Why Sodium Is More Reactive Than Lithium

Hey there, science curious friend! Ever wondered why some elements are, well, a bit more enthusiastic about reacting than others? Like, why does sodium go nuts when it hits water, but lithium is just… chill? Today, we're diving into the exciting world of alkali metals and unraveling the mystery of why sodium is the drama queen of the pair, while lithium’s more of a cool cat. No intimidating jargon, just a friendly chat about atoms and their atomic personalities!
So, we’re talking about two really cool, really reactive elements: sodium (Na) and lithium (Li). They’re both in the same family on the periodic table – the alkali metals. Think of them as siblings. They share some family traits, like being super eager to give away an electron (we'll get to that!), but they definitely have their own quirks. And today, we're focusing on sodium's extra-spicy reactivity compared to its slightly more laid-back brother, lithium.
The Atom's Inner Circle: Electron Shells
To understand this, we gotta peek inside an atom. Don’t worry, we’re not bringing microscopes or anything! Atoms have a nucleus in the center, kinda like the heart of the operation. And then, buzzing around this nucleus, are electrons. These electrons aren’t just randomly floating; they hang out in specific "shells" or "energy levels." Think of them like different floors in a building. The closer you are to the nucleus, the more energy you need to escape, and the more tightly you're held.
Must Read
Now, here’s the crucial bit: atoms love to have a full outer shell of electrons. It’s like a state of pure bliss for them, a cosmic equilibrium. When an atom has an incomplete outer shell, it’s got this constant itch to either grab some electrons or, more commonly for our alkali metal pals, to get rid of the single, lonely electron chilling in its outermost shell. This eagerness to achieve that perfect, full outer shell is what makes them so reactive!
Lithium's Humble Abode
Let’s start with lithium (Li). It's the smallest of the alkali metals. It has three protons in its nucleus and three electrons zipping around. Now, its electron arrangement is pretty neat: it has two electrons in its inner shell (which is full, like a cozy, sealed room) and just one electron in its outermost shell. This single electron is its ticket to becoming stable. All it needs to do is ditch that one extra electron, and poof! it has a full, stable inner shell. Easy peasy, right?
Imagine lithium as a tiny apartment building with two floors. The first floor is totally packed with tenants (electrons). The second floor, however, has only one tenant. This tenant is a bit of a nomad, always looking for a way out to join a place where all the apartments are full. It doesn't take much convincing for this one tenant to leave!

Sodium: The Slightly More Spacious Pad
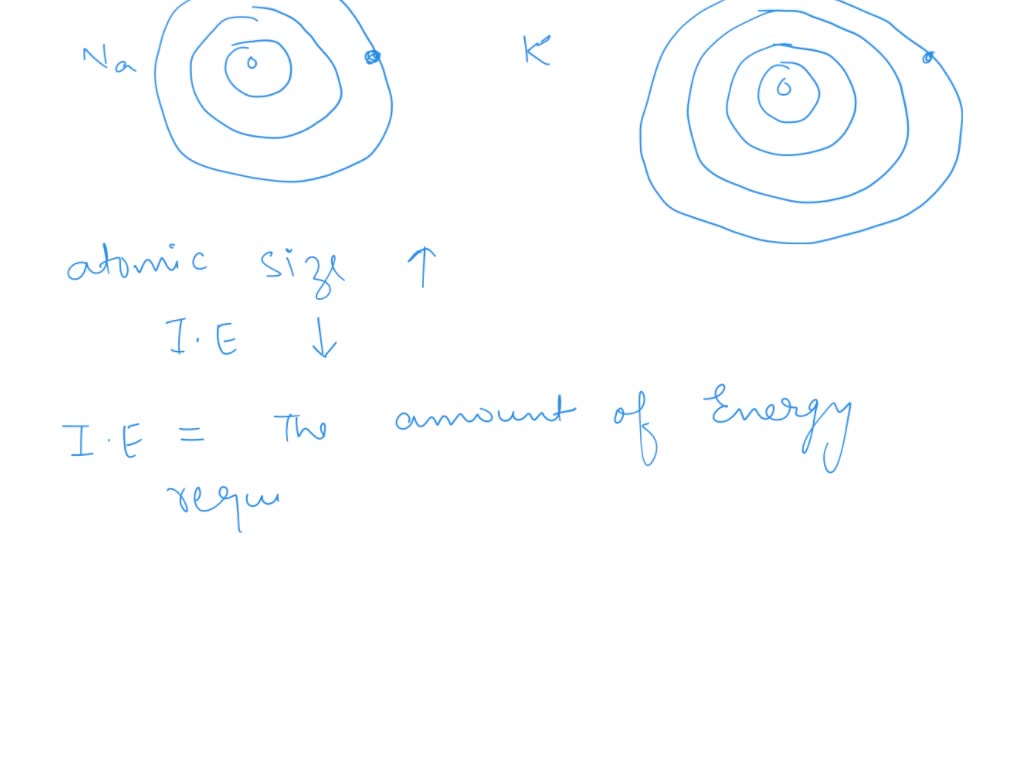
Now, let’s talk about sodium (Na). Sodium has 11 protons and 11 electrons. Its electron setup is a bit more spread out: two electrons in the first shell, eight in the second (both full, like perfectly occupied apartments), and then… one lonely electron in its outermost third shell. See the pattern? Just like lithium, it has that one single electron in its outer shell, just begging to be released to achieve stability.
So, why the difference in reactivity? It all comes down to distance. That single electron in sodium's outermost shell is further away from the positive pull of the nucleus than lithium’s single outer electron. Think of it like this: if you have a magnet (the nucleus) and a tiny metal ball (the electron), the closer the ball is to the magnet, the stronger the pull. The further away it is, the weaker the pull.
The Nucleus's Grasp
The nucleus of an atom is positively charged because of its protons. Electrons are negatively charged. Opposite charges attract, so the nucleus is constantly pulling on its electrons. In lithium, that outer electron is pretty close to the nucleus, so it’s held on with a decent amount of grip. In sodium, however, that outer electron is in a shell that’s further away. The nucleus's pull is significantly weaker on that electron.

This weaker pull means that it takes less energy to yank that outer electron away from a sodium atom than from a lithium atom. And in the world of chemistry, less energy needed for a reaction means more reactivity. Sodium is just a bit more willing to let go of its electron because it’s not as strongly attached.
The 'Oh Crap!' Moment: Reacting with Water
This is where we see the difference in action! Both sodium and lithium react with water, but sodium’s reaction is, shall we say, more dramatic. When you drop a tiny piece of sodium into water, it immediately starts to fizz, sizzle, and can even catch fire! It’s a whole show.
The sodium atom readily gives up its outer electron to a water molecule. This process releases a lot of energy, and the heat generated is often enough to ignite the hydrogen gas that's also produced in the reaction. It's like sodium is saying, "Here, take it! I'm done with this electron! Let's make some noise!" The reaction is so vigorous that it can be quite dangerous if not handled carefully.
Lithium's Polite Greeting
Now, lithium. When you put lithium in water, it’s a much gentler affair. It reacts, sure, and it produces hydrogen gas and lithium hydroxide, but it doesn't usually erupt in flames. It’s more of a polite nod and a handshake. The reaction still happens because lithium wants to lose that outer electron, but because that electron is held a tiny bit tighter by the nucleus, it takes a little more effort (energy) to get the job done.

Think of lithium as someone who’s willing to lend you a dollar, but they’ll probably keep it in their wallet for a bit. Sodium, on the other hand, is the friend who throws their wallet at you and says, "Just take it and buy us all ice cream!" (Though maybe not the best analogy for a scientific reaction, you get the vibe!) The energy released from lithium’s reaction is less intense, so you don't get that spectacular fiery display.
It's All About The Distance and Shielding
So, let's recap the main points:
- Electron Shells: Atoms like to have full outer electron shells for stability.
- Alkali Metals' Quirk: Sodium and lithium each have just one electron in their outermost shell, making them eager to shed it.
- The Magic of Distance: Sodium's outer electron is further from the nucleus than lithium's.
- Weaker Nuclear Pull: Because it's further away, the nucleus's grip on sodium's outer electron is weaker.
- Less Energy = More Reactivity: It takes less energy to remove sodium's outer electron, making it more reactive.
There's also a subtle thing called "shielding." Those inner electrons in both lithium and sodium act like a bit of a shield, blocking some of the nucleus's positive charge from reaching the outer electrons. Since sodium has more inner electron shells than lithium, this shielding effect is a bit stronger for sodium's outer electron, further weakening the nucleus's pull. It’s like having more people in between you and the person you're trying to reach – their voice gets muffled!

A Bigger Atom, A Bigger Splash
Essentially, sodium is a bigger atom. It has more "real estate" for its electrons. This extra space, combined with the shielding effect, makes its outermost electron a bit of a free spirit. It's not as deeply entrenched in the atomic structure and is therefore more easily coaxed into a reaction. Lithium, being smaller, has its outer electron closer to the nucleus and under a stronger influence, making it a tad more reluctant to participate in any explosive chemical drama.
It’s not that lithium isn’t reactive; it absolutely is! It’s just that sodium has that extra oomph. That extra electron is just that little bit more accessible, that little bit more ready to jump ship. So, when sodium meets water, it’s like the ultimate chemical party starter, while lithium is more of the polite guest who joins the dance a little later.
The Beauty of Gradual Change
And you know what's really cool about this? It shows us that even within the same "family" of elements, there's a beautiful spectrum of behavior. The periodic table isn't just a chart; it's a roadmap of predictable, yet fascinating, trends. Understanding these trends helps us predict how different elements will interact, which is super important for everything from developing new medicines to creating advanced materials.
So, the next time you hear about sodium’s dramatic flair or lithium’s calmer demeanor, you’ll know why! It’s all about the atomic dance, the interplay of charged particles, and the simple, yet profound, concept of distance. It’s a little reminder that even in the tiny world of atoms, there’s a whole lot of personality and a reason for everything. And isn’t that just a wonderfully mind-boggling thing to think about? Keep exploring, keep wondering, and remember, science is everywhere, and it's pretty darn fun!