Why Does Electronegativity Decrease Down A Group

Imagine atoms as tiny, bustling cities. Each city has a center, the nucleus, which is like the town hall, packed with important residents (protons and neutrons). Around this town hall, orbiting like commuters on a busy highway, are the electrons. Now, these electrons aren't just passive travelers; they have preferences, and they're attracted to the town hall's positive vibe (thanks to those protons).
The thing we're talking about today is electronegativity. Think of it as an atom's "clinginess" to its electrons, or how much it wants to hog them closer to its center. It's like a popularity contest for electrons – some atoms are super popular and can pull electrons towards them with all their might, while others are a bit more chill and let their electrons wander a bit further.
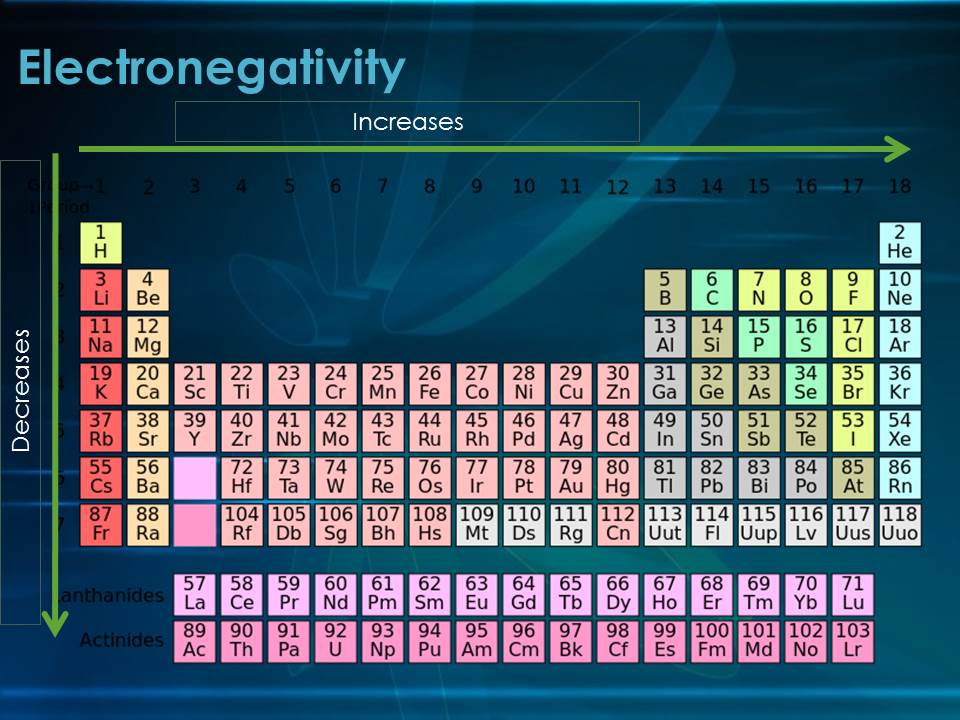
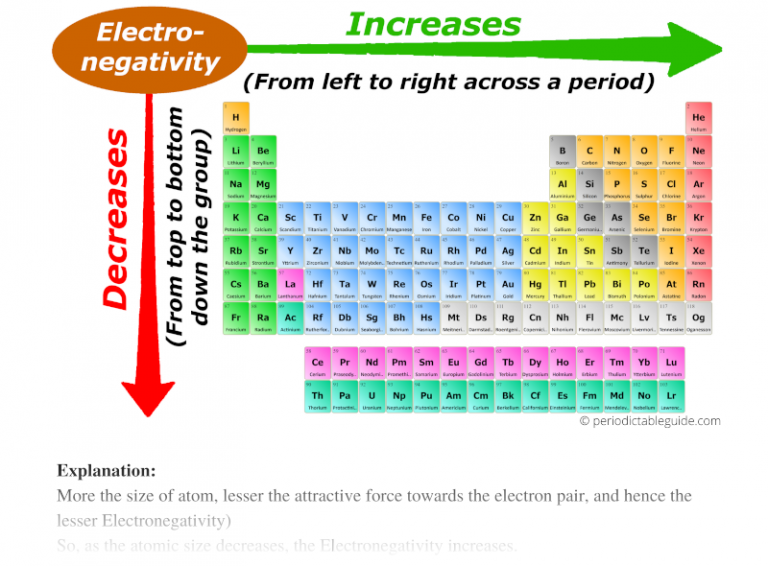
Now, let's talk about the periodic table. It's like a giant family photo album for all these atom cities. The table is organized into rows called periods and columns called groups. Today, we're going on a little road trip down one of these groups. Imagine you're visiting a family, and you start at the top of the house and keep going down to the basement.
Must Read
At the very top of a group, you have the most energetic, high-flying atoms. These are like the penthouse suites of the atomic world. They have their town hall (nucleus) and their commuters (electrons) zipping around relatively close by. Because the electrons are so close to the positively charged town hall, they feel a strong pull. It's like standing right next to a magnet – you feel its tug quite intensely. These top-of-the-group atoms are absolute electron-hogs. They're the ones who will shamelessly snatch an electron if they get the chance, making them super electronegative.
But then, as we travel down the group, things start to get… bigger. Think of it like adding more floors to our atomic building. Each new floor represents another electron shell, another layer of commuter highway. So, as you go down a group, each atom city gets a new set of electron shells added. The town hall (nucleus) is still there, but the commuters (electrons) are now taking longer routes, and there are more layers between them.

This is where the humor and maybe even a touch of heartwarming realization comes in. Imagine our nucleus is still the enthusiastic town hall mayor, eager to keep all the citizens (electrons) close. But as the city grows and adds more and more levels, it becomes harder for the mayor's voice to reach the furthest outposts. There are more buildings, more roads, and more traffic jams in between.
The electrons at the very outer edges of these bigger atoms are quite far from the town hall's strong, positive pull. It's like trying to hear your mom calling you from the other side of a huge mansion. You can still hear her, but the voice is definitely fainter. There are also more inner "walls" or electron shells acting as shields, buffering the positive pull of the nucleus from the outer electrons. These inner shells are like the security guards at the entrance of the mansion; they block some of the direct influence.

So, what happens to the atom's "clinginess"? It starts to fade. The outer electrons, being further away and a bit shielded, aren't as easily "clung" to by the nucleus. It's like the mayor is trying to hold onto a kite with a really, really long string in a gusty wind – the kite can just drift further away. The atom's ability to attract electrons from other atoms also weakens. It's just not as good at playing "keep away" anymore.
It’s like a parent trying to keep their teenagers close. When they’re little, they’re right there. As they grow up and have their own lives, they naturally get a bit further away, even though the love (the nucleus's pull) is still there!
Electronegativity - Periodic Trends
This is why electronegativity decreases as you go down a group. The atoms just get bigger, with more electron shells separating the outer electrons from the positively charged nucleus. The pull isn't as strong at that distance, and the shielding effect of the inner electrons makes it even less so. These larger atoms are less enthusiastic about grabbing extra electrons; they're more content with their own electron family, even if they're spread out a bit.
It’s a gentle reminder that as things grow and expand, the intensity of the central force can sometimes diminish. It’s a fundamental dance of attraction and distance, and it plays out beautifully across the rows and columns of the periodic table, making chemistry a never-ending, fascinating story.