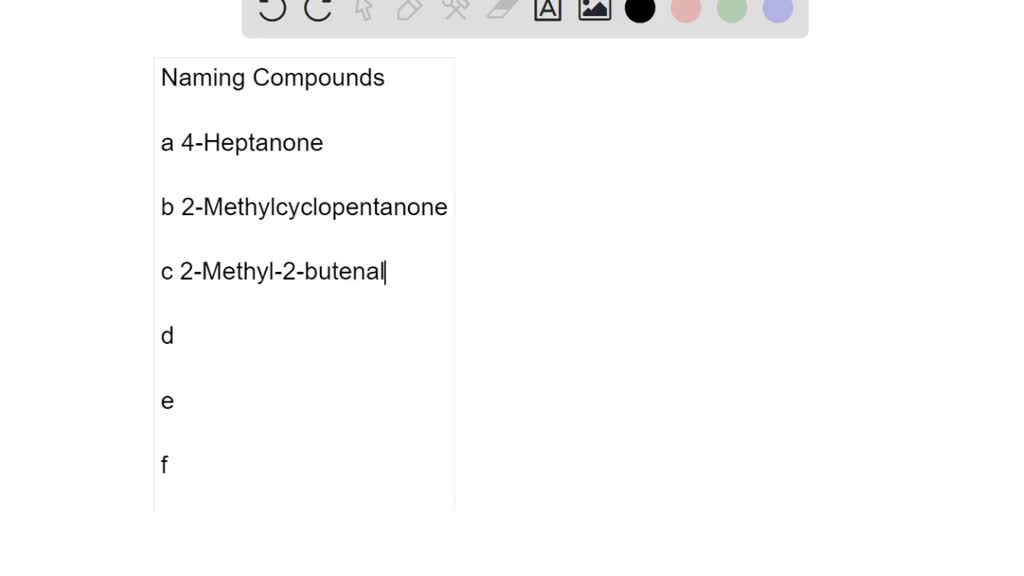
Write The Names For The Following Compounds
Hey there, fellow explorers of the weird and wonderful world of chemistry! Ever look at those long, complicated names for chemicals and think, "Who on earth comes up with these?" Well, today, we're going to peek behind the curtain and discover how chemists name stuff. It's not as scary as it looks, I promise. Think of it like cracking a secret code, where each part of the name tells you something specific about the molecule. Pretty neat, right?
We're going to dive into naming some common types of compounds. No need to grab your lab coat; we're keeping it super chill, like a coffee shop chat about the universe. We'll even throw in some fun analogies to make it stick. Ready to become a naming ninja? Let's go!
Ionic Compounds: The Perfect Pairing
First up, let's talk about ionic compounds. These are formed when one atom gives an electron to another. It's like a really generous friend handing over a valuable item. This creates two charged particles: a positively charged one (a cation) and a negatively charged one (an anion). And guess what? Opposites attract! They stick together like magnets, forming a stable compound.
Must Read
Naming these is actually quite straightforward. You take the name of the metal (the positive one, usually from the left side of the periodic table) and add the name of the non-metal (the negative one, usually from the right side). But here's the twist: the non-metal's name gets an "-ide" ending. So, if you have sodium (Na) giving an electron to chlorine (Cl), you don't call it "sodium chlorine." Nah, it becomes sodium chloride. Think of it like adding a suffix to make it sound official.
Why the "-ide" ending? It's basically a flag that says, "Hey, this element has gained electrons and is now part of a compound." It’s like giving a specific title to someone who's taken on a new role. So, if you see an "-ide" at the end of a name and the first part is a metal, you can bet it's an ionic compound.
Let's try another one. Magnesium (Mg) is a metal, and bromine (Br) is a non-metal. Magnesium gives electrons to bromine, and they form a compound. What do you think it's called? Yep, you guessed it: magnesium bromide. See? You're already getting the hang of it. It’s like learning the names of musical notes; once you know C, D, and E, you can start making melodies.
What about something like aluminum (Al) and oxygen (O)? Aluminum is a metal, and oxygen is a non-metal. So, the name would be aluminum oxide. Simple, right? The trick with ionic compounds is recognizing the metal and the non-metal, and then remembering that the non-metal gets that "-ide" treatment. It’s like a little linguistic handshake between the elements.

Covalent Compounds: The Sharing is Caring Crew
Now, let's move on to covalent compounds. These are a bit different. Instead of one atom completely giving up an electron, atoms in covalent compounds share electrons. It’s like a group of friends deciding to share their snacks instead of one person hogging them all. This sharing creates a strong bond, and these compounds are often found in everyday things like water and sugar.
Naming covalent compounds uses a system of prefixes to tell you exactly how many of each atom are present. This is where it gets really fun, because you’re being super specific. Think of these prefixes like numbers in code.
Here are some of the common prefixes:
- Mono-: One
- Di-: Two
- Tri-: Three
- Tetra-: Four
- Penta-: Five
- Hexa-: Six
- Hepta-: Seven
- Octa-: Eight
- Nona-: Nine
- Deca-: Ten
When naming a covalent compound, you name the first element, and then the second element gets an "-ide" ending, just like with ionic compounds. But here’s the key difference: the prefixes tell you how many of each element there are. You usually only use prefixes for the second element, but if there's more than one atom of the first element, you use a prefix for that too.
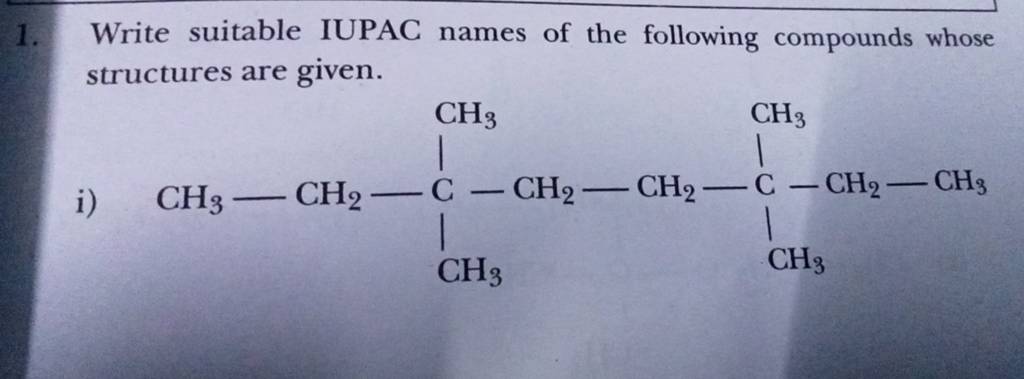
Let's take a classic example: water. Its chemical formula is H₂O. That means two hydrogen atoms and one oxygen atom. The first element is hydrogen, and the second is oxygen. Since there are two hydrogen atoms, we use the prefix "di-". And since there's only one oxygen atom, we don't need a prefix for it (unless it's the only atom of that element, then we use "mono-", but we usually drop the "o" in "mono-" before a vowel. So, for oxygen, it becomes "monoxide" but often just "oxide" if it's the second element and there's only one). So, H₂O is dihydrogen monoxide. Sounds fancy, right? But it's just water!
Another common one is carbon dioxide, CO₂. This means one carbon atom and two oxygen atoms. So, we have "carbon" (no prefix needed for the first element if there's only one) and then "di-" for the two oxygens, and the oxygen gets the "-ide" ending. Voilà: carbon dioxide. You breathe this out, and plants love to use it! It’s like saying "one carbon, two oxygens" in chemist-speak.
What about something like N₂O? That's two nitrogen atoms and one oxygen atom. So, we’d say dinitrogen monoxide. This one’s commonly known as nitrous oxide, or laughing gas! See how the prefixes are crucial for clarity? If we just said "nitrogen oxide," how would we know if it's N₂O or NO or N₂O₄? The prefixes are the disambiguators.
Let's try one more: SO₃. That's sulfur and three oxygen atoms. So, it's sulfur trioxide. This is a component of acid rain, which is a bit sad, but the naming is still pretty straightforward once you know the rules.
Acids: The Sour Bunch
Acids are a special category of compounds. They often have a distinct sour taste (think lemon juice, which contains citric acid) and can be quite reactive. The naming of acids depends on whether they contain oxygen or not.
If an acid doesn't contain oxygen, it's called a hydro-something-ic acid. The "hydro-" prefix tells you there's hydrogen involved, and it's usually bonded to a single non-metal element. For example, HCl in water is hydrochloric acid. The "chlor" part comes from chlorine, and the "-ic acid" ending tells you it's an acid without oxygen.
Now, if the acid does contain oxygen, the naming is based on the polyatomic ion it's derived from. This is where things get a little more involved, but the pattern is still there! If the polyatomic ion ends in "-ate," the acid name ends in "-ic acid." If the polyatomic ion ends in "-ite," the acid name ends in "-ous acid."
Let's take sulfuric acid. It comes from the sulfate ion (SO₄²⁻). Since sulfate ends in "-ate," the acid is sulfuric acid. Pretty neat, right? It’s like a family name: if the ion is a "Sulfate," the acid is a "Sulfuric."
And what about sulfurous acid? This comes from the sulfite ion (SO₃²⁻). Since sulfite ends in "-ite," the acid becomes sulfurous acid. So, the pattern is "-ate" becomes "-ic," and "-ite" becomes "-ous." Think of it like a slight variation in a tune.
What about nitric acid? It comes from the nitrate ion (NO₃⁻), which ends in "-ate." So, it's nitric acid. And nitrous acid? That comes from the nitrite ion (NO₂⁻), which ends in "-ite." So, it's nitrous acid.
These are just a few examples, but you can see the logic behind it. It’s all about recognizing the building blocks and following the naming conventions. It's like learning the grammar of chemistry; once you get the rules, you can construct almost any sentence – or name!
Why Does This Matter?
You might be thinking, "Okay, that's interesting, but why should I care about naming compounds?" Well, it's more than just memorizing. Understanding these names gives you a deeper appreciation for the world around you. Every medicine, every plastic, every food additive, and even the air you breathe is made of chemical compounds. Being able to decipher their names is like having a key to unlock their secrets.
It helps us communicate clearly in science, ensuring everyone is talking about the same substance. Imagine trying to order a specific chemical without a standardized name – chaos! These naming systems are the backbone of chemical communication. So, the next time you see a long chemical name, don't be intimidated. Take a breath, look for those prefixes, suffixes, and element combinations. You might just find yourself decoding the language of the universe, one compound at a time. And that, my friends, is pretty cool.