Which Neutral Atom Is Isoelectronic With F+

Ever found yourself wondering about the secret lives of atoms? It might sound a bit niche, but delving into the world of atomic relationships can be surprisingly fun and even a little bit mind-bending. Today, we're going to explore a question that might pique your curiosity: Which neutral atom is isoelectronic with F+? Now, don't let the fancy terms scare you off! This is all about understanding how atoms can share certain fundamental characteristics, like having the same number of electrons. It's like finding out that two seemingly different people have the same favorite song – a little connection that's interesting to discover!
The concept of being isoelectronic is incredibly useful in chemistry and physics. At its core, it means that two different species – atoms or ions – have the same number of electrons. This similarity in electron configuration can lead to some fascinating similarities in their chemical behavior, even if their overall electrical charge is different. Think of it as two siblings who, despite having different personalities, share a remarkable talent for playing the same instrument. Understanding this allows scientists to make predictions about how certain atoms will react or behave under different conditions. It's a cornerstone for grasping the periodic trends and the nuances of chemical bonding.
So, why is this relevant beyond a textbook? Well, this fundamental understanding underpins many scientific endeavors. In education, teaching students about isoelectronic species is a fantastic way to solidify their understanding of atomic structure, electron shells, and the concept of ionization (when an atom gains or loses electrons to become an ion). It helps demystify why certain elements behave the way they do. In a broader sense, understanding isoelectronic relationships is crucial for fields like materials science, where scientists design new materials with specific properties, and in analytical chemistry, where identifying unknown substances often involves understanding their electronic structures. Even in understanding how light interacts with matter, electron configurations play a vital role!
Must Read
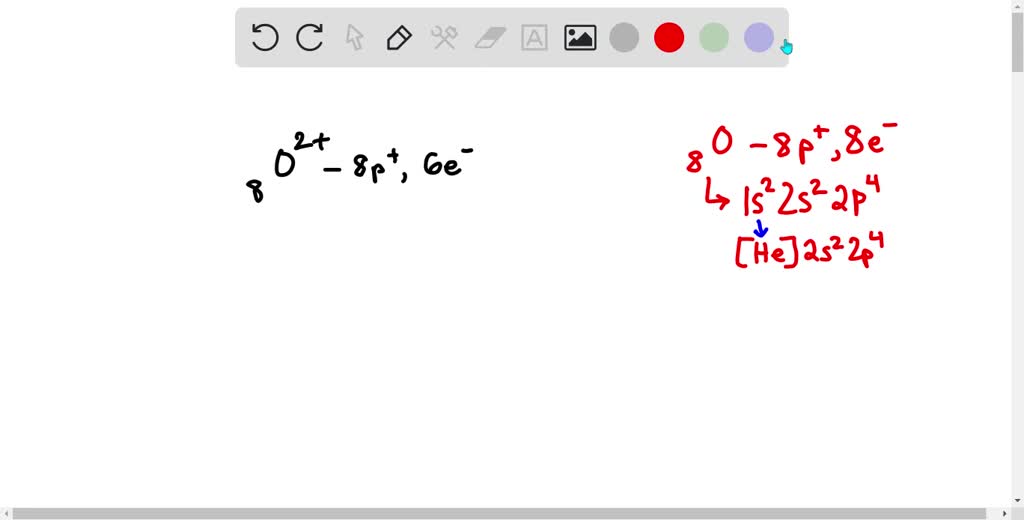
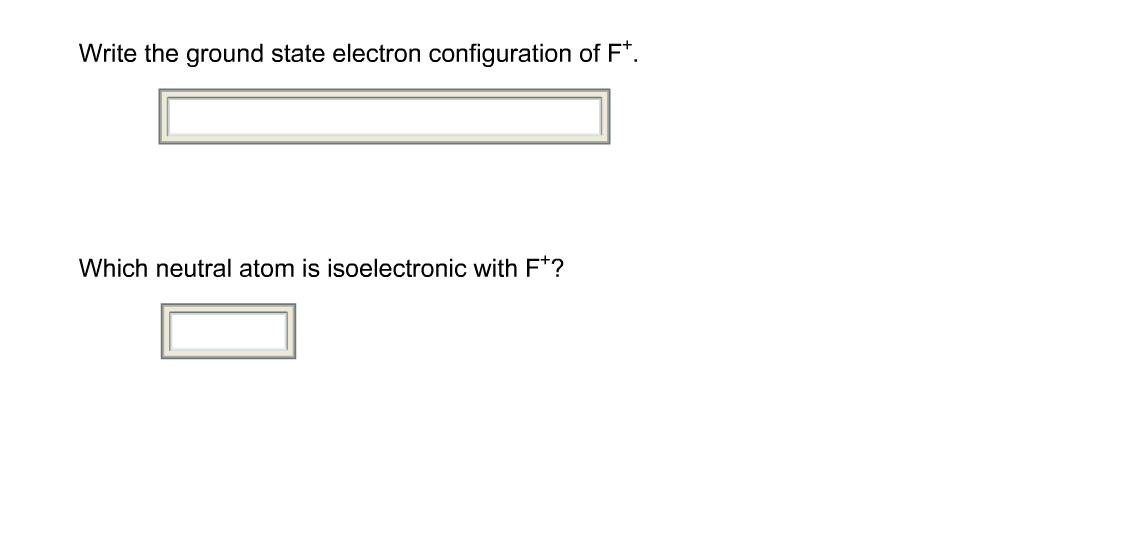
Now, back to our specific question: Which neutral atom is isoelectronic with F+? To figure this out, we first need to know how many electrons F+ has. Fluorine (F) is in the second period and group 17 of the periodic table, meaning a neutral fluorine atom has 9 protons and thus 9 electrons. The '+' sign in F+ indicates that it has lost one electron. So, F+ has 9 - 1 = 8 electrons. Now, we need to find a neutral atom that also has 8 electrons. Looking at the periodic table, the neutral atom with 8 electrons is oxygen (O). Therefore, a neutral oxygen atom is isoelectronic with the fluorine cation (F+).
Exploring this concept further is easier than you might think! The best way to start is by getting a periodic table and a calculator (or just your brain!). For any ion, find the neutral atom and then add or subtract electrons based on its charge. For example, try figuring out which neutral atom is isoelectronic with O2- or Na+. You can also look up lists of isoelectronic species online – it's like a fun scavenger hunt for atomic connections! The more you practice, the more intuitive it becomes, and the more you'll appreciate the elegant order of the atomic world.