Which Molecule Could Have An Expanded Octet

Hey there, curious minds! Ever found yourself staring at a molecule and wondering, "Hmm, what's its deal?" We're going to dive into a little corner of the chemistry world that’s a bit like a party trick for atoms – the idea of an expanded octet. Don't let the fancy name scare you; it's actually pretty neat and, dare I say, even a little bit relevant to why the world around you works the way it does.
So, what's an octet anyway? Think of it like atoms wanting to be super stable, kind of like us wanting a comfy couch after a long day. To achieve this ultimate chill state, most atoms – especially the smaller ones like carbon, nitrogen, and oxygen – like to have eight electrons hanging out in their outermost shell. It's like they’ve got their "perfect number" of friends for a game of cards. Once they hit eight, they’re generally pretty content and don't go looking for more drama.
Imagine your favorite coffee shop. It has a certain number of seats, and when all the seats are filled, it's a buzz of activity, but also kind of at its capacity, right? Atoms with a full octet are similar. They're happy, balanced, and not itching to grab more electrons.
Must Read
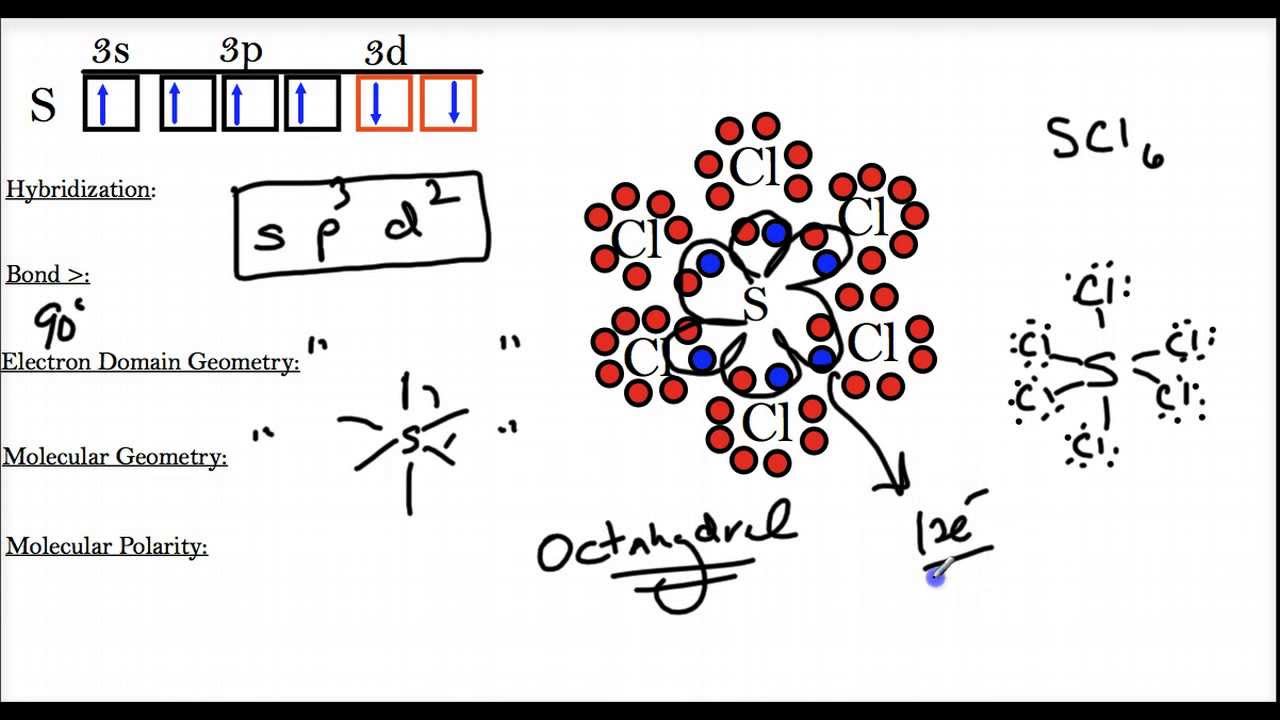
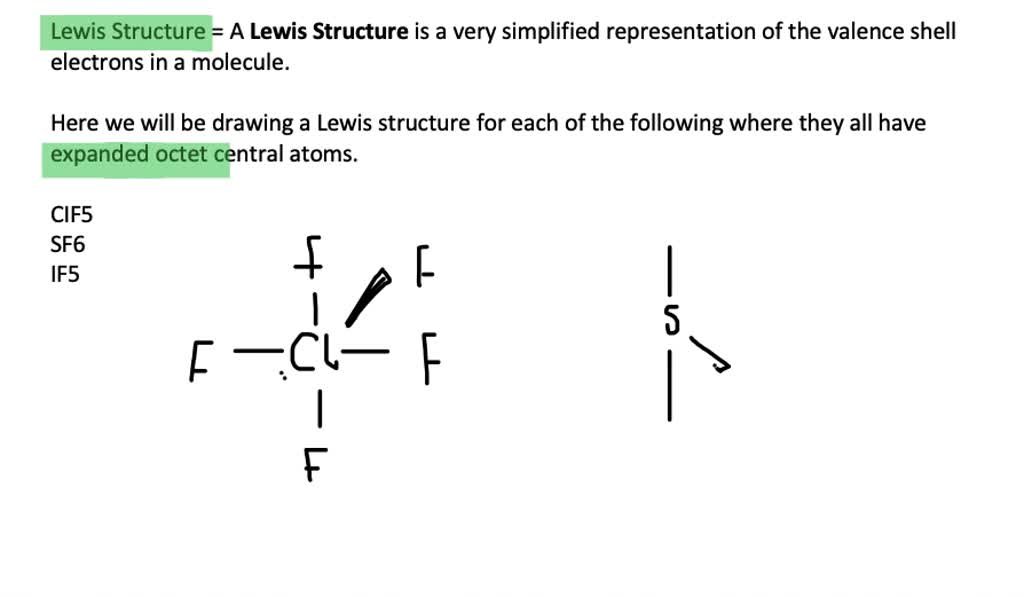
But here's where the fun begins! Some atoms, particularly those in the third row and below on the periodic table (think elements like sulfur, phosphorus, and chlorine), are a bit more… spacious. They have bigger "apartments" to live in, meaning they have more room to add extra electron buddies. These guys can actually go beyond the magic number eight and accommodate more than eight electrons in their outer shell. This is our star of the show: the expanded octet.
Why Should We Care About an Expanded Octet?
Okay, so some molecules have a few extra electrons. Why is this a big deal for us, who are just trying to figure out what to have for dinner? Well, it’s all about understanding how molecules behave. These expanded octets can change the shape of a molecule, its reactivity, and how it interacts with other molecules. And molecules are everywhere!
Think about the air you breathe. Oxygen is pretty standard with its octet. But what about the molecules that make up your delicious food? Or the medicines that help you feel better? Or even the plastics that make up your phone case? All of them have specific structures and properties because of the way their atoms bond and arrange themselves, and sometimes, an expanded octet plays a crucial role in that.
Let’s take a little story. Imagine you’re packing for a trip. You’ve got your suitcase (the atom's electron shell). For most of your clothes, a standard suitcase is perfectly fine. You can fit all your essentials. But what if you’re going on a long expedition and need to pack way more gear? You might need a bigger suitcase, or maybe even a trunk! Elements that can have an expanded octet are like those travelers with the bigger luggage capacity.
Who Are Our Expanded Octet Stars?
The usual suspects for expanding their octet are atoms from the third period (row) and beyond. Why? Because they have access to d orbitals. These are like extra rooms or storage spaces in their electron shells that smaller atoms just don't have.
One of the most common examples is sulfur. You know that distinct smell of eggs or even a skunk? That's often due to sulfur-containing compounds. Take sulfur dioxide (SO2). Sulfur can bond with two oxygen atoms, and in some representations, it's shown with more than eight electrons around it. This flexibility allows sulfur to form different types of bonds and create a variety of molecules with unique properties.
Another familiar face is phosphorus. Think about the phosphorus in your toothpaste that helps keep your teeth strong, or the phosphorus in fertilizers that helps plants grow. Phosphorus can also pull off the expanded octet trick. Phosphorus pentachloride (PCl5) is a classic example. Phosphorus is bonded to five chlorine atoms, and to make that happen, it needs to go beyond the usual eight electrons.
And let’s not forget iodine. You might have encountered iodine in your kitchen as part of iodized salt, which is important for your thyroid health. Iodine can also form compounds where it has an expanded octet, like in the iodide ion (I3-), which is made of three iodine atoms. It’s like a little electron party that keeps going!
Why This Matters (Beyond the Lab Coat)
So, why should you, the everyday reader, give a hoot about expanded octets? Because it's a fundamental concept that helps explain the diversity and complexity of the chemical world.
Imagine you're trying to build with LEGOs. If all your bricks were the same size and shape, you'd have limited options. But if you have a variety of bricks – big, small, with different connectors – you can build so much more! Expanded octets are like having those extra, versatile LEGO bricks in the molecular world. They allow for a wider range of molecular structures and, therefore, a wider range of functions and properties.
Understanding these concepts helps chemists design new materials, develop more effective drugs, and even understand the chemical reactions happening in our own bodies. When a molecule can be more flexible in its electron arrangement, it can participate in different kinds of reactions, leading to all sorts of fascinating outcomes.
It’s also a good reminder that sometimes, the rules we learn aren't always absolute. Just like a talented chef can bend a recipe to create something extraordinary, some atoms can bend the "octet rule" to create incredibly diverse and important molecules. So, the next time you marvel at a vibrant flower, enjoy a tasty meal, or use a piece of technology, remember that hidden within the microscopic world, molecules are doing their thing, and sometimes, that involves a little extra electron power!
It’s a little peek into the amazing, often invisible, world of chemistry that makes our lives possible and so interesting. So, next time you hear about an expanded octet, you can smile and think, "Ah, yes, those electron-packing superstars!"