Why Does Copper Oxide Have A High Melting Point
.jpg)
So, you're curious about copper oxide, huh? And its super-duper high melting point? Like, what’s the big deal? Let’s grab a virtual coffee, settle in, and spill the beans, shall we?
Because, honestly, who wouldn't be a little intrigued by a substance that laughs in the face of your average stovetop heat? It's not like your grandma’s butter, melting into a puddle at the slightest warmth. Nope. Copper oxide is built different.
Imagine trying to melt a brick. Okay, maybe not that extreme, but you get the picture. It’s a bit of a stubborn character when it comes to changing its state from solid to liquid. And we’re talking seriously high temperatures here. We’re not even close to boiling water territory, people. We’re in the realm of things that glow red, then orange, then maybe even a sort of fiery white.
Must Read
So, what’s the secret sauce? Why is this common compound such a powerhouse of heat resistance? It all boils down to its inner workings, you know, its molecular dance. It’s not just a random jumble of atoms; there’s a whole lot of structure going on.
Think of it like building with LEGOs. You can make a wobbly tower that falls over easily. Or, you can build something super solid, interlocked, and tough. Copper oxide, my friend, is definitely in the latter category. It’s got this incredibly strong grip on its bits and pieces.
The Atomic Hug of Death (Not Really, But Close!)
Okay, "death" is a bit dramatic, but you get it. The bonds between the copper and oxygen atoms in copper oxide are seriously, seriously strong. We’re talking about ionic bonds, which are like the ultimate commitment in the chemical world. They’re not just a casual handshake; it’s more like a super-glue embrace that’s really hard to break.
These bonds form a crystal lattice. Picture a perfectly organized grid, like a super-neat spreadsheet, but in 3D. Every copper atom is surrounded by oxygen atoms, and vice-versa, all held together by these powerful ionic attractions. It's a real neighborhood lock-in, where everyone is tightly packed and not keen on moving out.
To melt something, you need to give its atoms enough energy to break free from this organized structure. You need to overcome those strong attractions. And with copper oxide, that’s a tall order. It takes a massive amount of energy, which translates to a super high temperature, to get those atoms wiggling and jiggling enough to say, "Okay, fine, I’ll move over there."

Cupric Oxide vs. Cuprous Oxide: A Tale of Two Melting Points
Now, here’s where it gets a tiny bit more nuanced. You can have different types of copper oxide. The most common ones are cupric oxide (CuO) and cuprous oxide (Cu₂O). And guess what? They have slightly different melting points. It’s like having two slightly different recipes for the same dish – close, but not identical.
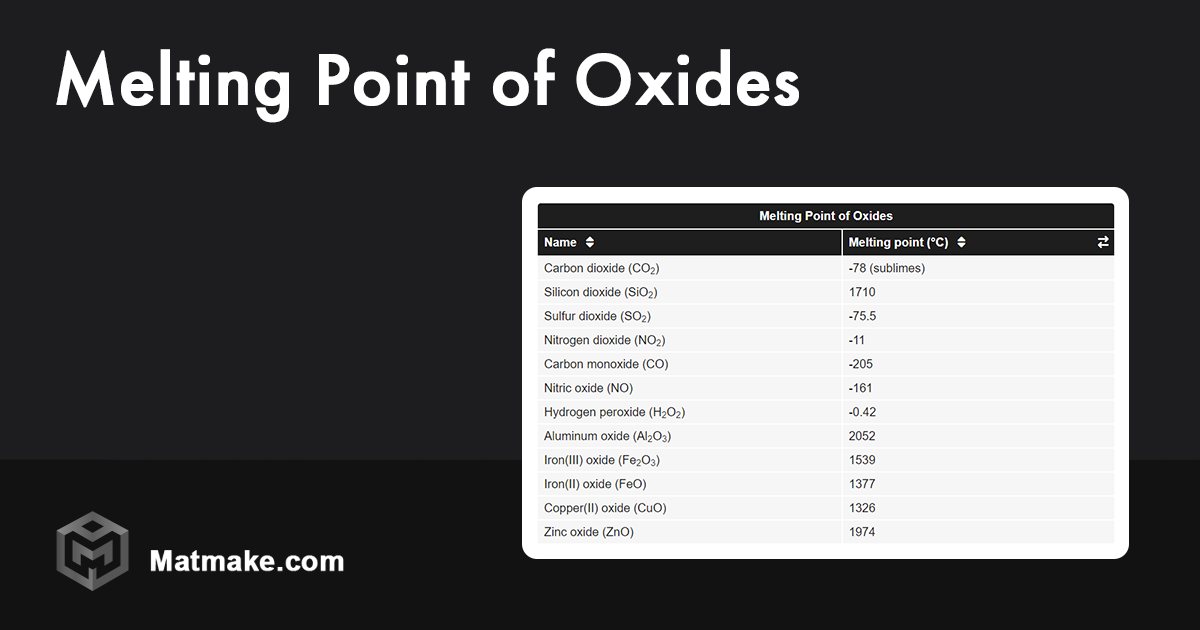
Cupric oxide (CuO) is the one that typically melts around a whopping 1326 degrees Celsius (that’s like 2419 Fahrenheit, for those who prefer the fahrenheit scale – or just like big numbers!). That’s hot. Like, really hot. Hotter than any oven you've probably ever used, unless you're secretly a blacksmith working in a volcano.
Cuprous oxide (Cu₂O), on the other hand, has a melting point that’s even higher, often cited as decomposing at around 1235 degrees Celsius before it truly melts. But wait, isn’t that lower? Ah, but this is where the "decomposition" part comes in. Sometimes, instead of a clean melt, these compounds decide to break down into other things at really high temperatures. It’s like they’re saying, "You want me to melt? Nah, I’d rather become something else!" It's a bit of a chemical tantrum, really.
So, while CuO has a straightforward, sky-high melting point, Cu₂O does its own thing. But the underlying principle of those strong ionic bonds is still the main character in this melting point saga.
Why Does It Matter, Anyway?
Okay, so it melts at a bazillion degrees. Big deal, right? Well, actually, yes! This high melting point isn't just a quirky fact for your next pub quiz. It's actually super useful.
Think about all the places where you need materials that can handle serious heat. We’re talking about industrial furnaces, high-temperature ceramics, and even some specialized electronics. If you’re trying to create something that needs to withstand extreme conditions, you need materials that won’t just give up and melt into a gooey mess.
![Melting points of metal oxides [Refs. 8,79] | Download Table](https://www.researchgate.net/profile/Benjaram_Reddy2/publication/286179231/figure/tbl1/AS:667069817294864@1536053246922/Melting-points-of-metal-oxides-Refs-8-79.png)
Copper oxide, with its robust structure and those unyielding ionic bonds, is a fantastic candidate for these kinds of applications. It's like the reliable workhorse that just keeps going, no matter how much pressure (or heat!) you throw at it. It's not going to sag or deform when things get tough.
Plus, it’s relatively abundant and not too expensive, which is always a bonus in the world of industrial materials. Imagine if the only thing that could withstand that much heat was something mined from the moon and cost a gazillion dollars per ounce. That would be… less than ideal.
The Crystal Lattice: A Super-Strong Scaffold
Let’s dive a little deeper into this crystal lattice idea. It’s not just a vague concept; it’s the physical manifestation of those strong ionic bonds. In CuO, for example, the copper ions (Cu²⁺) and oxide ions (O²⁻) arrange themselves in a specific, highly ordered structure. It’s a bit like a perfectly stacked pyramid, but repeated endlessly in all directions.
This arrangement maximizes the electrostatic attraction between the oppositely charged ions. Think of it as each positive ion being surrounded by negative ions, and vice versa, all pulling on each other with all their might. There’s very little wasted space, and everything is locked into its position.
To melt it, you need to inject so much thermal energy into this system that the ions start vibrating so violently that they can overcome these powerful attractions. It's like trying to shake a tightly packed crowd of people so hard that they all decide to go their separate ways. It takes a lot of effort!

The strength of these attractions is directly related to the charges of the ions and the distance between them. In copper oxide, the charges are quite significant (2+ and 2- in CuO), and the distances are relatively small, leading to a very strong overall attraction. This is why it takes so much heat to break them down.
Beyond Just Melting: Other High-Temperature Talents
But it's not just about the melting point, is it? Copper oxide has other tricks up its sleeve when it comes to high temperatures. It's generally quite stable at elevated temperatures, meaning it doesn't readily decompose or react with its surroundings unless you push it to extreme limits.
This stability is crucial in many manufacturing processes. For instance, in the production of certain types of glass and ceramics, copper oxide can be used as a pigment or as a flux. Its ability to withstand the high temperatures involved in firing these materials without degrading is key to achieving the desired properties and colors.
Imagine adding a colorant to your pottery clay, firing it in a kiln hotter than the surface of Venus, and your colorant just vanishes or turns into something completely different. That would be a disaster! Copper oxide, though, with its stoic resilience, is up to the task.
It’s like having a superhero material that doesn't flinch when the heat is on. It just performs, reliably and consistently, even in the most demanding environments. And that’s pretty darn cool, if you ask me.
The Magic of Ionic Bonding, Simplified
Let's try and simplify this ionic bonding thing even further. Imagine you have two magnets. One is a positive charge (copper), and the other is a negative charge (oxygen). When you put them together, they snap into place and hold on tight. Now imagine you have a whole bunch of these magnets, arranged in a neat grid, all pulling on each other. To pull them apart, you'd need to exert a massive amount of force, right?
That's essentially what's happening at the atomic level in copper oxide. The copper atoms and oxygen atoms have opposite electrical charges, and they're attracted to each other very strongly. This attraction, the ionic bond, is what holds the solid structure together. The more energy (heat) you put in, the more the atoms vibrate, but they resist breaking free from this magnetic-like pull.
It's this inherent, powerful attraction that makes copper oxide so stubborn when it comes to melting. It's not just a matter of getting them to wiggle; it's about overcoming that fundamental electrostatic hug that keeps them locked in their crystalline embrace.
So, What's the Takeaway?
Basically, copper oxide has a high melting point because it’s built like a fortress. It’s got incredibly strong atomic bonds, specifically ionic bonds, that hold its structure together with an iron (or, well, copper and oxygen) grip. These bonds create a highly ordered crystal lattice that requires a tremendous amount of energy – hence, a very high temperature – to break down.
It’s not magic; it’s just clever chemistry and physics at play. And the fact that it’s so tough at high temperatures makes it a valuable player in various industrial and technological fields. It’s a material that truly stands the heat!
So, next time you hear about copper oxide and its melting point, you can nod sagely and think, "Ah yes, the strong ionic bonds and the super-organized crystal lattice." You’ll be the smartest person at the coffee shop, I guarantee it. Or, at least, you'll have a much cooler explanation for why it's so darn hard to melt.
Isn’t science just fascinating? It’s like a constant puzzle, and this high melting point is just one piece of a much bigger, very hot picture. Cheers to enduring materials and the reasons behind their awesomeness!