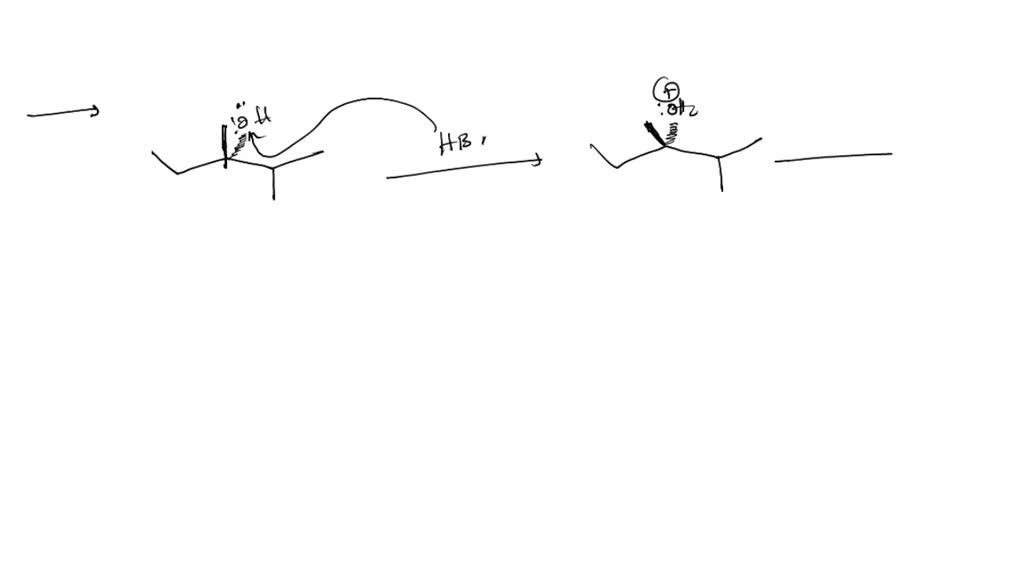
Why Is Racemic Mixture Not Optically Active Exam Question

Alright, let's talk about something that sounds a bit fancy but is actually super cool and surprisingly important in the world of chemistry. You know how sometimes things come in pairs, like your left and right socks? Well, in the tiny world of molecules, some things also come in these mirrored pairs. We call these enantiomers. Think of them like your hands – they look identical but are mirror images, and you can't superimpose them perfectly. Pretty neat, right?
Now, here's where the exam question fun begins. Imagine you've got a bunch of these mirrored molecule pairs. What happens if you mix them all up, exactly 50/50? This magical 50/50 blend is called a racemic mixture. It's like having an equal number of left and right shoes all jumbled together in a big box. Easy enough to picture!
So, the big question in chemistry exams, the one that gets students scratching their heads a bit, is: Why is a racemic mixture not optically active? It sounds like a riddle, doesn't it? And honestly, it's one of those "aha!" moments when you finally get it. It's not some complex, scary scientific concept. It's actually quite elegant and has a really simple explanation, once you let go of any intimidation.
Must Read
Let's dive into why this is so entertaining. The whole field of optical activity is about how certain molecules interact with light. Specifically, they can twist or rotate polarized light. Think of polarized light like a wave that's only moving up and down, not side to side. When this special light passes through a sample of an optically active substance, it's like the substance gives the light a gentle nudge, making it twist in a particular direction. Some molecules twist it to the right (we call this dextrorotatory, or d), and others twist it to the left (we call this levorotatory, or l).
This light-twisting ability is super important. It's how scientists can tell different enantiomers apart. Imagine you have two drugs that are enantiomers. One might be a fantastic medicine, while the other could be completely useless or even harmful! So, being able to identify and separate them is a big deal in pharmaceuticals. It’s like having a secret code that only one version of the molecule can read or write.

Now, back to our racemic mixture. We've got our 50/50 blend of the left-handed and right-handed molecules, or the d and l forms. Here's the delightful bit: they're like two dancers doing opposite moves on the dance floor. One is twirling clockwise, and the other is twirling counter-clockwise at the exact same speed and intensity. What happens to the overall light when it encounters both of them?
The d enantiomer, with its right-twisting power, rotates the polarized light. Let's say it rotates it 10 degrees to the right. At the same time, the l enantiomer, with its equal and opposite left-twisting power, rotates that same light 10 degrees to the left. So, you have one force pushing the light one way, and an equally strong force pushing it the other way. It's a perfect tug-of-war, and guess who wins? Nobody!
The net result is that the light, after passing through the entire racemic mixture, experiences no overall rotation. It's like the movements cancel each other out completely. The light might get nudged left and then nudged right, but it ends up exactly where it started, direction-wise. This is why a racemic mixture is declared "not optically active." It's not that the individual molecules aren't doing anything; it's that their effects perfectly neutralize each other when present in equal amounts.
Isn't that a wonderfully simple and elegant explanation? It’s like a balanced equation in chemistry, where everything adds up to zero. This concept is often presented in introductory organic chemistry, and it's a fantastic way to introduce students to the concept of chirality and its consequences. It’s a moment where a seemingly complex idea becomes surprisingly clear and logical.
![[ANSWERED] A mixture of equal amounts of two enantiomers OA Racemic](https://media.kunduz.com/media/sug-question-candidate/20220506003049434003-4189330.jpg?h=512)
The beauty of this question lies in its simplicity and its ability to test fundamental understanding. It’s not about memorizing endless facts; it's about understanding a principle. When you see this question on an exam, it’s an invitation to show you understand the delicate balance of forces at the molecular level. It’s a chance to appreciate how nature, even at its smallest, can have such perfect symmetry that it cancels out observable effects.
So, the next time you hear about a racemic mixture or optical activity, don't feel overwhelmed. Think of it as a dance of light and molecules, where sometimes, equal and opposite moves lead to a perfectly still outcome. It’s a little bit of chemistry magic that explains a whole lot about the world around us, from medicines to materials. It’s a prime example of how even a seemingly "neutral" mixture can hold significant, albeit balanced, power.
The fact that this is a common exam question just highlights its importance. It’s a gateway to understanding more complex concepts in stereochemistry. It’s a foundational piece that, once grasped, opens up a whole new perspective on how molecules behave. So, if you ever get a chance, delve a little deeper into optical activity. You might find it surprisingly engaging!