Which Phrase Best Defines A Covalent Bond
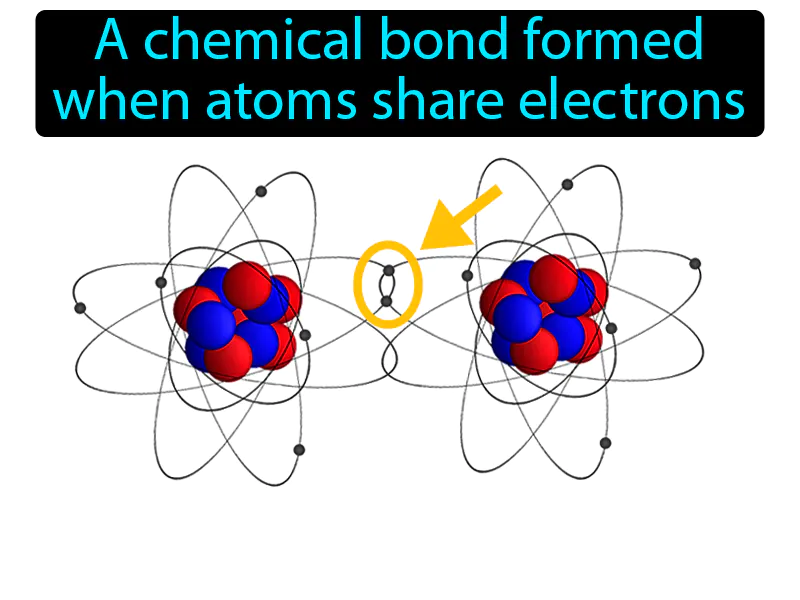
Hey there, science curious folks! Ever found yourself staring at a bunch of letters like H₂O or CO₂ and wondered, "What in the molecular world is going on there?" Well, buckle up, buttercups, because today we're diving headfirst into the dazzling world of covalent bonds! Think of it as the ultimate molecular potluck, where everyone brings something to the table. No, seriously, it’s like atoms are all saying, "Hey, you’ve got an electron? Cool, I’ve got one too! Let’s totally share and be besties forever!"
So, what exactly IS a covalent bond, you ask? Imagine you're playing a game of "keep away" with your favorite toy. Now, imagine two friends who both really want to play with that toy. What do they do? They might end up playing with it together, right? They're sharing, cooperating, and generally making sure neither of them is left out. That’s basically what’s happening at the atomic level with covalent bonds. Atoms are sharing their electrons to become more stable and happy. It's like a little electron love story!
Now, if we had to pick one phrase that perfectly sums up a covalent bond, it's a tough call because there are so many awesome ways to describe it. But if I were forced to choose, and believe me, the pressure is immense (cue dramatic music!), I'd lean towards something that emphasizes the sharing aspect. You know, that whole "we're in this together" vibe.
Must Read
Let's break down some of the contenders, shall we? We've got:
"Sharing is Caring"
This one’s a classic, right? It’s what your kindergarten teacher probably drilled into your head, and for good reason! In the context of covalent bonds, it’s spot on. Atoms that form covalent bonds are literally sharing their electrons. They’re not stealing them, they’re not giving them away completely, they’re sharing. It’s a beautiful display of inter-atomic altruism, or perhaps just good old-fashioned self-preservation. Either way, it works!
Think of it like this: an atom might have a certain number of electrons, but it feels a bit… incomplete. Like a puzzle with a few missing pieces. Other atoms are in the same boat. So, they get together and say, "Hey, if we pool our resources – our electrons – we can both feel more complete and stable!" It’s like two lonely singles deciding to share a cozy apartment. Suddenly, rent is cheaper, and there's always someone to watch Netflix with. Ah, the joys of partnership!
This "sharing is caring" mantra is so fundamental that it’s practically the atomic equivalent of a hug. And who doesn’t love a good hug? Especially when it leads to a more stable and less grumpy atom. Because, let’s be honest, no one likes a grumpy atom. They tend to cause all sorts of chemical chaos.
"Electron Synergy"
Ooh, fancy! "Synergy" means that the combined effect is greater than the sum of the individual effects. In covalent bonding, when atoms share electrons, they achieve a more stable electron configuration. This is often a full outer shell, which makes them much less reactive and, therefore, more content. It’s like a band where all the musicians play together to create amazing music – the music is much better than any single instrument playing alone.
This phrase highlights the outcome of sharing. It’s not just about the act of sharing, but the incredibly beneficial result. It's the reason why molecules stick together and form the building blocks of everything around us. From the air we breathe to the pizza we eat, it’s all thanks to this electron synergy. Pretty mind-blowing, right? It’s like atoms are running a secret, super-efficient cooperation club.
So, while "sharing is caring" is sweet and simple, "electron synergy" is a bit more sophisticated, hinting at the powerful chemical magic that happens. It's the difference between saying "Let's share our toys" and "Let's collaborate to build an awesome LEGO castle that’s way cooler than anything we could build alone." Both are true, but one is a bit more… architectural.
"Electron Partnership"
This one is also a strong contender. It emphasizes that the electrons are not just being casually shared, but are actively partnered between the atoms. It implies a more committed and stable relationship than a fleeting encounter. Think of it as a business partnership, where both parties contribute and benefit mutually.
In a covalent bond, the shared electrons spend their time orbiting both atomic nuclei. They’re not permanently attached to one atom or the other; they’re doing a little dance, a synchronized swimming routine, around both. This continuous sharing creates a strong attraction that holds the atoms together. It’s like a long-term lease on an electron, where both landlord (atom) and tenant (electron) are happy with the arrangement.
This "partnership" idea is really important because it differentiates covalent bonds from other types of chemical interactions. It’s not a one-sided deal. It’s a cooperative venture. Imagine two chefs deciding to combine their best ingredients and techniques to create a Michelin-star meal. That’s an electron partnership at its finest!

What makes this phrase so great is the sense of mutual benefit and stability it conveys. It’s the atoms working together, each getting what they need to achieve a more stable electron configuration. It’s the atomic equivalent of finding your perfect co-pilot for life's grand chemical adventures.
"A Bond of Shared Electrons"
This is perhaps the most direct and literal description. It simply states what’s happening: a bond is formed because electrons are shared. It's like saying "a friendship is built on mutual respect." True, but maybe a little less evocative.
This phrase is like the no-frills, get-to-the-point explanation. It’s undeniably accurate, but it might lack a little of the playful charm we’re aiming for. However, for clarity and precision, it’s hard to beat. It cuts through the fluff and tells you exactly what you need to know.
Think of it as the chemist’s version of a dictionary definition. It’s essential, it’s correct, but it might not make you giggle with delight. Still, a solid contender for its sheer accuracy.
"The Great Electron Exchange Program"
Okay, maybe I made this one up, but I kind of like it! It conjures up images of atoms going abroad to study and pick up new electrons, bringing them back to their home country (nucleus) to improve their electron-domes. It’s a bit whimsical, but it captures the essence of atoms getting what they need from each other.
This phrase injects a bit of fun and adventure into the concept. It’s like atoms are jet-setting around, collecting electrons like souvenirs. While not strictly scientifically accurate in the travel sense, it gets the idea across that electrons are moving between the atoms in a way that benefits both. It’s a more dynamic and lively way to think about it.
It’s the kind of phrase that makes you picture little electron passports and atom-sized luggage. It’s the epitome of a fun and easy-to-read explanation, even if it’s not the most formal. And let’s be honest, who doesn’t love a good exchange program?
But if we really have to pick the absolute best phrase, the one that encapsulates the core of what a covalent bond is in a way that’s both accurate and delightfully understandable, I’m going to have to go with:
"Sharing Electrons to Stick Together"
Why this one? Because it hits all the key points with maximum charm and minimum jargon!
"Sharing Electrons" – This is the absolute heart of the matter. It’s the core mechanic, the atomic equivalent of a handshake or a high-five. It’s what makes a covalent bond covalent. No ambiguity here, just pure, unadulterated sharing.

"to Stick Together" – This explains the purpose and the result. Atoms don’t just share electrons for the sheer joy of it (though who knows, maybe they do!). They share to become more stable, to achieve a more complete outer electron shell, and in doing so, they stick together. This is what forms molecules. It’s the glue that holds the universe together, at least on a molecular level.
It's a phrase that's easy to remember, easy to explain, and gets the scientific concept across without making your brain do a triple somersault. It’s the atomic equivalent of saying, "Let’s hold hands and walk across the street together so we don’t get lost!"
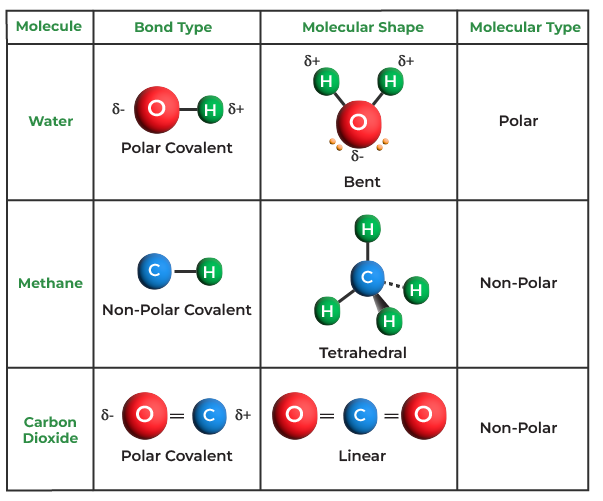
Think about water (H₂O). Those two hydrogen atoms and one oxygen atom aren't just hanging out. They're sharing electrons in a way that makes them all happier and more stable, and that sharing is what allows them to stick together to form that vital molecule we call water. Without that sharing, they'd be off doing their own solitary electron thing, and we'd have a very dry planet, indeed!
It’s a phrase that speaks to cooperation, to mutual benefit, and to the formation of something greater than the sum of its parts. It’s not just about the electrons; it’s about what those shared electrons achieve. They achieve stability, and they achieve connection. They achieve the very essence of what it means to be part of a molecule.
So, the next time you see a chemical formula, or you’re pondering the amazing world of chemistry, remember this simple, elegant phrase. It’s the key to understanding how atoms become friends, how they form bonds, and how they ultimately build the entire universe, one shared electron at a time.
And that, my friends, is pretty darn cool. It's a reminder that even at the tiniest, most fundamental level, cooperation and sharing are what make things work. It’s a beautiful lesson, really. So, go forth, be merry, and share your electrons (metaphorically speaking, of course!) and make the world a more chemically bonded place! You’ve got this!





