happy equilibrium.
When Things Get a Little… Ambitious
What happens if you really push it? If you add a TON of acid or base, even a buffer can be overwhelmed. It’s like trying to bail out a sinking ship with a teacup. Eventually, you're going to get swamped. This is called exceeding the buffer capacity.

The range of a buffer is also important. The NH3/NH4Cl buffer works best when the pH is close to the pKa of the ammonium ion. The pKa is a fancy way of saying the pH at which the acid and its conjugate base are present in equal amounts. For NH4+, this is around 9.25. So, this buffer is your go-to for keeping things in the slightly alkaline range. Not for super acidic stuff, though. You wouldn't use a teacup to put out a bonfire, right?
Think of it this way: a buffer is like a shock absorber. It absorbs the bumps. But if you hit a massive pothole, even the best shock absorber will struggle.
Fun Facts You Never Knew You Needed
Did you know that ammonia is used in cleaning products for a reason? Its basic nature helps break down grease and grime. And when it reacts with acids, it neutralizes them. So, in a way, even your Windex is playing a bit of a buffering game!
Ammonia is also a key component in making fertilizers. Happy soil needs the right pH for plants to thrive. So, our little NH3/NH4Cl pals are helping grow your dinner. You're welcome, food.

The ammonium ion (NH4+) can sometimes be mistaken for a proton (H+). They look similar in chemical formulas, but they're different players in the buffering game. It's like mistaking a look-alike actor for the real star. Close, but not quite the same impact.
And here's a quirky one: In ancient Rome, ammonia was collected from urine. Yes, urine. It was used in tanning leather and even as a cleaning agent. So, the next time you smell ammonia, maybe just think about ancient Rome. It makes it… interesting?
The ability of buffers to resist pH change is a cornerstone of chemistry and biology. It’s a principle that underpins so much of what we understand about how the world works, from the tiniest cell to industrial processes. And it all comes down to a simple, elegant interplay between a base and its conjugate acid.
So, the next time you encounter ammonia or ammonium chloride, give them a nod. They’re the quiet heroes, the chemical lifeguards, keeping everything in check. They’re part of a fascinating chemical world that’s always buzzing with activity, and you're now a little more in on the secret. Pretty neat, huh?