Why Does Sodium Oxide Have A High Melting Point

Hey there, curious minds! Ever found yourself staring at a kitchen counter, maybe making a cup of tea, and suddenly wondering about the super-strong bonds that hold everyday stuff together? Today, we're diving into something that sounds a bit science-y but is actually all around us: sodium oxide and its surprisingly high melting point. Don't let the name scare you; think of it as a peek behind the curtain of how the world works, in a way that's as cozy as your favorite armchair.
So, what is this sodium oxide thing? Imagine tiny, invisible building blocks. Sodium oxide is made up of two types of these blocks: sodium, which you know from table salt (sodium chloride), and oxygen, the stuff we breathe. When these two get together, they form a kind of super-strong handshake, a bond that’s really hard to break. Think of it like those really, really stubborn magnets that snap together with a satisfying thunk and you can barely pull them apart. That's kind of what's happening at the atomic level with sodium oxide.
The Big Hug: Why Things Stick Together So Tightly
This "super-strong handshake" is called an ionic bond. It's like a tiny theft happening where one atom (sodium) gives away a little piece of itself, an electron, to another atom (oxygen). This act makes one positively charged and the other negatively charged, and opposites attract, right? They become like two magnets with their opposite poles facing each other, clinging on for dear life. In sodium oxide, you have lots and lots of these sodium and oxygen bits, all arranged in a neat, repeating pattern, like a perfectly stacked pile of LEGO bricks. Each little brick is holding onto its neighbors with all its might.
Must Read
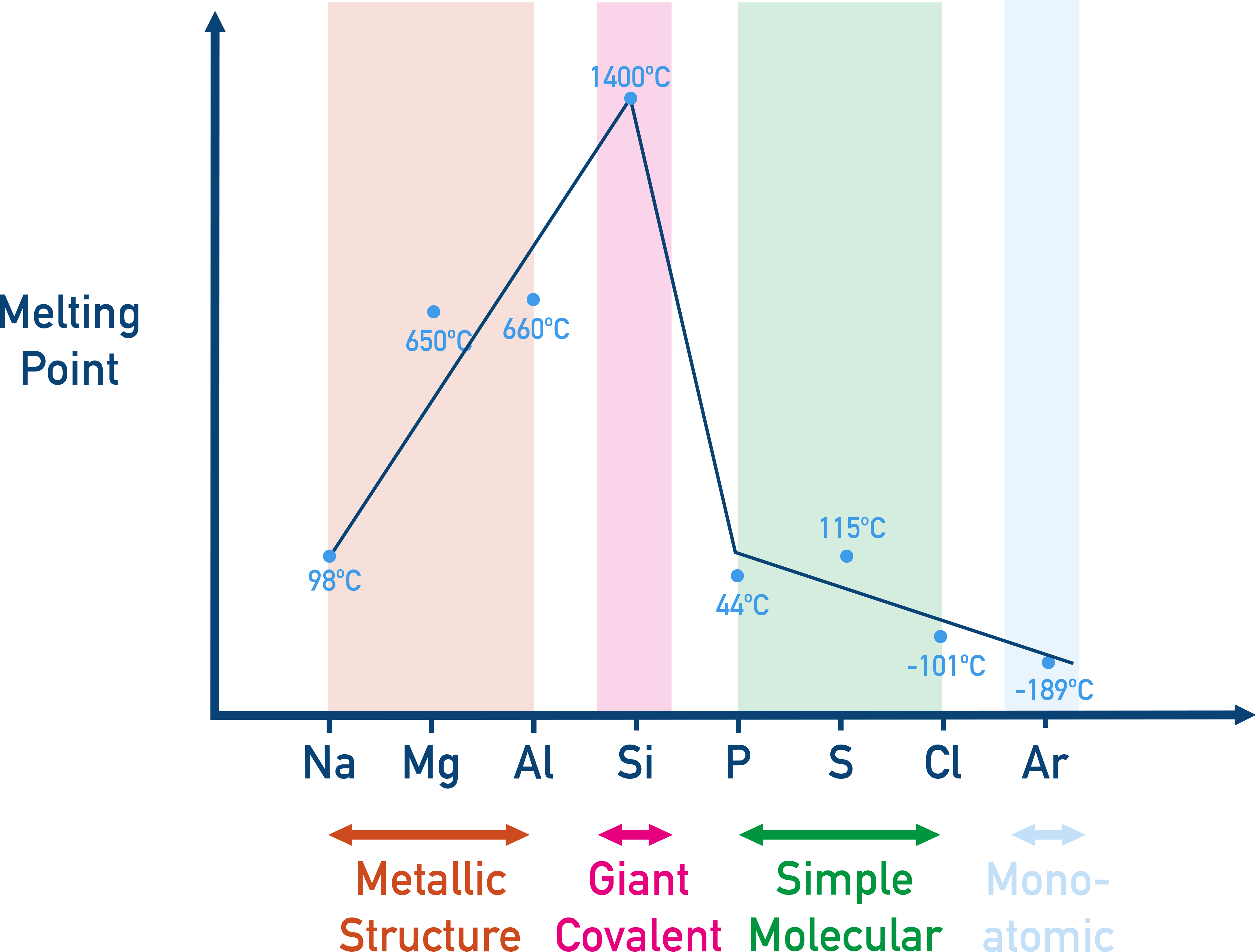
Now, imagine trying to break down that LEGO castle. You'd need a lot of force, wouldn't you? That's exactly why sodium oxide has such a high melting point. Melting isn't just about making something go from solid to liquid; it's about breaking those bonds. It's about getting those tiny building blocks to loosen their grip and start sliding past each other. For sodium oxide, those ionic bonds are so strong, like a superhero holding a building together during an earthquake, that it takes an enormous amount of heat to make them let go.
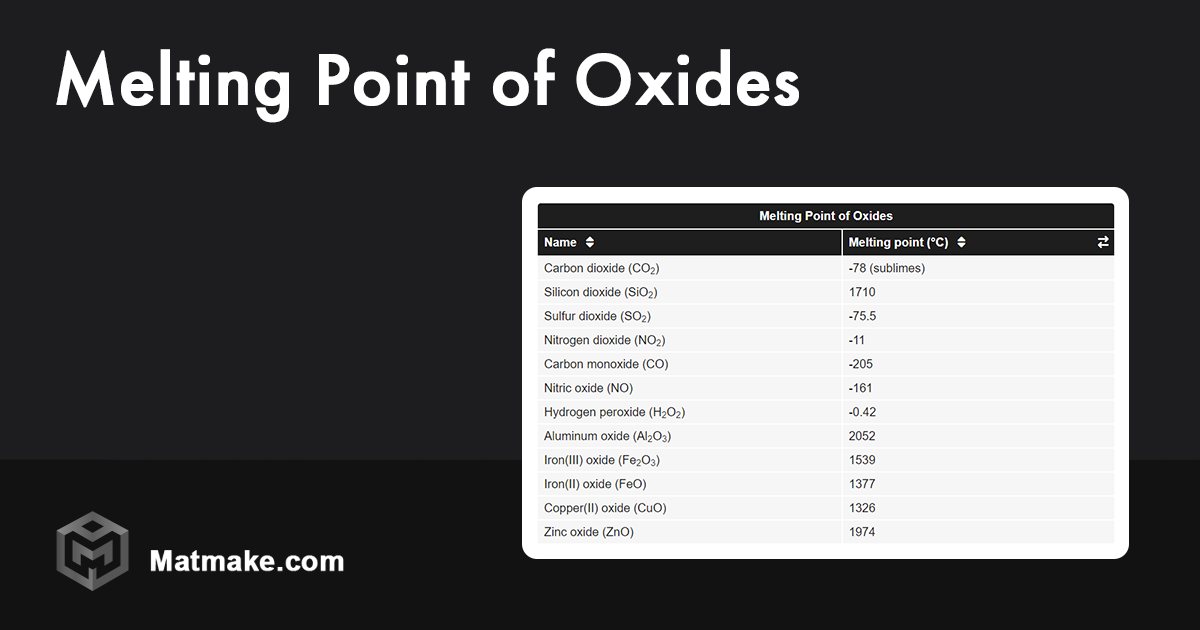
We’re talking temperatures that would make your oven look like a chilly room. We're talking about reaching over 1100 degrees Celsius. To put that in perspective, if you were trying to melt sodium oxide in your kitchen oven, you'd need to turn it up hotter than the surface of an active volcano! You’d probably melt the oven itself before the sodium oxide even thought about becoming a puddle.
Everyday Magic: Where Does This Matter?
Okay, okay, you might be thinking, "That's interesting, but why should I care about hot rocks?" Well, it's all about understanding the building blocks of our world and how they behave, which often impacts things we use every single day, sometimes in surprising ways.
Think about glass. Yep, glass! Many types of glass are made using silica (which is basically sand) and other ingredients, and the way these ingredients fuse together, and how they withstand heat, is related to similar strong bonding principles. When you heat up the raw materials for glass, you're essentially trying to overcome these powerful attractions to create that smooth, transparent material we use for windows, bottles, and smartphone screens.
Or consider ceramics. Those pretty tiles on your bathroom floor or the sturdy mug you drink your morning coffee from? They often involve compounds with strong ionic bonds, giving them their durability and resistance to heat. If they didn't have these tough structures, your mug would warp the moment you poured in hot coffee, and your tiles would crumble under your feet!
Even in more industrial settings, understanding these high melting points is crucial. In manufacturing, scientists and engineers need materials that can withstand extreme temperatures. If they're designing a furnace or a piece of equipment that will be used in a really hot environment, they need to choose materials that won't melt or degrade. Sodium oxide, or compounds with similar bonding, are part of the puzzle in creating these robust materials.
A Little More About Those Tiny Charges
Let’s circle back to those charged particles, the sodium ion (Na+) and the oxide ion (O2-). The "2-" on the oxide ion tells us it's holding onto two extra electrons, making it a bit greedier than the sodium ion, which has lost one. This difference in charge, and the fact that they're packed so closely and regularly, creates a really strong network of attraction. It's like a whole village where everyone is holding hands really tightly. To get anyone to move, you'd have to break a lot of those handholds simultaneously.

Imagine a dance floor packed with couples. If they're all holding hands in a tight circle, it's going to take a lot of effort to get any one couple to break away and start dancing solo. The more couples there are, and the tighter they're holding hands, the more energy (heat!) you'd need to get the party started properly.
This strong attraction isn't just about melting. It also affects other properties, like how easily sodium oxide dissolves in water (it doesn't dissolve easily, by the way!) or its electrical conductivity. But for today, the melting point is our star. It's a direct consequence of those incredibly tenacious ionic bonds.

Why It's Cool to Know This Stuff
So, why should you, the everyday awesome human, care about sodium oxide's melting point? Because it’s a tiny window into the fundamental forces that shape our physical world. It helps us appreciate the materials around us – the glass in our windows, the ceramic in our sinks, the concrete in our sidewalks. It's the science behind why some things are incredibly tough and heat-resistant, while others melt with a gentle nudge.
It’s like understanding a little bit of magic. You see a beautiful glass vase, and now you know that its creation involved bringing together elements that, at a microscopic level, were locked in a super-strong embrace. It's a testament to the power of nature's building blocks and how we, as humans, have learned to harness and shape them.
Next time you’re enjoying a warm drink in your favorite ceramic mug, or looking out through a crystal-clear window, you can have a little mental nod to those tiny, determined sodium and oxygen atoms, locked in their high-temperature handshake. It’s a small thing, but it's a reminder that even the most ordinary objects have extraordinary science behind them!

,h_400,q_80,w_640.png)
