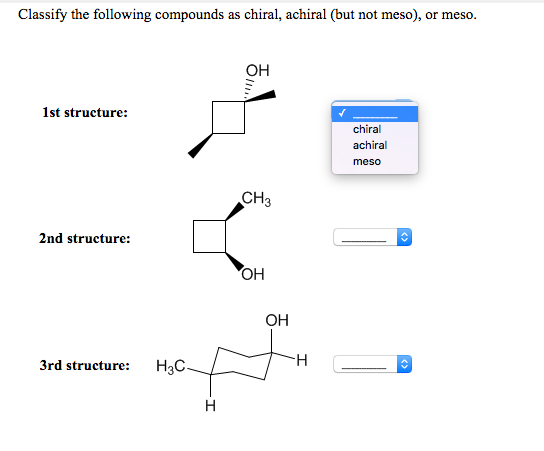
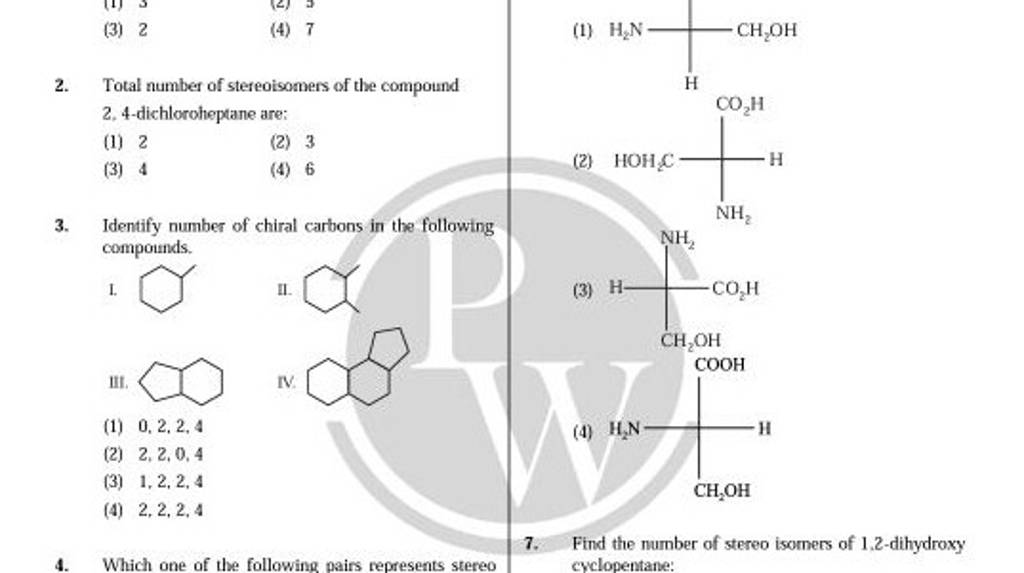
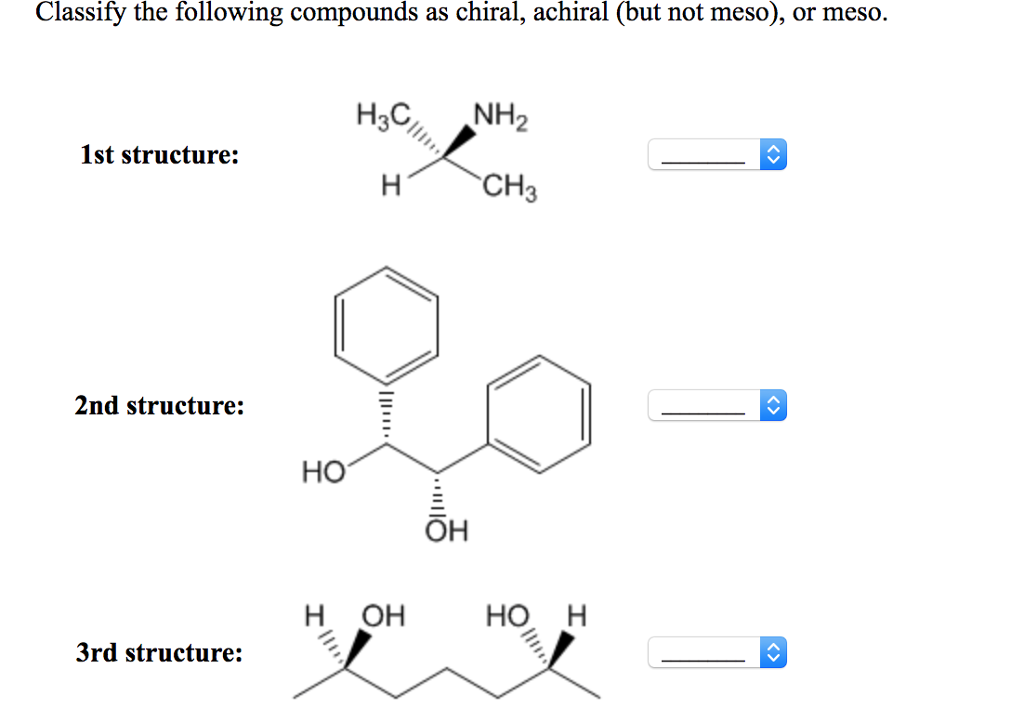
Which Of The Following Compounds Is Chiral
Alright, gather 'round, you magnificent caffeine-fueled thinkers! Today, we're diving headfirst into the wonderfully weird world of chemistry, and specifically, a question that might sound like it was plucked from a particularly baffling episode of "The Big Bang Theory": Which Of The Following Compounds Is Chiral? Now, I know what you're thinking, "Chiral? Is that like, a fancy brand of artisanal cheese?" Close, but no cigar, my friends! Think more like a tiny, molecular dancer. Or a superhero. Or your left and right hand.
See, our universe is absolutely brimming with molecules. They're the building blocks of everything. Your comfy chair? Molecules. That suspiciously delicious doughnut you're eyeing? Molecules. Your uncanny ability to find the last slice of pizza? Yep, molecules doing their thing. And among these molecular maestros, some have this peculiar trait called chirality. It's a word that sounds like it belongs in a Greek tragedy, but it basically means a molecule is non-superimposable on its mirror image. Think of your hands again. They look identical, right? But try as you might, you can't stack your left hand perfectly on top of your right hand so all the fingers and thumbs line up. One's always a bit…off. That's chirality in a nutshell, but on a microscopic, mind-bending scale!
The Mystery of the Mirror Image Twins
So, why should we care about these molecular twins? Well, turns out, in the grand ballroom of life, these chiral molecules are like identical twins with wildly different personalities. They can have the exact same atoms, in the exact same order, but if they're arranged slightly differently in 3D space – bam! – you've got two different molecules with potentially very different jobs. It's like having two identical keys. One opens your front door, the other…well, it might open a portal to Narnia, or it might just get stuck in the lock. You just don't know until you try!
Must Read
This is especially crucial in biology. Our bodies are built on a foundation of chiral molecules. Think enzymes, proteins, even our DNA! They have specific shapes, and they can only interact with other molecules that have complementary shapes. It's like a lock and key situation, but way more intricate and, frankly, more terrifying if you mess it up. For instance, one enantiomer (that's the fancy term for one of the mirror-image twins) of a drug might be a lifesaver, while its chiral counterpart could be a total dud, or worse, a ticking time bomb.
Imagine a drug designed to, say, soothe your nerves. If it's chiral, and you accidentally ingest the wrong enantiomer, instead of a calming lullaby, you might get a disco ball in your brain and the urge to yodel opera. It's molecular sabotage! And it's happened before, folks. Thalidomide, a drug given to pregnant women for morning sickness in the late 1950s and early 1960s, is a stark and tragic example. One enantiomer helped with nausea; the other caused severe birth defects. Talk about a bad trip!
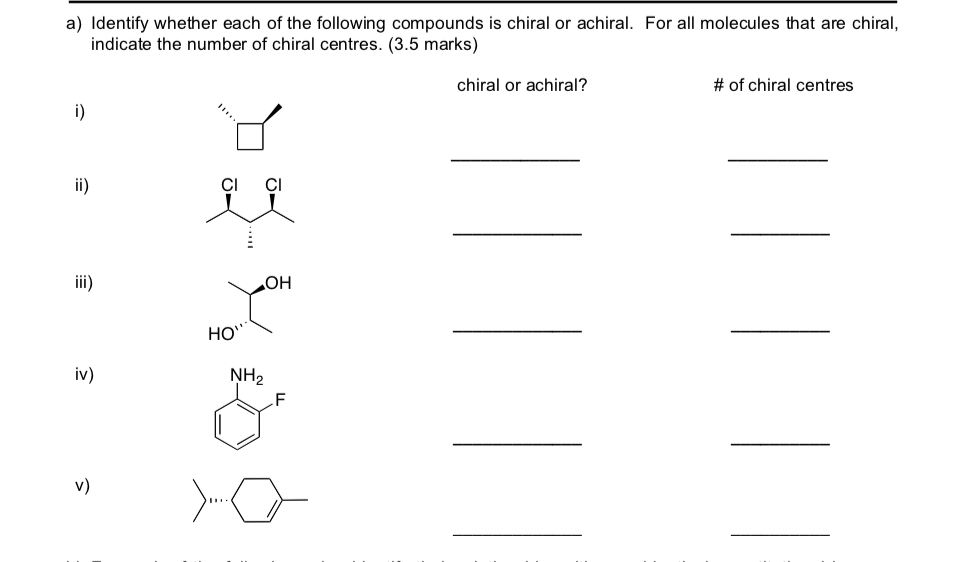
Let's Play "Spot the Chiral"
So, how do we, the humble observers of the molecular world, figure out which compound is our chiral dancer? It's all about looking for a specific feature: a chiral center. Typically, this is a carbon atom that's bonded to four different groups. Think of our carbon atom as the center of a tiny, molecular fidget spinner. If all four prongs of the fidget spinner are different colors, shapes, or textures, then it's likely that spinning fidget spinner is chiral. If two or more of those prongs are the same, then it’s probably not going to have that mirror-image weirdness.
Let's break down some imaginary compounds. Imagine we have Compound A. It's got a carbon atom with a hydrogen, an oxygen, a carbon, and another carbon attached. Hmm, two carbons. Are they different? Let's say one is part of a simple methyl group (CH3) and the other is part of a much larger, more complicated ethyl group (CH2CH3). Aha! Four different things attached to that carbon. Our little carbon friend is likely performing a solo pirouette in the chiral ballet. Compound A is probably chiral!
Now, let's look at Compound B. This one has a carbon atom with two hydrogens, a chlorine, and a bromine attached. Two hydrogens, you say? That's a red flag, my friends! It's like showing up to a masquerade ball wearing the same mask as your date. Not unique enough for chirality. Compound B is likely achiral (the opposite of chiral, meaning it is superimposable on its mirror image, the boring, predictable twin).

What about Compound C? This carbon has a chlorine, a fluorine, a bromine, and a hydrogen. Four completely different atoms. This little carbon atom is a molecular rockstar, juggling four unique partners. Compound C is definitely chiral! It's doing a triple axel with a quadruple twist, and its mirror image is doing the exact same thing, but in reverse. It’s a molecular tango!
Beyond the Basic Carbon
Now, it's not always about carbon atoms, though they are the undisputed champions of chirality. Other atoms like nitrogen, phosphorus, or even sulfur can be chiral centers under certain circumstances. But for our general audience adventure today, focusing on that carbon atom with its four distinct amigos is your golden ticket to identifying chirality. It's like a molecular detective game, and you've just been handed your magnifying glass!

And here's a fun fact to blow your minds: most biological molecules are homochiral. This means they are predominantly made up of only one type of enantiomer. DNA, for instance, is almost exclusively made of "right-handed" sugar molecules. Imagine if your DNA was a jumbled mess of both left and right-handed components – you'd probably be doing the cha-cha instead of walking! Our bodies are incredibly picky about their molecular builders.
So, next time you're pondering the mysteries of the universe, remember the humble chiral molecule. It's the reason why some medicines work and others don't, why your sense of smell can distinguish between pleasant and foul odors (often due to the different chiral forms of scent molecules), and why your left sock never quite matches your right sock. It's the little details, the subtle differences, that make the world go 'round, and in the molecular realm, chirality is the ultimate detail.
So, to recap our cafe chat: when faced with the question, "Which Of The Following Compounds Is Chiral?", you're looking for that carbon atom with four unique attachments. That's your giveaway! It's the molecular equivalent of a handshake, and if all four people shaking hands are different, then you've got yourself a chiral handshake. Keep an eye out for these fascinating, non-superimposable mirror images – they're everywhere, quietly orchestrating the symphony of existence, one chiral spin at a time!