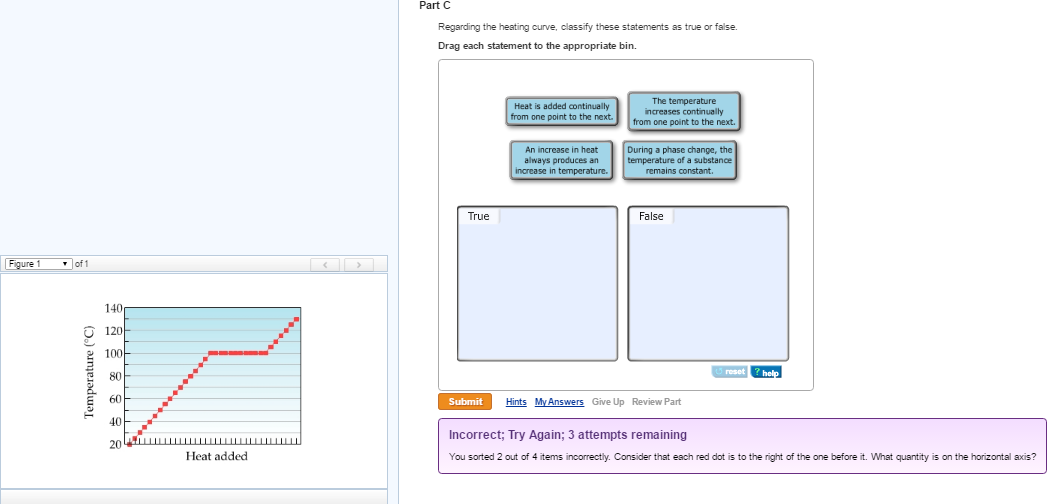
Which Statement Is True For A Heating Curve

Ever stared at a heating curve and felt like you were trying to decipher ancient hieroglyphics? Yeah, me too. It’s one of those sciencey things that pops up and makes you question your life choices. Like, why do we need to know this? But then, sometimes, just sometimes, a little spark of understanding flickers.
So, let’s pretend we’re all sitting around a cozy campfire, sipping hot cocoa. Except, instead of spooky stories, we’re dissecting a heating curve. Because, let’s be honest, is there anything more thrilling than watching a line go up on a graph? Prepare for some groundbreaking revelations. Or at least, a good chuckle.
The Big Mystery: What is a Heating Curve Anyway?
Basically, a heating curve is a graph. It shows what happens to a substance when you give it some heat. Think of it like a temperature tracker for your soup. You crank up the stove, and the temperature climbs, right?
Must Read
Our heating curve does just that, but for all sorts of stuff. From water doing its thing to... well, whatever else scientists like to heat up. It’s a story told in lines and points, with temperature on one axis and time or heat added on the other. Simple enough, right? Until it isn't.
The Plot Twists: Plateaus and Jumps
Now, here’s where things get interesting. A heating curve isn’t just a straight, boring climb. Oh no. It’s got drama! It has plot twists that would make a soap opera jealous.
Sometimes, the temperature just stops going up. It’s like the substance is taking a little nap. This is called a plateau. It’s not stuck; it’s busy doing something else. Something important, I’m sure.
And then, it’s back to business, climbing higher and higher. It’s a roller coaster for your molecules. You get excited for the next big jump, and then bam! Another nap.
The Unpopular Opinion: Plateaus Are the Real Heroes
Here’s my totally unpopular, potentially controversial opinion. While everyone is marveling at the rising temperatures, I’m over here giving a standing ovation to the plateaus. They’re the silent achievers.
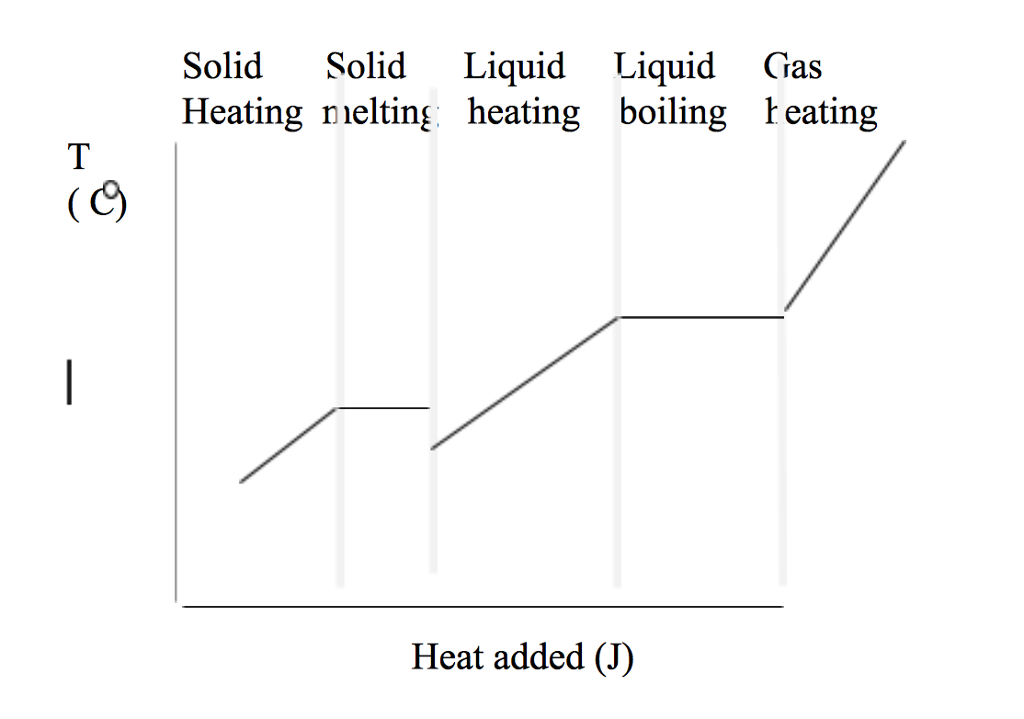
While the temperature is taking a break, the substance is actually undergoing a transformation. It’s like when you’re learning a new skill. You hit a plateau, and you might feel like you’re not progressing. But behind the scenes, your brain is busy rewiring itself. You’re becoming a master.
On a heating curve, these plateaus represent phase changes. Think melting ice or boiling water. The temperature stays the same, but the substance is switching its outfit from solid to liquid, or liquid to gas. It’s a costume change, powered by heat!
Melting: The Cozy Transition
The first plateau you usually see is the melting point. This is when a solid starts to get cozy and turn into a liquid. Imagine your ice cream on a hot day. It’s not getting hotter; it’s just… becoming more ooey-gooey.
The energy you’re adding isn’t raising the temperature anymore. It’s being used to break the bonds holding the solid together. It’s like paying the entrance fee to a cool new club – the liquid club.
Once all the solid has transitioned, then the temperature can start climbing again. It’s a necessary pit stop before the next adventure. And who doesn’t love a good pit stop?

Boiling: The Energetic Leap
Next up, we have the boiling point. This is when a liquid decides it’s had enough and wants to become a gas. It’s a much more dramatic affair than melting.
Think of water vigorously boiling. Those bubbles aren’t just for show; they’re tiny packets of gas trying to escape. The substance is using all that added heat energy to break free from its liquid bonds and zoom around as a gas.
This plateau is where the real party starts. Molecules are bouncing off the walls, and the substance is expanding to take up way more space. It’s the ultimate freedom.
And once again, once all the liquid has become gas, the temperature can resume its ascent. It’s like the applause after a particularly impressive dance move. The performer can finally take a bow and get ready for the next act.
The "Why Bother?" Moments
So, you might be asking, why do we even bother with these curves? Isn’t it enough to just know that heat makes things hotter? Well, science would say no. And who am I to argue with science?

Heating curves tell us specific things about substances. They reveal their melting points and boiling points. These are like fingerprints for different materials. Knowing these points helps us identify substances and understand their properties.
It’s how scientists know that water boils at 100 degrees Celsius. And that something else might boil at a much higher or lower temperature. It’s all about the details, you see.
The "But What If?" Scenarios
What if you’re heating something that’s already a gas? Well, the curve will just keep going up, showing the gas getting hotter. No exciting plateaus there, just a steady climb.
What if you’re cooling something down? Then you get a cooling curve, which is basically a heating curve in reverse. The temperature drops, and you see plateaus where the gas condenses into a liquid, and the liquid freezes into a solid.
It’s like watching a movie backwards. Sometimes it’s less exciting, but you still get the whole story.

My Personal Take: Embrace the Curve
Look, I’m not saying you need to become a heating curve expert overnight. But next time you see one, give it a nod. Appreciate its subtle nuances. Admire the dedication of the plateaus.
Think of it as a tiny, organized adventure. A journey from solid to liquid to gas, all mapped out for your viewing pleasure. It’s a story of transformation, of energy, and of things getting warmer.
And if nothing else, you can impress your friends at your next science-themed party by pointing out a plateau and saying, "Ah, yes. The phase change. Truly the unsung hero of thermodynamics." They might just be slightly bewildered, but hey, at least you'll be entertained.
So, which statement is true for a heating curve? Well, the truest statement, in my humble, slightly quirky opinion, is that it’s way more interesting than it looks. And that, my friends, is a scientific fact. Probably.
The true magic of a heating curve lies not just in the rising temperatures, but in the silent, significant work happening during those plateaus. It’s where the real change, the true transformation, takes place.
Remember, science doesn't always have to be serious. Sometimes, it's just about observing the world around us, finding the humor in the details, and maybe, just maybe, appreciating a good plateau.