Why Does Graphite Have A High Melting Point

Ever stared at a pencil lead? That’s graphite doing its thing. It’s pretty ordinary stuff, right? Used for writing, drawing, making marks.
But here's a secret: graphite is a bit of a diva. It’s incredibly tough when it comes to heat. Seriously tough.
Most things melt when they get hot. Butter, chocolate, ice cream on a sunny day. They just… goooey.
Must Read
But graphite? It just shrugs. "Oh, you think this is hot? Cute."
Why is it such a heat-wimp, or rather, a heat-survivor? It’s all about its atomic structure. Think of it like a super-organized city.
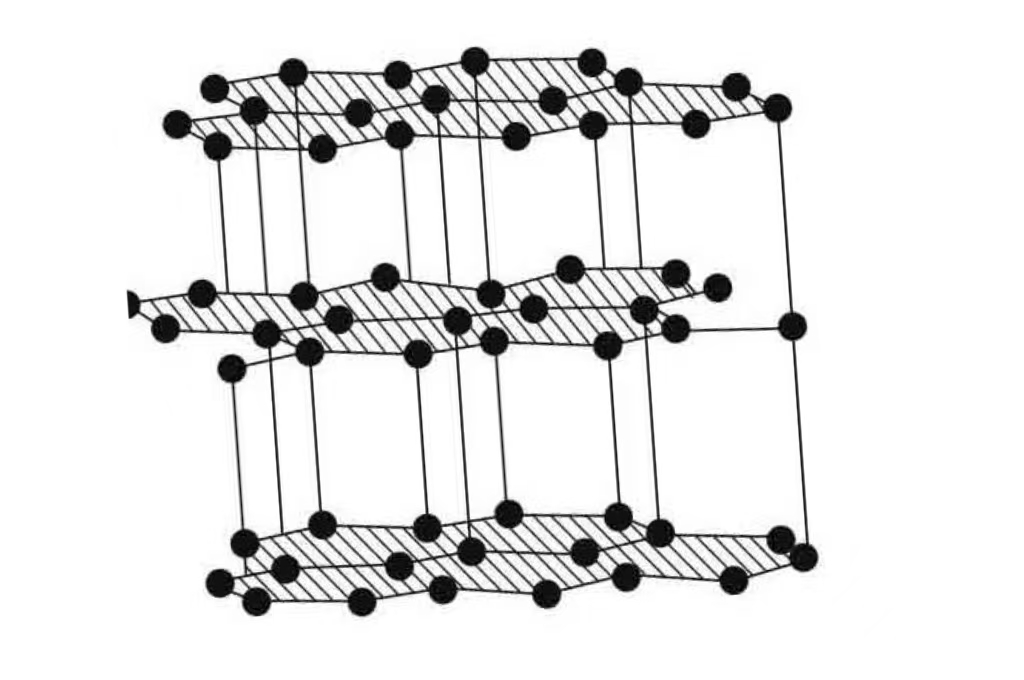
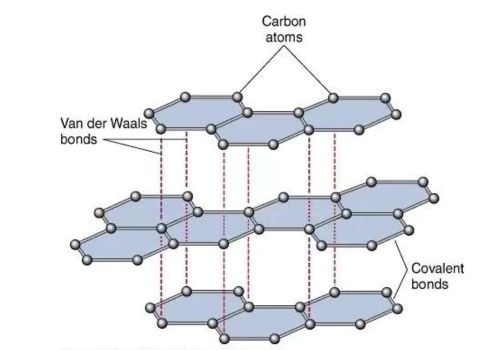
In graphite, the carbon atoms are arranged in flat layers. These layers are like super-strong hexagonal grids. They are locked together really tightly.
Imagine a bunch of tiny hexagonal trampolines. All stacked neatly on top of each other. That's kind of what graphite looks like, microscopically speaking.
The carbon atoms within each layer are bonded together with what scientists call covalent bonds. These are the strongest type of chemical bonds. They are like super-glue for atoms.
These covalent bonds are incredibly difficult to break. They require a tremendous amount of energy. Energy that heat provides.
So, when you try to melt graphite, you're essentially trying to break these super-glue bonds. It’s like trying to pull apart a brick wall with your bare hands.
Even though the layers can slide past each other (that’s why pencils write so smoothly!), the bonds within the layers are the real challenge. They hold the fort.
To melt graphite, you need to reach temperatures of around 3,652 degrees Celsius. That's about 6,600 degrees Fahrenheit.

To put that in perspective, the surface of the sun is about 5,500 degrees Celsius. So, graphite is tougher than the sun's surface. Mind. Blown.
Your oven at home? It maxes out around 250-300 degrees Celsius. It wouldn't even tickle graphite.
Think about it. You can use a pencil in a kiln. You can use it in a furnace. And it will just sit there, being all black and smug.
This high melting point isn't just a fun fact for trivia night. It makes graphite incredibly useful. It’s used in places where things get really, really hot.
Like in the brakes of race cars. Or in the nozzles of rocket engines. These things experience insane temperatures. Graphite is like their cool, unflappable friend.
It’s also used in crucibles, which are special pots used for melting metals. You can’t use just any old pot for that, right? It would melt first.
So, while you’re scribbling notes or doodling, remember the extraordinary power hidden within your humble pencil lead. It's a tiny piece of material that laughs in the face of extreme heat.
It's almost like it’s saying, "Is that all you’ve got? I’ve seen hotter Tuesdays."
The weak links in the graphite structure are the forces between the layers. These are called van der Waals forces. They are much weaker.
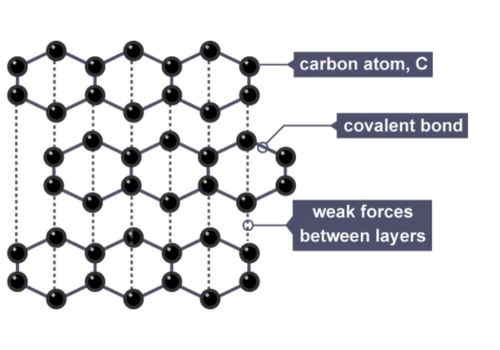
These forces are like a gentle handshake between the layers. They allow the layers to glide. That's what gives graphite its slipperiness.
But melting is a different beast. Melting requires breaking those strong internal bonds. The ones holding the hexagons together.
It’s like having a perfectly arranged deck of cards. The cards within each row are stuck together really well. But the rows themselves can slide apart easily.
When you heat graphite, you’re giving those internal bonds a serious workout. They resist. They refuse to budge.
Until, eventually, at those ridiculously high temperatures, they finally give up the ghost. But it takes a heck of a lot of persuading.
So, next time you're using a pencil, give it a little nod of respect. It's not just a writing tool. It’s a miniature heat-defying champion.
It’s a material that has mastered the art of staying solid under pressure. Extreme pressure, in this case.
And it does it all with such quiet dignity. No dramatic melting, no fiery demise. Just a stoic refusal to surrender to the heat.
It’s a lesson in resilience, really. A lesson taught by a pencil lead. Who would have thought?
The structure of graphite is called a hexagonal lattice. It’s a beautiful, repeating pattern. Like a perfectly tiled floor.
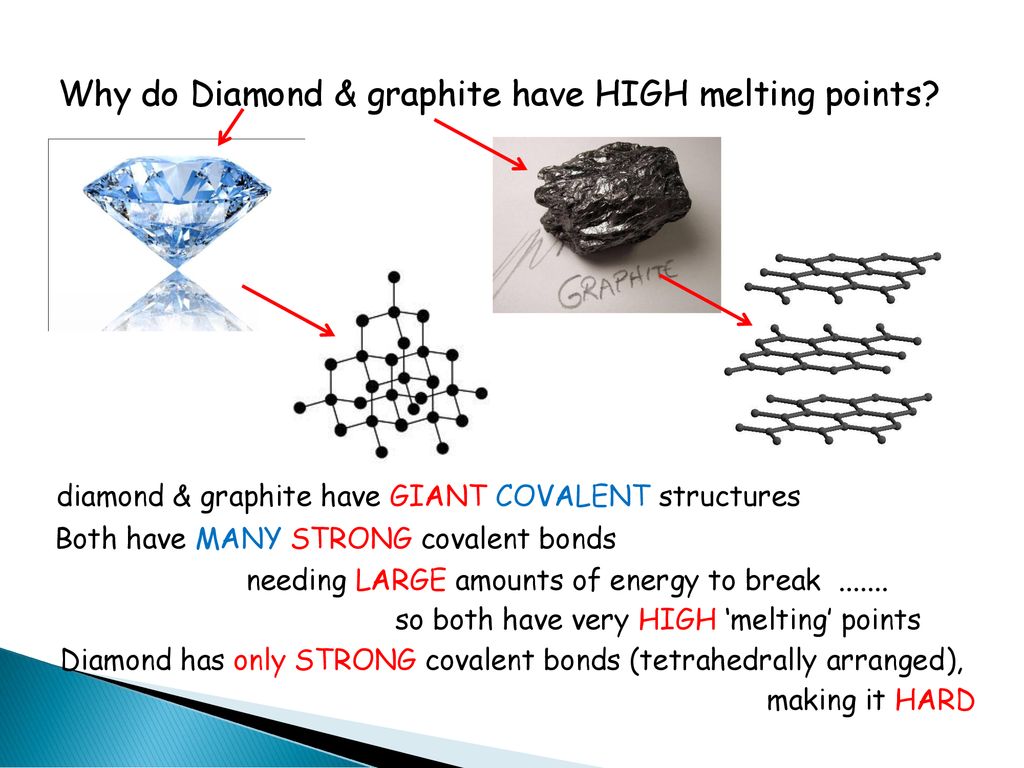
These repeating units are held together by those strong covalent bonds. They are the backbone of the structure.
When heat energy is applied, the atoms vibrate. In most substances, this vibration eventually shakes the bonds apart.
But in graphite, the covalent bonds are so robust, they can absorb a lot of this vibrational energy. They just wobble a bit.
It’s like a sturdy building that can withstand a strong earthquake. It might shake, but it won’t crumble easily.
The layers of graphite are arranged in an ABAB pattern. This stacking is efficient and stable.
The van der Waals forces between these layers are weak. This is why graphite is soft and can be used as a lubricant.
But again, melting is about breaking those strong internal bonds. It’s not about separating the layers.
So, when people talk about melting graphite, they are often referring to a phase change. Where it transforms into something else.
In some extreme conditions, graphite can turn into diamond. That's another story for another day. But it also requires immense heat and pressure.

The fact that graphite has such a high melting point is a testament to the strength of chemical bonds. It’s a fundamental principle of chemistry at play.
And it all starts with those humble carbon atoms, arranged just so. In their perfect, hexagonal little worlds.
So, the next time you draw a line, remember you're wielding a material that’s practically fireproof. It’s a small, black superhero.
It’s easy to underestimate things we see every day. Like a pencil. Or the power of strong chemical bonds.
But graphite proves that even the most ordinary-looking things can have extraordinary properties. They just need the right structure.
And a whole lot of heat to even think about breaking them. It’s a quiet, smoky strength.
It’s a material that makes us look good when we’re trying to look smart. By drawing diagrams. And it doesn’t melt under pressure.
So, there you have it. Your pencil lead is a tiny bit of a legend. A legend of heat resistance.
It’s the reason why some things work, and why some things are just plain… well, not graphite. And that’s a good thing.
A really good thing, if you ask me. Because who wants a melting pencil? Not me, that’s for sure.