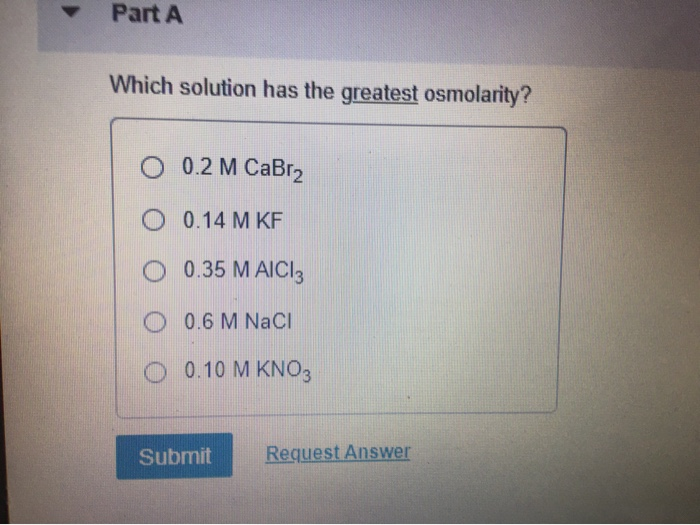
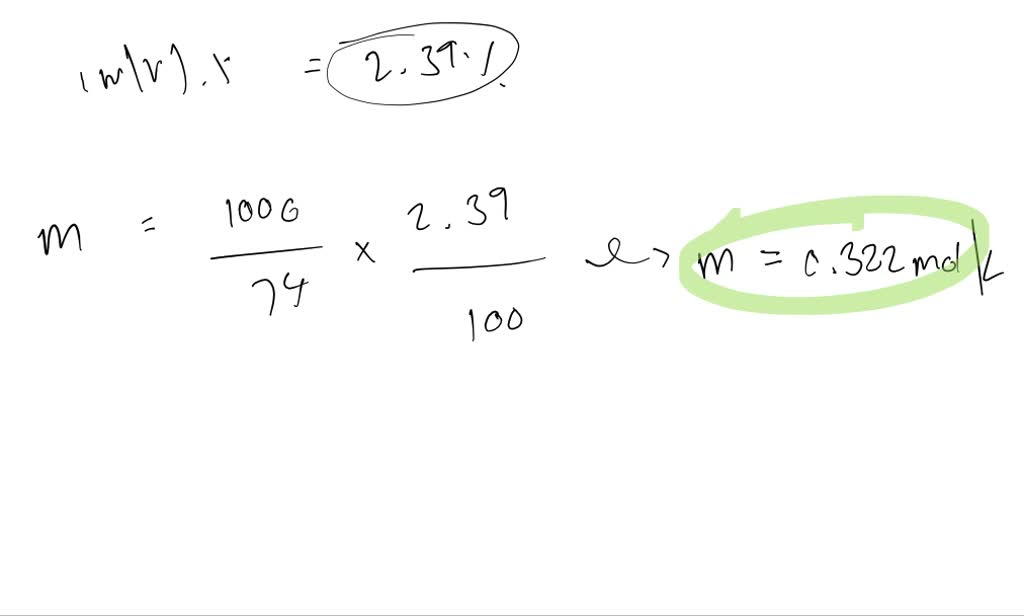Which Solution Has The Greatest Osmolarity
Hey there, science nerds and curious cats! Ever stop to think about what's really going on in all those liquids we chug? Like, beyond just "wet" and "tasty"? Today, we're diving headfirst into the wonderfully weird world of osmolarity. Sounds fancy, right? But don't worry, it's actually pretty cool, and we're going to find out which everyday solution is the king of them all!
So, what IS osmolarity? Think of it like this: it's a measure of how much stuff is dissolved in a liquid. Not just any stuff, mind you. We're talking about tiny particles called solutes. These can be salts, sugars, proteins – basically, anything that's not the water itself. The more of this dissolved "stuff" you have, the higher the osmolarity.
Why should you care? Well, imagine your cells are like little balloons. They have a semi-permeable membrane – a fancy way of saying it lets some things through and keeps others out. Osmolarity is like the boss of water movement across these membranes. Water loves to go where the most "stuff" is, trying to balance things out. It's like water saying, "Whoa, that side's got a lot going on! I'm gonna chill over there!"
Must Read
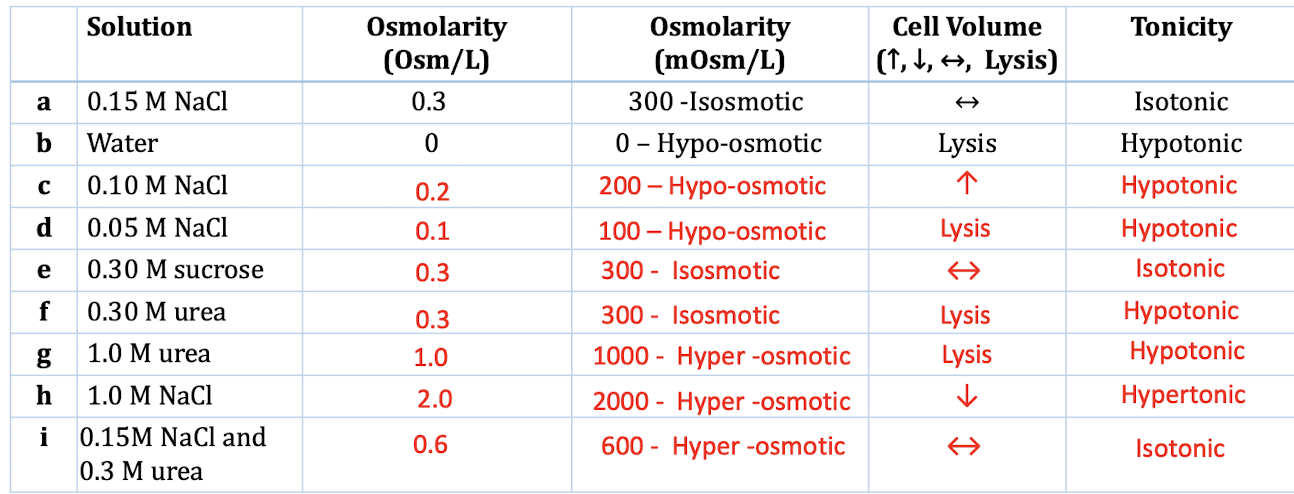
This is super important in our bodies. Think about your blood. It needs to have a specific osmolarity to keep your cells happy and functioning. Too much or too little "stuff" and your cells can either shrivel up like a raisin (dehydration, yikes!) or swell up like a water balloon and potentially burst (lyse, double yikes!).
But we're not just talking about your insides! This concept pops up everywhere. From the sports drinks you gulp after a workout to the saline solution used in hospitals. And that's where the fun begins. We're going to rank some common liquids and see which one packs the biggest osmolarity punch. Get ready for some surprises!
The Contenders: What Liquids Are We Talking About?
Let's round up our potential osmolarity champions. We've got some old favorites and a few that might make you do a double-take. We'll keep it simple, focusing on things you'd likely encounter in your daily life.
First up, the classic: Pure Water. This is our baseline, our zero-point. It's mostly just H2O, so its osmolarity is practically zero. It's the ultimate chill liquid. No dissolved solutes, no drama.
Next, the king of hydration (supposedly): Sports Drink. Think Gatorade or Powerade. These guys have water, sugar, and electrolytes (like sodium and potassium). They're designed to replenish what you lose when you sweat. But how much stuff are they really packing?

Then, we have the everyday hero: Cola (or any sugary soda). Another sugary beverage, but often with more sugar and less electrolytes than a sports drink. Will the extra sugar push it to the top?
Let's not forget the salty side: Broth. Whether it's chicken or beef, broth has water, salt, and some protein bits. Salt is a big player in osmolarity, so this one could be a dark horse.
Now for something a bit more medicinal: Saline Solution. This is what they use to give you IVs. It's basically salt water. But what concentration? This is where things get interesting, because there are different types of saline.
And finally, a couple of real curveballs: Urine. Yep, your own bodily fluid. What's going on in there? And the ultimate, the super-concentrated, the one that makes your eyes water: Tears. Believe it or not, your tears have a job to do, and osmolarity plays a role!
The Osmolarity Showdown: Who's Got the Most Oomph?
Alright, drumroll please! Let's start comparing. Remember, we're looking for the liquid with the most dissolved stuff, the highest osmolarity.

Pure Water, as expected, is at the bottom. It's the tranquil pond in our osmolarity lake. Nothing much is happening here, and that's perfectly fine for diluting things.
Sports Drinks are next. They've got sugars and salts, so they're definitely more concentrated than pure water. This is why they're good for rehydration – they help your body absorb water more efficiently. But are they the most concentrated?
Colas and Sodas are often similar to sports drinks, sometimes even a bit higher due to their sugar content. They're basically sugary water with some fizz and flavoring. They'll give you a sugar rush, but maybe not the most efficient hydration.
Now, let's talk Broth. This is where it gets interesting. Broth, especially a well-seasoned one, can have a surprising amount of dissolved solids, including salt and small protein molecules. This can push its osmolarity up there.
But here’s where things get really scientific (but still fun!).
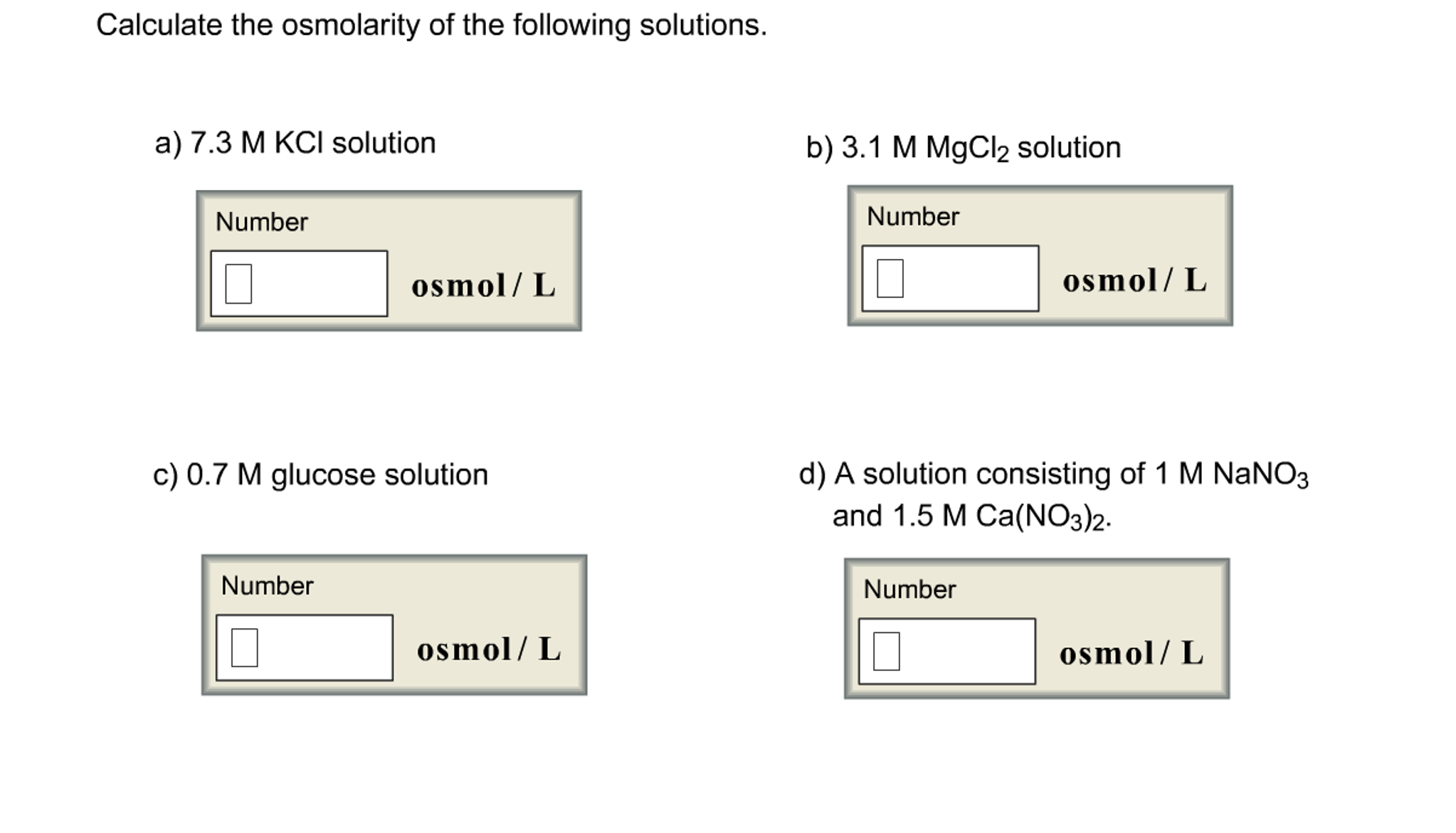
Saline Solution is a fascinating case. There’s isotonic saline (0.9% NaCl). This is designed to be roughly the same osmolarity as your blood. It’s the ultimate gentle solution for your cells. It's designed not to mess with water movement too much.
Then there’s hypertonic saline. This stuff has more salt than your blood. Think 3% or even 7.5% saline. This is the heavy hitter. Because it has so much dissolved salt, water will be pulled out of cells and surrounding tissues. This is why it’s used to reduce swelling in the brain, for example. This is a serious contender for the highest osmolarity among common solutions we'd intentionally put in our bodies.
What about our bodily fluids? Let's get a bit… personal.
Urine can vary wildly in osmolarity. If you're well-hydrated, it's pretty dilute. But if you’re dehydrated, your kidneys are super efficient and try to conserve water. They’ll dump more waste products into the urine, making it much more concentrated. So, on a hot day after a long hike with minimal water? Your urine might surprise you with its osmolarity!
And then… we have Tears. This is where things get a little emotional, literally! Tears aren't just salty water. They contain electrolytes, antibodies, and enzymes. Their osmolarity is actually quite similar to blood, around 300 mOsm/L. This is crucial! If your tears were way more concentrated, they'd suck the water right out of your eyeballs, which would be… well, uncomfortable at best. However, when you cry a lot, the salt concentration can slightly increase.

The Champion Revealed (with a Twist!)
So, who wins the osmolarity crown? When we're talking about common, intentionally consumed or applied solutions, the clear winner for the highest osmolarity is often hypertonic saline. That 7.5% saline solution is packing a serious salty punch. It’s designed to draw water out, and it does so with gusto.
But here’s the quirky part: if we’re talking about a worst-case scenario in terms of concentration of dissolved stuff, it's hard to beat some highly concentrated medical solutions or even extremely dehydrated bodily fluids. For instance, if you were to somehow get some extremely concentrated sugary syrup or very, very concentrated salt solution directly into your system, its osmolarity would be astronomical.
Think about it: a super-duper sugary syrup for pancakes or a thick, syrupy cough medicine. These can have incredibly high sugar concentrations. Or imagine a dried-up salt lick! While we wouldn't want to drink those, they highlight the extreme end of osmolarity.
The fun in osmolarity isn't just about finding the highest number. It's about understanding how these invisible forces affect everything, from your cells to the drinks you choose. It's a little bit of physics, a little bit of biology, and a whole lot of why water moves the way it does.
So next time you grab a drink, or even shed a tear, remember the fascinating world of osmolarity. It's a silent, powerful force that keeps our bodies in balance and makes the world of liquids so much more interesting. Science is everywhere, even in your water bottle!