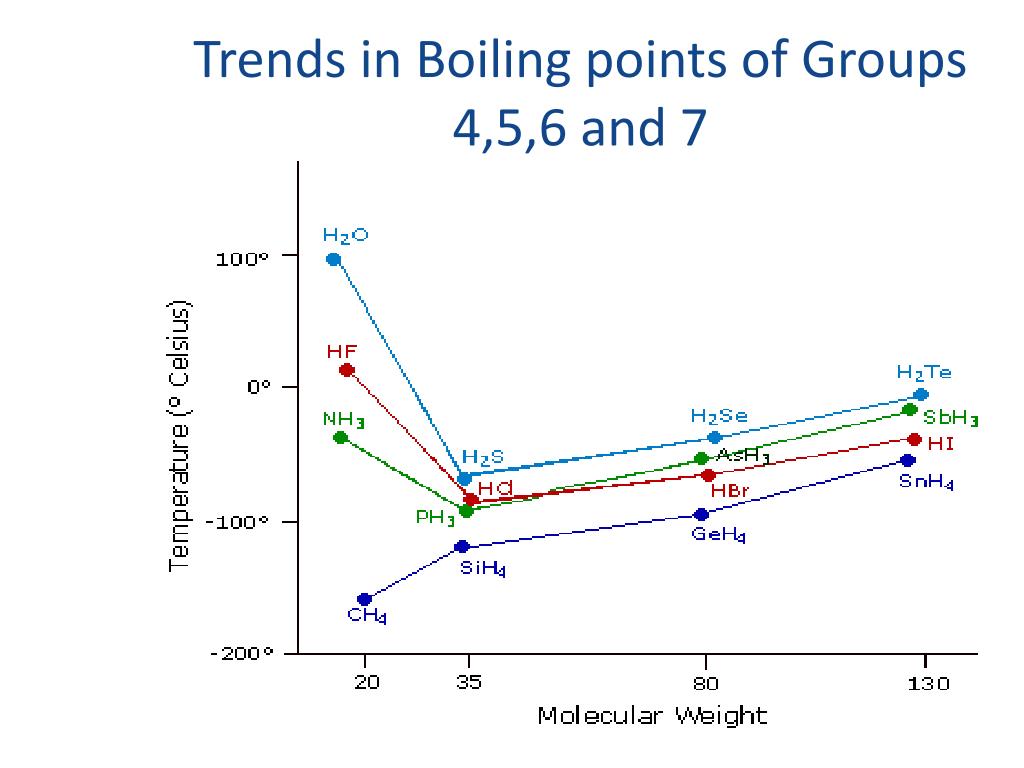
Why Does Boiling Point Increase Down Group 7

Ever find yourself staring at a pot of water, perhaps contemplating the existential dread of laundry day or, more likely, wondering what to make for dinner? Well, let's take a moment to gently pivot our gaze from the mundane to the magnificent. Today, we’re dipping our toes into the fascinating world of chemistry, specifically, the rather intriguing behavior of Group 7 elements – you know, those lively halogens like fluorine, chlorine, bromine, and iodine. They’re the quirky characters of the periodic table, and they’ve got a little secret that makes them behave in a rather predictable, and dare we say, cool way. It’s all about their boiling point, and how it likes to take a little hike as you go down the group. Intrigued? You should be!
Think of it like this: imagine a bunch of friends heading out for a weekend getaway. Some are light and breezy, happy to travel with just a small backpack. Others need a whole suitcase, maybe even a U-Haul, to haul all their essentials. In the chemical world, these “essentials” are called intermolecular forces – the invisible sticky stuff that holds molecules together. And for our Group 7 pals, these forces get progressively stronger as we move down the line.
The Unseen Hugs: Van der Waals Forces Explained
So, what exactly are these intermolecular forces? For the diatomic molecules of Group 7 (meaning they exist as pairs, like F₂, Cl₂, Br₂, and I₂), the primary players are something called Van der Waals forces. Now, don't let the fancy name scare you! These forces are essentially temporary, weak electrical attractions that arise because electrons, those zippy little particles whizzing around atoms, aren't always perfectly evenly distributed. At any given moment, they might clump up on one side of a molecule, creating a fleeting, temporary dipole.
Must Read
This temporary dipole in one molecule can then nudge the electrons in a neighboring molecule, inducing a similar, temporary dipole. Voilà! You’ve got a weak attraction, like a polite little handshake between molecules. It’s not a strong, unbreakable bond, but it’s enough to keep them from drifting off into the ether all on their own. Think of it as the subtle social graces that keep a crowd from scattering at a party.
Size Matters: The Bigger They Are, The Stronger the Grip
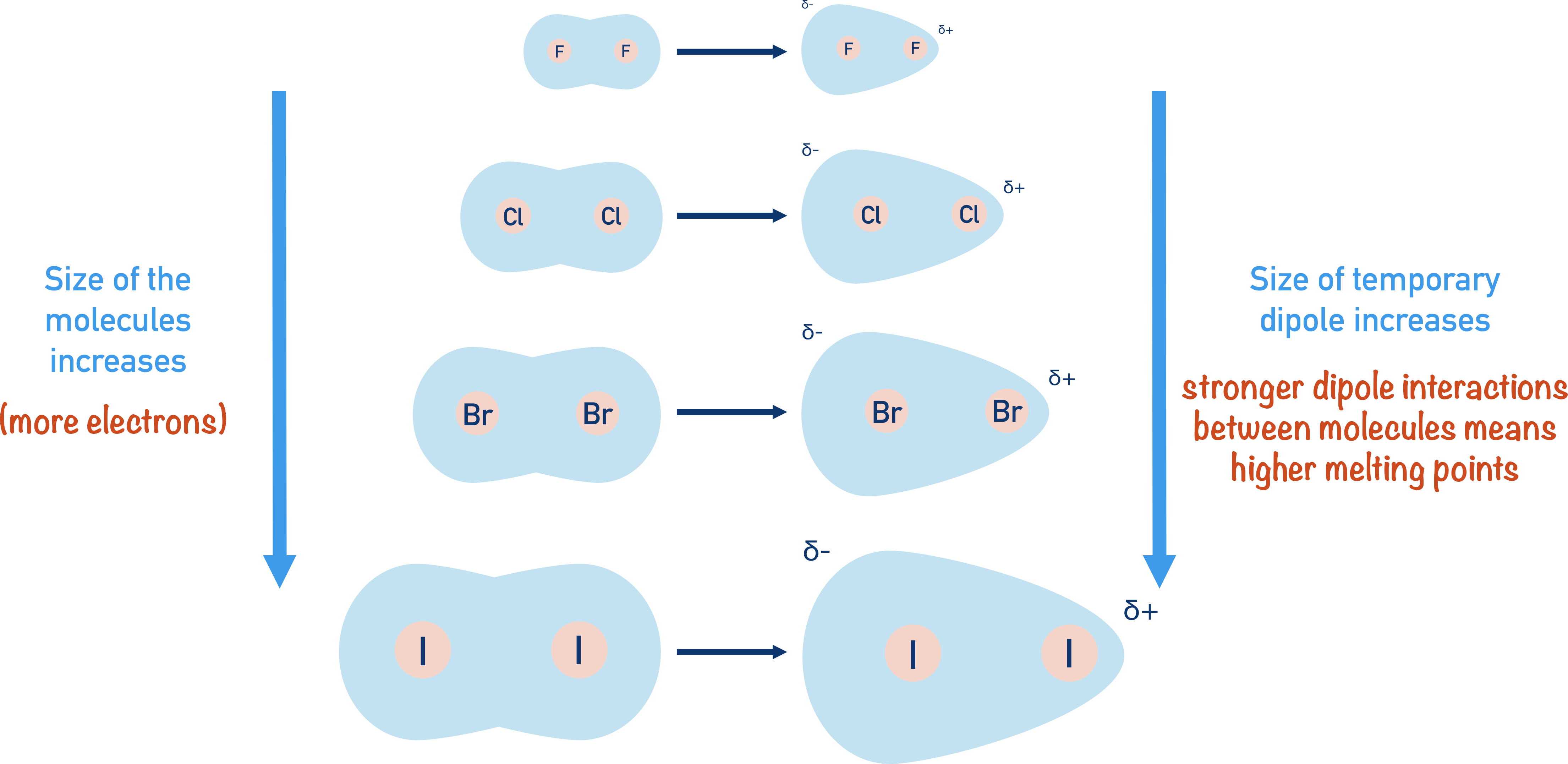
Now, here’s where the "down the group" part comes in. As you descend Group 7, the atoms get bigger. Fluorine is the smallest, a veritable compact car of an atom, while iodine is more like a generously proportioned SUV. This increase in size isn't just about looking more imposing; it means there are more electrons in each atom, and therefore, more electrons in the diatomic molecule.
And here’s the key: more electrons mean a greater potential for those temporary dipoles to form and to be larger. Imagine a tiny pebble versus a large boulder. When the pebble’s electrons shift, the resulting temporary dipole is pretty minor. But with a boulder’s abundance of electrons, the shift can create a more significant and longer-lasting temporary dipole. This, in turn, leads to stronger induced dipoles in neighboring molecules. It’s like the difference between a gentle nudge and a more substantial push.

The Electron Cloud Dance: Polarizability is Key
Chemists have a term for this: polarizability. It's basically how easily the electron cloud of a molecule can be distorted or polarized. Larger molecules with more electrons have more diffuse electron clouds that are much more easily squished and stretched. Think of a stretched rubber band – it’s more easily distorted than a stiff piece of metal. Our larger halogens, like bromine and iodine, have very polarizable electron clouds.
So, as we go from fluorine (F₂) to chlorine (Cl₂) to bromine (Br₂) to iodine (I₂), the size of the molecules increases, the number of electrons increases, and the polarizability of their electron clouds increases. This means the Van der Waals forces between these larger molecules become significantly stronger. They're like those friends who absolutely *need all their comfort items for the trip – the stronger the intermolecular attraction, the more energy it takes to pull them apart.
From Gas to Liquid to Solid: The Boiling Point Journey
What does this mean in practical terms? Well, it directly affects their boiling points. Boiling, at its heart, is the process of giving molecules enough energy to overcome these intermolecular forces and escape into the gaseous phase. If the forces holding the molecules together are weak, it doesn't take much energy (or heat) to break them. If the forces are strong, you need a lot more energy.
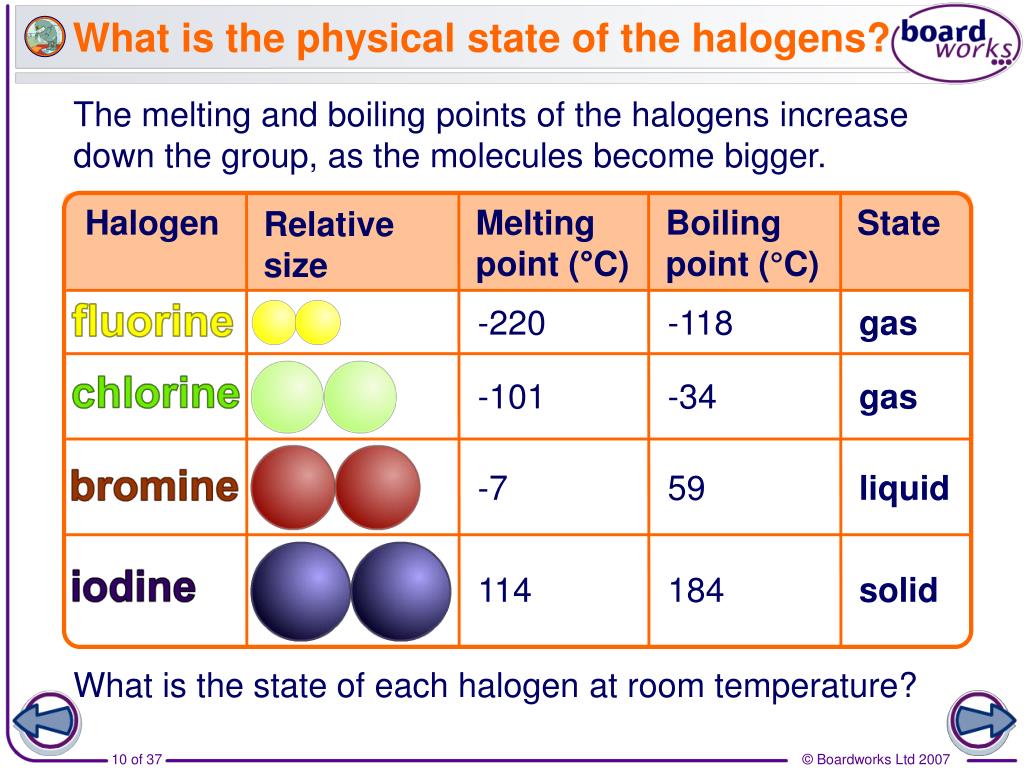
Fluorine and chlorine are at the smaller end of the spectrum. Their Van der Waals forces are relatively weak, so they exist as gases at room temperature. Think of them as the energetic youngsters who are always ready to jump up and run around. They don't need much coaxing to become airborne, so to speak.

Bromine is a bit larger and has stronger intermolecular forces. It’s the middle child, a bit more settled but still prone to bursts of energy. It’s a liquid at room temperature, a bit more grounded than fluorine and chlorine, but still capable of becoming a gas with a bit of warmth.
And then there’s iodine. Iodine molecules are the largest and have the strongest Van der Waals forces. They are the wise elders of the group, requiring significant effort to get them moving. They exist as a solid at room temperature. To get iodine to boil, you need to apply considerably more heat to break those substantial intermolecular attractions.
Fun Facts and Cultural Tidbits
These differences in boiling points aren't just academic; they have real-world implications! For instance, chlorine gas, a diatomic molecule that's a gas at room temperature, is a well-known disinfectant used in swimming pools and water treatment plants. Its gaseous nature makes it easy to disperse and mix. (Though, remember, it's also quite toxic in higher concentrations, so safety first, folks!)
Bromine, being a liquid, is used in various applications, including flame retardants and photographic chemicals. Its liquid state makes it easier to handle and transport in certain industrial processes compared to a gas. Ever seen those old, slightly eerie black-and-white photos? Bromine might have played a small role in their creation.
Iodine, the solid of the group, is essential for human health. Our thyroid glands need it to produce hormones that regulate metabolism. That’s why you’ll find iodine added to table salt – it’s a crucial nutrient that we get thanks to its solid, easily manageable form. A pinch of salt, a dash of health, all thanks to the humble iodine crystal!
And let's not forget fluorine! As the smallest and most reactive halogen, its compounds are used in everything from toothpaste (to strengthen enamel) to non-stick cookware (hello, Teflon!). It’s the tiny powerhouse of the group, always eager to react and make its presence known. Just like that friend who’s always first to try something new.
Practical Tips for Everyday Chemistry Enthusiasts
While we’re not exactly suggesting you start boiling elemental halogens in your kitchen (please, don't!), understanding these principles can make everyday observations more interesting. Next time you’re watching a kettle boil, you’re witnessing water molecules (which have hydrogen bonding, a much stronger force than Van der Waals) gaining enough energy to overcome their attractions and become steam. It's a powerful reminder of the invisible forces at play all around us.
Even simple cooking can be a subtle lesson. Think about how different substances behave when heated. Some melt easily, others require a lot more heat. This is all down to their molecular structures and the strengths of the forces holding them together. It’s like different ingredients having different personalities – some are shy and melt away with a little warmth, others are more robust and need a serious sizzle.
Consider the packaging of different products. Gases are often stored in pressurized containers, liquids in bottles or cans, and solids in bags or boxes. This packaging reflects the physical state of the substance at room temperature, which, as we've seen, is directly linked to its intermolecular forces and, consequently, its boiling point.
A Moment to Reflect: The Quiet Strength in Connection
So, the next time you’re looking at a chart of the periodic table, or even just boiling water for your morning tea, take a moment to appreciate the subtle dance of molecules. The increasing boiling points down Group 7 are a beautiful illustration of how size and electron count influence intermolecular forces. It’s a reminder that even in the seemingly chaotic world of atoms, there’s a predictable elegance, a gentle progression governed by fundamental principles.
It’s a bit like our own lives, isn't it? We grow, we gather experiences (more electrons, if you will), and as we do, our connections with others and the world around us can deepen. Those bonds might not always be obvious, but they shape who we are and how we interact. Just as iodine needs more energy to change its state, perhaps we, too, require a little more effort and understanding to navigate significant shifts in our lives. The universe, from the smallest atom to the grandest star, is full of these quiet, powerful connections, constantly influencing each other in fascinating ways.