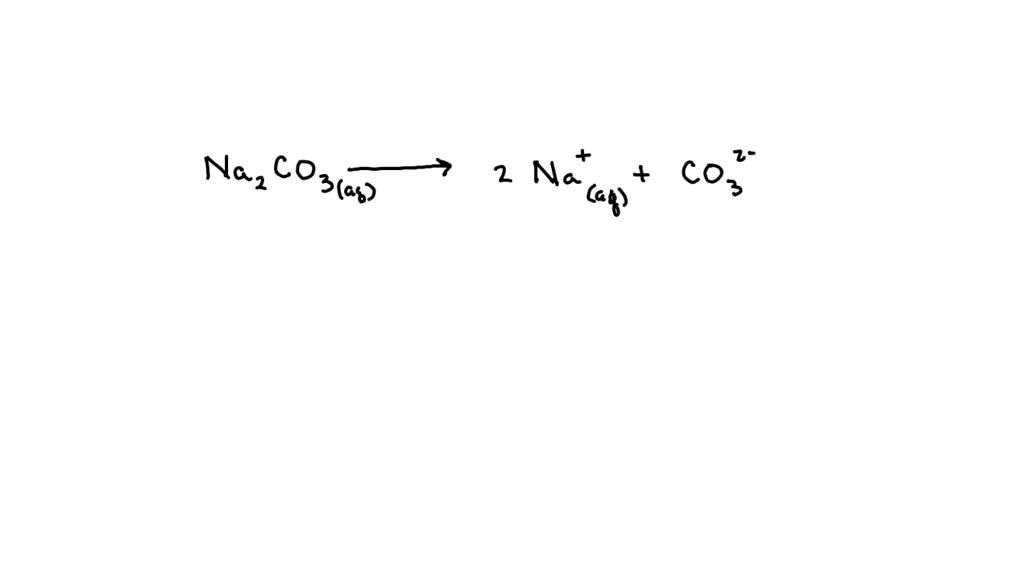
Write The Ions Present In Solution Of Na2co3
Ever find yourself staring at a box of baking soda, or maybe you're a closet laundry-booster enthusiast? You know, the stuff that makes your cookies fluffier and your whites brighter? Well, guess what? You're already playing chemist in your own kitchen, and today we're going to peek behind the curtain of one of those common helpers: sodium carbonate, or as some of us affectionately call it, washing soda. It's the unsung hero of a clean home and a happy bake sale.
Now, imagine you’ve got a big ol' glass of water. It’s just sitting there, minding its own business. Then, you decide to sprinkle in some of that magical sodium carbonate powder. What happens next? It doesn't just sit there like a shy guest at a party. Nope! It starts to get a little… excited.
See, when sodium carbonate (which we can write down as Na₂CO₃ if we’re feeling fancy) hits water, it’s like dropping a handful of bouncy balls into a ball pit. Everything starts to break apart and mingle. It’s not a violent explosion or anything, more like a friendly mingling session where everyone finds their own little corner to hang out in. And these little guys that break apart? They’re called ions. Think of them as the individual personalities or the little LEGO bricks that make up the original sodium carbonate compound.
Must Read
So, what are these party guests, these ions, that show up when sodium carbonate dives into the water? We’ve got a couple of main characters, and a couple of supporting cast members who are always around for the ride.
The Star of the Show: Sodium Ions
First up, we have the sodium ions. These are the happy, positively charged chaps. We write them down as Na⁺. Now, why are they positive? It's a bit like they've got a little extra pep in their step, a tiny spark of energy that makes them want to be a bit outgoing. They've shed an electron, and that's made them a little bit… well, positive!
Think of them as the really popular kids at school. They’re everywhere, they’re friendly, and they tend to stick around. When you put Na₂CO₃ in water, for every one molecule of sodium carbonate, you get two of these Na⁺ ions. It’s like opening a pack of gum and finding two pieces instead of one. Double the fun, right?
These little sodium guys are like the cheerleaders of the solution. They’re bouncing around, spreading good vibes, and generally keeping things lively. They don’t do a whole lot of the heavy lifting when it comes to, say, cleaning your shirt or making your cake rise, but they are absolutely essential. They’re the foundation, the ones who make sure the whole operation can even happen.
Imagine you’re trying to organize a surprise party. You need the decorations, the cake, the music, but you also need people to be there. The sodium ions are those enthusiastic guests who show up early, ready to help set things up. They might not be the ones mixing the frosting, but their presence makes everything possible.
And here’s a fun fact: these sodium ions are pretty much everywhere. They’re in our tears, our sweat, and even that salty snack you shouldn't have had but totally did. They’re incredibly common, which is why they’re such reliable companions in our chemical concoctions.
The Real MVP: Carbonate Ions
Now, let’s talk about the other main player, the one who really gets down to business. These are the carbonate ions. They have a bit of a different vibe. They’re carrying a negative charge, which we show as CO₃²⁻. That little "2-" is important, because it means they're not just a little bit negative, they're twice as negative. They've got a bit of a "take charge" attitude, and that's what makes them so useful.
Think of these carbonate ions as the master organizers or the skilled artisans of the group. They're the ones who actually do the work. When you use washing soda to tackle a greasy pan, it's these CO₃²⁻ ions that are the real MVPs. They're like tiny cleaning superheroes, swooping in to neutralize acids and break down stubborn grime. They’re the reason your laundry smells fresh and your sink sparkles.
Why are they so good at their job? Well, they're a bit like a really good hug. They can grab onto things that are a bit acidic (which are often the "bad guys" in cleaning situations) and neutralize them. It’s a chemical reaction, but think of it as a friendly handshake that diffuses a tense situation. Acids have a positive-leaning personality, and the negatively charged carbonate ions are just perfect for balancing them out.

When you're baking with baking soda (which is a different form of sodium carbonate, but the principle is similar!), the carbonate part is what reacts with acidic ingredients to create carbon dioxide gas. This gas is what makes your cookies puff up and your cakes rise like a proud parent watching their kid graduate. It's the CO₃²⁻ ions doing their magic!
Imagine you’re at a potluck, and someone brings a dish that’s a little too sour. The carbonate ion is like the person who brings a little extra sugar or baking soda to balance it out. They don't just stand there; they actively make things better. They’re the problem-solvers.
The Supporting Cast: Water Molecules
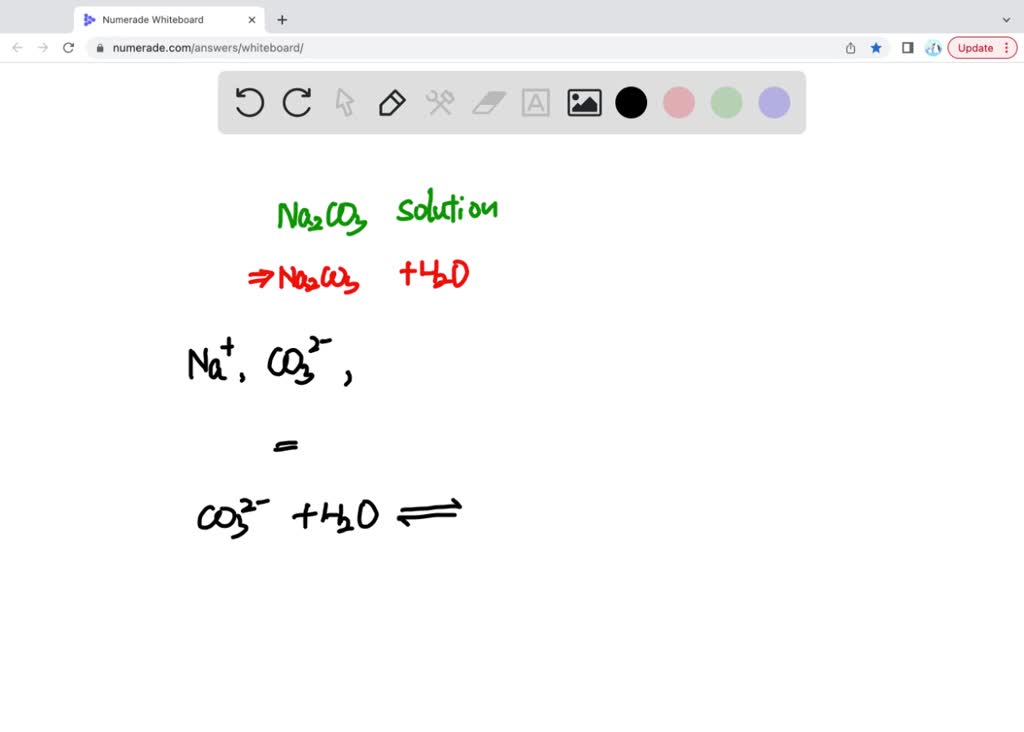
Now, while we're talking about the stars of the show, we can't forget the stagehands, the ones who make the whole performance possible. These are the water molecules, represented by H₂O. They’re the ones who create the environment for the sodium and carbonate ions to do their thing.
Water is a pretty amazing solvent. It’s like the ultimate social lubricant for chemicals. When sodium carbonate dissolves in water, it's the water molecules that surround and separate the Na⁺ and CO₃²⁻ ions. They're the friendly crowd that helps everyone feel comfortable and spread out.
Think of water as the dance floor. The sodium ions and carbonate ions are the dancers. Without the dance floor (water), they’d just be standing around awkwardly. Water molecules are polar, meaning they have a slightly positive end and a slightly negative end. This makes them really good at attracting and surrounding charged ions, pulling them apart from each other and keeping them dispersed.
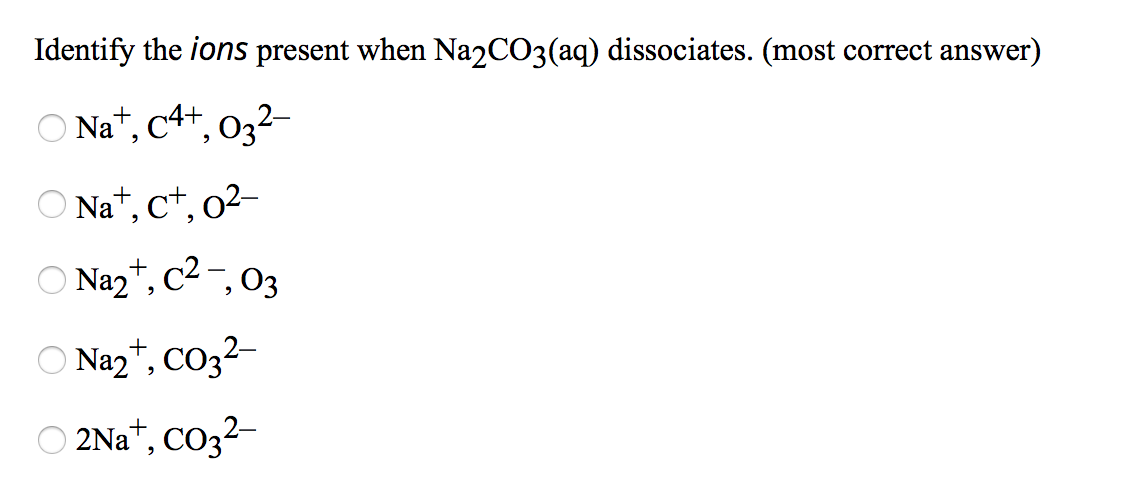
This is why not everything dissolves in water. Some things are just loners, or they have a personality that doesn't quite gel with water's. But sodium carbonate? Oh, it loves water! It’s like a popular kid finding their best friends at a party. The water molecules are the ones who make sure everyone has enough space to move and groove.
A Bit of Nuance: Hydroxide Ions
Here's where things get a tiny bit more interesting, and it's where the "easy-going" part might need a little stretch, but bear with me! When those carbonate ions are hanging out in the water, they can actually do something rather cheeky. They can steal a little bit of hydrogen from a water molecule.
So, a CO₃²⁻ ion can snatch a hydrogen atom (which has a positive charge, remember?) from an H₂O molecule. When this happens, the water molecule is left behind with one less hydrogen, turning it into a hydroxide ion, which has a negative charge and is written as OH⁻. And the carbonate ion, now with an extra hydrogen, becomes a bicarbonate ion (HCO₃⁻).
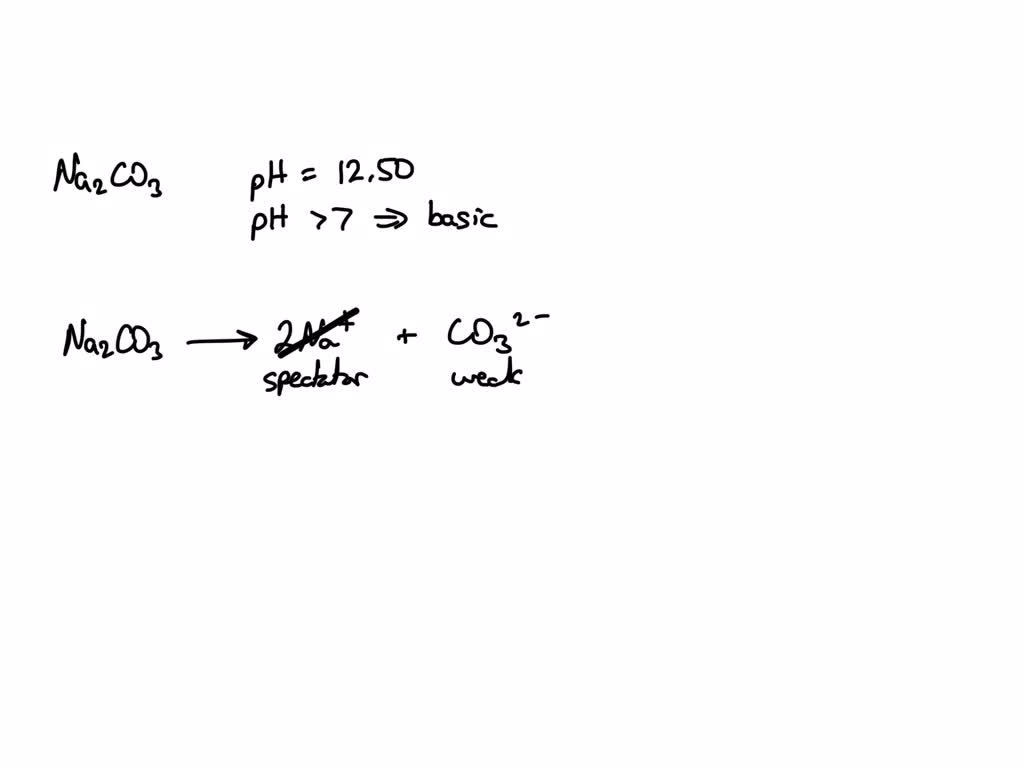
This is why solutions of sodium carbonate are a bit alkaline or basic. The OH⁻ ions are the reason for that. They’re the ones that make your skin feel a little bit slippery if you get concentrated washing soda on it (don't do that, by the way!). They're the opposite of acids; they're the ones that neutralize acids.
Think of it like this: the carbonate ions are super friendly, and they're so eager to interact that they sometimes break up a perfectly good water molecule. When they do, they leave behind these hydroxide ions, who are also a bit negatively charged and contribute to the overall "basic" nature of the solution. It’s like a chain reaction of social interaction!
This is a crucial point for cleaning. Many stains are acidic. So, the presence of these OH⁻ ions (along with the CO₃²⁻ ions doing their direct attack) helps to combat those acidic grime monsters. It’s a team effort, with the CO₃²⁻ and OH⁻ ions working together to create a clean environment.
Putting It All Together: The Ion Party!
So, when you’ve got your sodium carbonate (Na₂CO₃) dissolved in water, you're not just looking at a clear liquid. You've got a bustling microscopic party going on! You have:
- Lots and lots of happy, positive Na⁺ (sodium) ions, like the energetic guests.
- The hardworking, negatively charged CO₃²⁻ (carbonate) ions, the real cleaners and bakers.
- Some OH⁻ (hydroxide) ions, created by the carbonate ions being a little too friendly with water, which make the solution basic.
- And of course, plenty of H₂O (water) molecules, acting as the dance floor and the facilitators of all this interaction.
The bicarbonate ions (HCO₃⁻) are also around, as a result of the carbonate ions grabbing a hydrogen. They're like the slightly modified versions of the original guests, still mingling and contributing.
It’s a whole ecosystem of charged particles, all zipping around, bumping into each other, and doing their specific jobs. It’s not about the solid Na₂CO₃ anymore; it’s about the individual personalities of these ions that make it so effective.
Next time you reach for that box of washing soda or marvel at your perfectly risen cake, take a moment to appreciate the incredible world of ions that's at play. It’s a tiny, invisible party happening in every drop of that solution, and it’s responsible for so much of the clean and tasty magic in our lives!