Why Is Electrolysis Used To Extract Some Metals
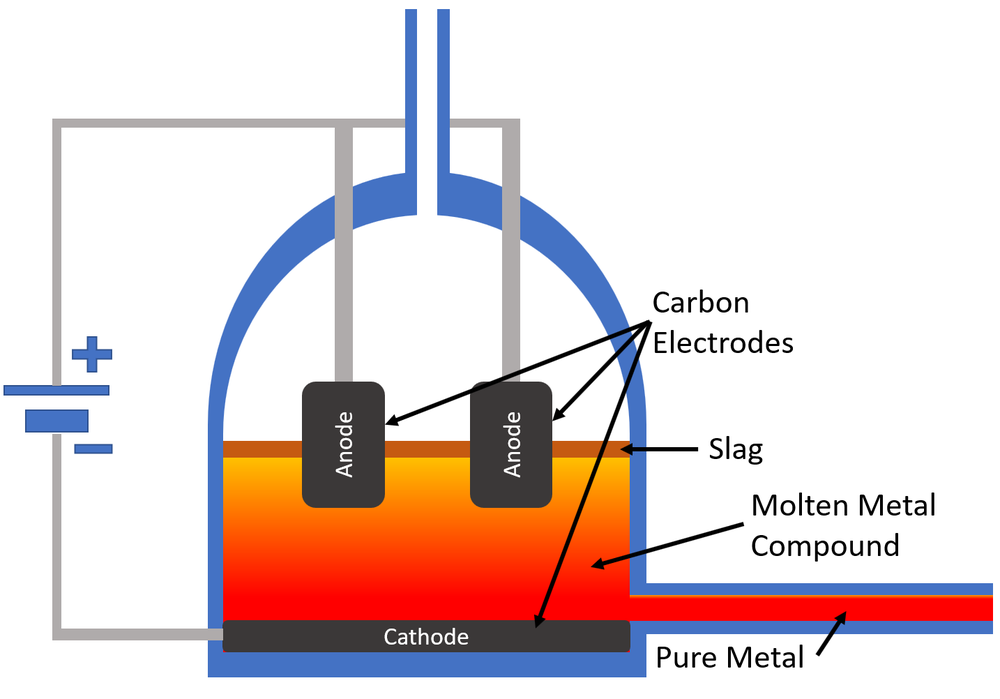
Ever looked at your trusty aluminum foil, the shiny copper wires in your phone charger, or even that fancy titanium bike frame and wondered, "How on earth did they get that metal out of the ground?" It’s a great question, and while some metals are pretty chill and practically fall out of rocks, others are a bit more… stubborn. They’re like those picky eaters who refuse to leave their comfy spot unless you really convince them. That’s where a rather zappy method called electrolysis comes in, and it’s surprisingly important for a lot of the stuff we use every single day.
Think of it like this: many of these super useful metals, when they’re found in nature, aren’t just hanging out as pure metal. Nope, they’re usually all bundled up with other elements, like oxygen or chlorine. Imagine a kid who’s really attached to their favorite teddy bear. You can’t just yank the teddy away, can you? You need a gentle, but firm, approach. Electrolysis is that approach for metals.
A Little Jolt of Genius
So, what exactly is this “electrolysis” thing? At its heart, it’s using electricity to break things apart. Picture it as using a tiny, controlled lightning bolt to persuade a stubborn metal atom to leave its buddies. It sounds dramatic, but it’s a really clever process that’s been around for ages.
Must Read
Imagine you have a salt dissolved in water. Not just any salt, but a salt made of a metal you want, like aluminum. You get a couple of electrodes – think of them as special rods – and stick them into this salty water. Then, you run electricity through them. One electrode is super positive (let’s call it the "grabber") and the other is super negative (the "pusher").
The metal atoms, which are a bit like little magnets in this situation, get excited by the electricity. The ones we want to extract will get attracted to the "grabber" electrode, and the unwanted stuff gets pushed away or attracted to the "pusher" electrode. It’s like a dance party, but with atoms and electricity!
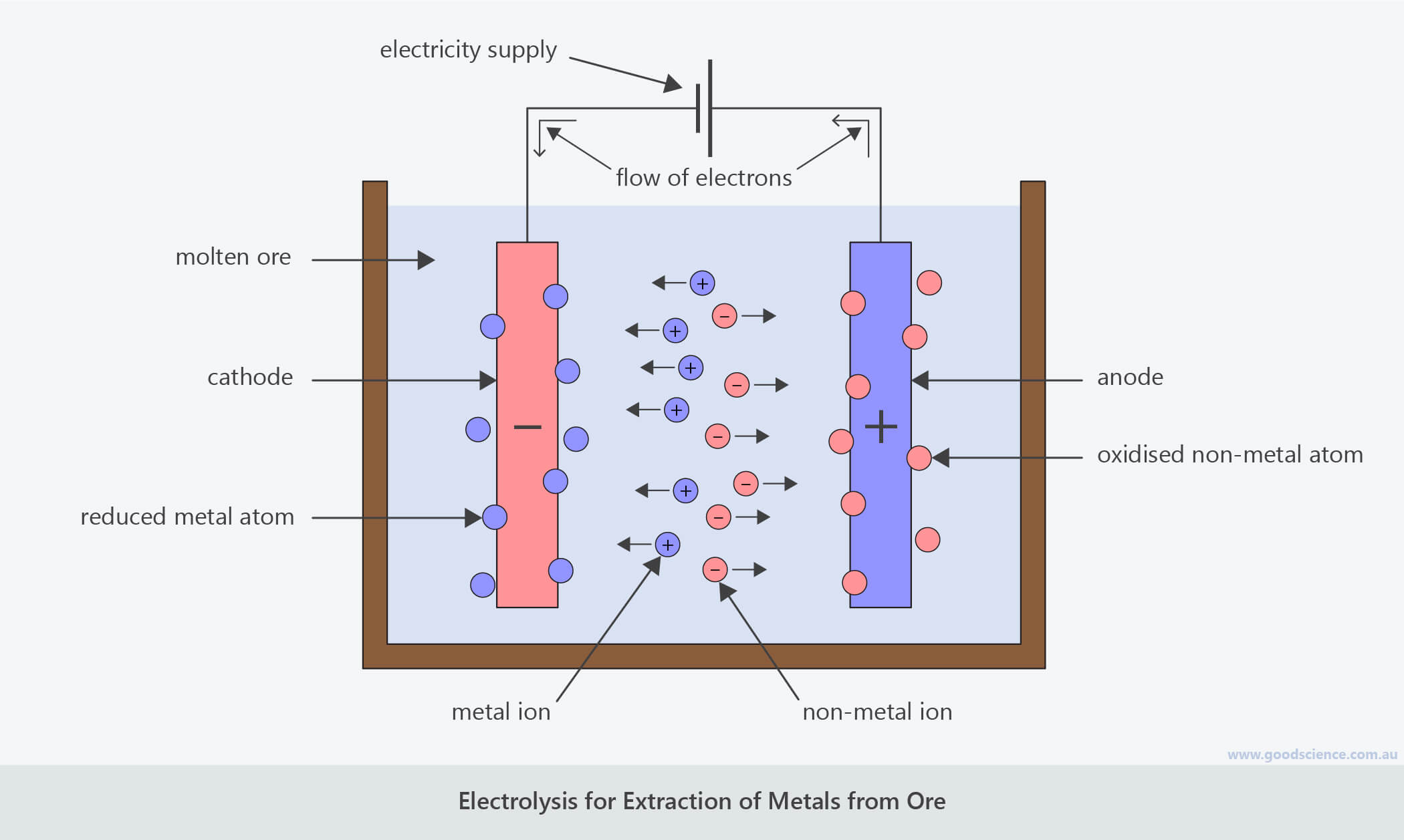
When the metal atoms reach the "grabber" electrode, they’re so happy to be there that they give up their extra bits (called electrons) and settle down as pure, shiny metal. Ta-da! You’ve just coaxed metal out of its mineral hiding spot.
Why Bother With All This Zapping?
Now, you might be thinking, "Why go through all this trouble? Can’t we just melt it or something?" Well, for some metals, like iron (which is used in most steel), heating it up in a big furnace does the trick. But for metals like aluminum, it’s a different story. Aluminum is incredibly useful. It’s lightweight, strong, and doesn’t rust easily. Think of all the soda cans you’ve recycled, the airplane parts that keep us flying, and the cookware in your kitchen – that’s all thanks to aluminum.
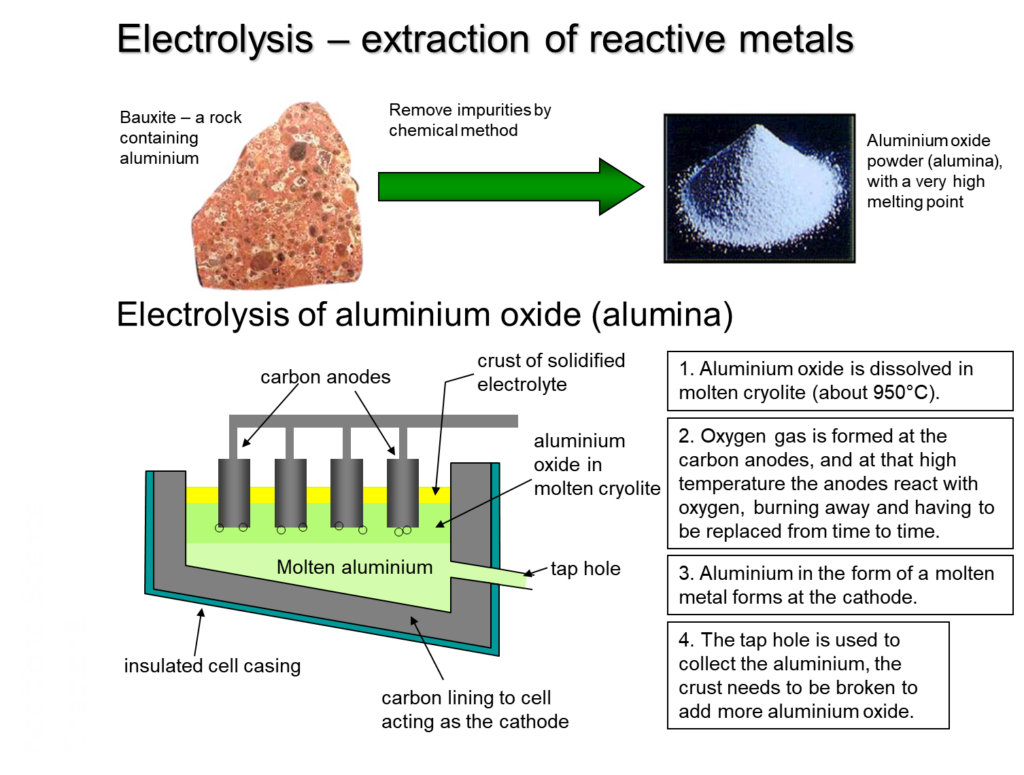
However, aluminum is really tightly bound to oxygen in its natural form, which is usually a rock called bauxite. Heating it up isn’t enough to break that strong bond. It’s like trying to pry open a piggy bank with just a gentle nudge. You need something more powerful, something that can truly get into those tight bonds.
Electrolysis is that powerful something. It’s the only practical way to get pure aluminum out of bauxite on a large scale. Without it, those soda cans would be a lot rarer, and your coffee pot might be made of something much heavier and less convenient. It’s the reason we have the lightweight, modern world we do.
Another metal that often uses electrolysis is magnesium. Magnesium is also super light and strong, making it fantastic for things like car parts, cameras, and even fireworks (for that dazzling sparkle!). Like aluminum, magnesium likes to hang out with other elements and needs a good electrical push to be set free. It’s found in seawater, which is pretty amazing – we’re basically pulling useful metals out of the ocean with electricity!
And let’s not forget titanium. This metal is a superhero of the material world. It’s incredibly strong, yet surprisingly light, and it doesn’t corrode. Think of the medical implants that help people walk again, the high-performance sporting equipment, and even parts of jet engines. Extracting titanium is a complex process, and electrolysis plays a crucial role in purifying it and getting it ready for all those amazing jobs. It’s like giving titanium its cape and boots so it can go off and do its heroic work.
It’s More Than Just Pretty Shine
So, why should you, the everyday reader, care about electrolysis? Well, it’s all about the stuff that makes our lives easier, safer, and more fun. Every time you use your smartphone, hop on a plane, or even enjoy a fizzy drink, there’s a good chance electrolysis was involved in getting the metals that make it possible.

It’s a behind-the-scenes hero. It allows us to harness the power of electricity to unlock valuable resources that are hidden within the Earth’s crust. It’s a testament to human ingenuity, finding clever ways to work with nature rather than against it.
Think about the future, too. As we look for lighter, stronger, and more sustainable materials, metals like aluminum and titanium will become even more important. Electrolysis will continue to be a key player in making these materials accessible and affordable. It’s not just about extracting metal; it’s about enabling innovation and building a better future, one zappy atom at a time.
So, next time you see a shiny aluminum foil or a strong titanium bike, give a little nod to electrolysis. It’s the unsung hero that helps bring these amazing materials from the ground to your hands, making our modern lives possible and, dare we say, a little bit brighter.


