Why Do Ionic Compounds Conduct Electricity When Molten

Have you ever marveled at the intricate, shimmering beauty of stained glass windows, the dazzling glow of a neon sign, or even the simple, reliable spark of a car battery? These seemingly disparate wonders all share a common, fascinating principle: the ability of certain substances to conduct electricity, often when they're in a state of flux. Today, let's dive into the world of ionic compounds and discover why they become little electrical highways when they're molten, opening up a universe of creative possibilities and inspiring a deeper understanding of the world around us.
For artists, hobbyists, and the perpetually curious casual learner, understanding this phenomenon isn't just about science; it's about unlocking a new perspective. Imagine the vibrant colors trapped within molten glass, a medium that relies on the precise arrangement and movement of charged particles – our ionic compounds! Or consider the magical transformation of raw materials into conductive pathways for electronics projects, a realm where understanding the fundamentals can lead to groundbreaking innovations. For the DIY enthusiast, it’s about demystifying the magic and empowering yourself to experiment. It allows you to appreciate the engineering behind everyday objects, from the simple heating element in your toaster to the complex circuitry of your smartphone. This knowledge can spark inspiration and fuel your creative endeavors.
Think about the diverse applications. In the realm of art, the ability of molten ionic compounds to conduct electricity is crucial for processes like electrolysis used in metal plating for jewelry or decorative objects. Even within traditional glassblowing, the controlled heating and cooling of silica-based ionic compounds, which have unique conductive properties when hot, contribute to their malleability and the stunning visual effects achievable. In electronics, it's the bedrock of everything from batteries (which use molten salts or electrolytes containing ions) to industrial smelting processes. For hobbyists building custom electronics, understanding how different ionic compounds behave when heated allows for more precise control and safer experimentation with conductivity.
Must Read
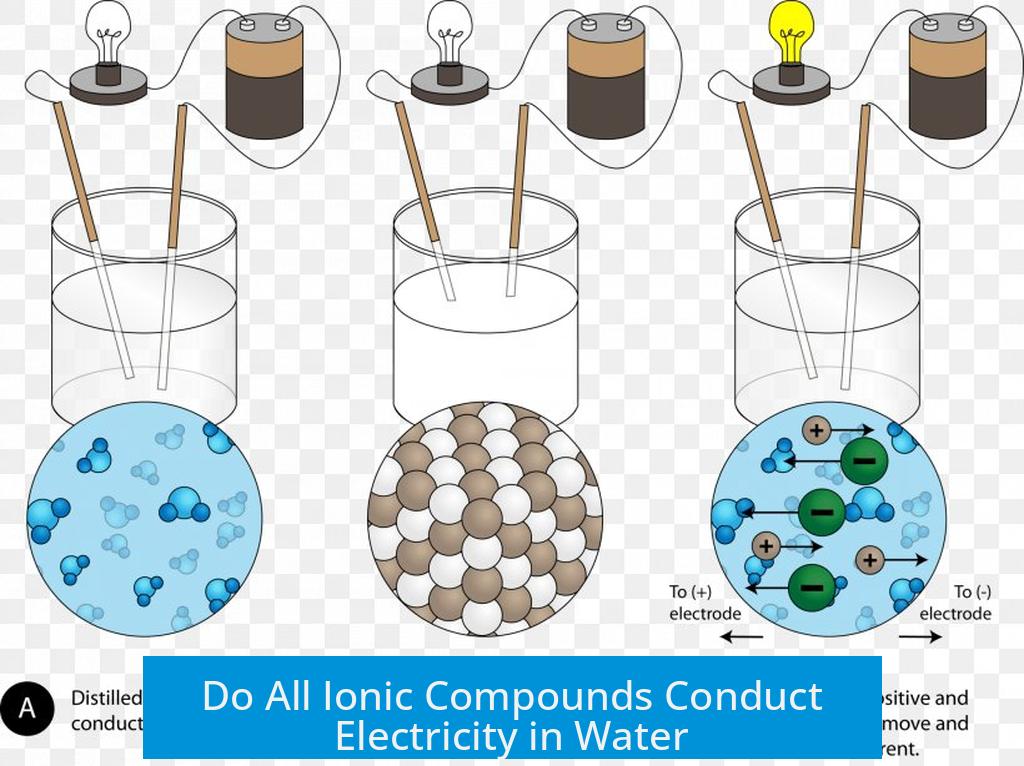
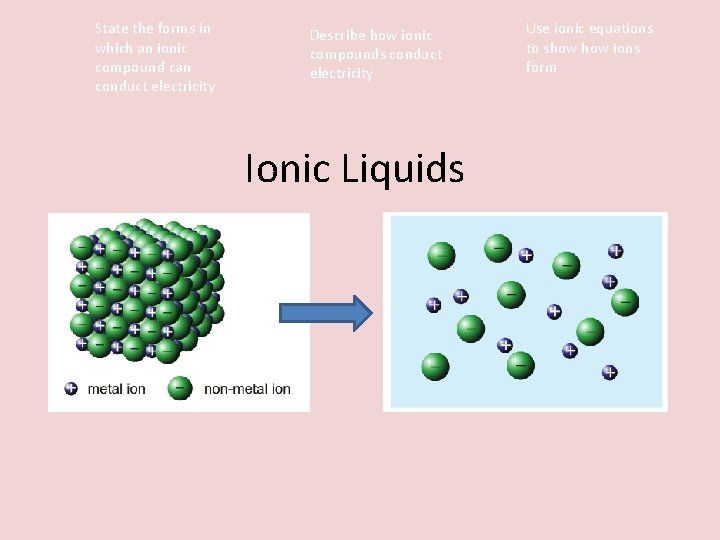
So, why exactly do these compounds conduct electricity when molten? It all comes down to their structure. In their solid state, ionic compounds are like tightly packed cities where charged particles (ions) are held rigidly in place. They’re charged, yes, but they can’t move freely. However, when you heat them up enough to melt them, these ions break free from their fixed positions. Suddenly, they’re like a bustling crowd on a city street, free to move and mingle. When you introduce an electrical current, these mobile, charged ions become the charge carriers. The positively charged ions are attracted to the negative electrode, and the negatively charged ions are attracted to the positive electrode. This synchronized movement of charges is electricity flowing!

Ready to explore this yourself? While working with molten ionic compounds can require specialized equipment and safety precautions, the underlying principles can be explored in safer ways. For instance, you can experiment with dissolving common table salt (sodium chloride, an ionic compound) in water. The water dissolves the crystal lattice, freeing the sodium (Na+) and chloride (Cl-) ions. You’ll find that saltwater conducts electricity much better than pure water. This simple experiment demonstrates the core concept of mobile ions carrying charge, offering a tangible, accessible way to engage with this fascinating scientific principle right at home. Remember, safety first! Always supervise children and use appropriate safety gear for any science experiment.
Ultimately, understanding why molten ionic compounds conduct electricity is more than just memorizing facts. It's about appreciating the dynamic nature of matter and the invisible forces that shape our world. It’s about seeing the science behind the magic, fostering a sense of wonder, and perhaps, sparking your next creative breakthrough. The next time you see a vibrant piece of art or a functioning piece of technology, you'll have a deeper appreciation for the charged dance happening within.