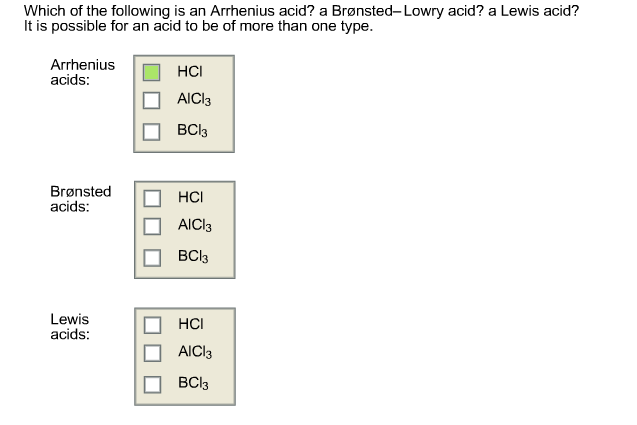
Which Of The Following Is An Arrhenius Acid
Alright, gather 'round, folks, and let's talk about something that sounds a little like it belongs in a dusty textbook, but trust me, it's way more exciting. We're diving headfirst into the wonderful world of Arrhenius acids. Now, I know what you're thinking: "Acids? Isn't that just something that makes my stomach hurt or dissolves my favorite jeans?" Well, yes and also… kinda no. Think of it like this: if chemistry were a superhero league, Arrhenius acids would be the ones who’ve been around the block, the classic heroes with a predictable superpower. And today, we’re going to figure out which of our mysterious candidates is the real deal, the genuine Arrhenius article. It’s like a chemical "Who’s Who?" but with more fizz and less drama. Probably.
So, what exactly is an Arrhenius acid? Imagine a molecule that’s just itching to shed a hydrogen ion, also known as a proton. It’s like that one friend at a party who’s always ready to give away free hugs, but in this case, the hug is a positively charged H+ ion. And where does this ion go? Straight into water, of course! Water, our universal solvent and the queen of everything, is the stage for this whole Arrhenius acid show. If a substance, when dissolved in water, increases the concentration of those little H+ ions, BAM! You’ve got yourself an Arrhenius acid. It’s as simple as that. No complicated algorithms, no secret handshakes, just a good old-fashioned donation of a proton to the water party.
Now, some of you might be wondering, "Are there other kinds of acids?" Oh, you bet your sweet organic chemistry textbook there are! There's the Brønsted-Lowry crew, who are a bit more liberal with their definition of acids (they just need to donate a proton, period, not necessarily to water). And then there are Lewis acids, who are the real wild cards, accepting electron pairs. But for today, we’re sticking with the OG, the Arrhenius. Think of them as the founding fathers of acid theory. They laid the groundwork, and without them, we’d all be swimming in a sea of confusion… and possibly very neutral water.
Must Read
Let’s talk about some characteristics. Arrhenius acids often taste sour. Think of that sharp tang of a lemon or the bite of vinegar. That’s your hint! However, I must strongly advise against tasting random chemicals to identify them. Your taste buds are precious, and while a lemon might be an Arrhenius acid, that unlabeled beaker in the back of the chem lab is probably not your friend. They also tend to be good conductors of electricity when dissolved in water. Why? Because those free-floating H+ ions are like tiny little electrically charged race cars zooming around, making the solution zippy. It’s like adding glitter to a clear liquid; suddenly, everything’s a lot more… sparkly and conductive.
Here's a fun, slightly terrifying fact: the pH scale, that handy little tool that tells us if something is acidic, neutral, or basic, is directly related to the concentration of hydrogen ions. A lower pH means more H+ ions, which means… you guessed it! A stronger Arrhenius acid. So, that super-sour lemon juice? It’s got a low pH because it’s practically overflowing with those H+ giveaway artists.
Now, the million-dollar question: which of the following is an Arrhenius acid? We're about to play a little game of chemical detective. Imagine we have a lineup. On one side, we have potential suspects. On the other, the evidence. We need to see who fits the bill. Remember our criteria: dissolves in water, and releases H+ ions. It’s all about that water interaction. It’s like a dating app for molecules, and water is the matchmaker.
Let’s say our lineup includes a few classic characters. Perhaps we have Hydrochloric Acid (HCl). Now, this guy is a textbook example, a real poster child for Arrhenius acids. When you plop HCl into water, it splits faster than a teenager’s phone screen. Poof! You get an H+ ion and a Cl- ion. The H+ immediately finds a friend in a water molecule, forming something called a hydronium ion (H3O+), which is basically a water molecule giving a piggyback ride to a proton. So, HCl? Definitely an Arrhenius acid. It’s like the celebrity of the Arrhenius world, always making headlines.

What about something like Sodium Hydroxide (NaOH)? This one is the exact opposite. It’s a base! Instead of releasing H+ ions, it’s all about those hydroxide ions (OH-). Think of it as the anti-acid. It’s the villain in our Arrhenius acid story, or at least, the character who plays a very different role. So, NaOH? Nope, not an Arrhenius acid. It’s more of an Arrhenius base, which is a whole other kettle of fish, or rather, a whole other beaker of bubbly solution.
Then we might encounter something like Sugar (C12H22O11). Now, sugar is sweet, it’s yummy, it makes everything taste better. But does it release H+ ions into water? Not really. It dissolves, sure, but it’s more of a passive participant. It’s like that friend who comes to the party but just stands in the corner, not really contributing to the main event. Sugar is not an Arrhenius acid. It’s just… sweet.

And how about Sulfuric Acid (H2SO4)? Oh boy, this one is a heavyweight! Sulfuric acid is incredibly strong, like a bodybuilder in the Arrhenius acid gym. When dissolved in water, it liberates a whole army of H+ ions. It’s a very enthusiastic Arrhenius acid, perhaps a little too enthusiastic for some applications. So, H2SO4? Absolutely, unequivocally, an Arrhenius acid. It practically screams "I'm an acid!"
So, to recap our detective work: we’re looking for a substance that, when tossed into the welcoming arms of water, starts spitting out those precious H+ ions. Hydrochloric acid? Check. Sulfuric acid? Double-check. Sodium hydroxide? Big fat no. Sugar? Nope, too sweet and passive.
The beauty of the Arrhenius definition is its simplicity. It’s about the production of H+ in aqueous solution. That's the golden ticket. So, the next time you're faced with a lineup of chemical suspects and asked to identify an Arrhenius acid, just remember: who’s the biggest proton-giver-outer in the water party? That’s your guy! And trust me, once you get the hang of it, it’s a lot more fun than wrestling with a stubborn jar lid. Almost.