Which Compound Matches The Ir Spectrum Best
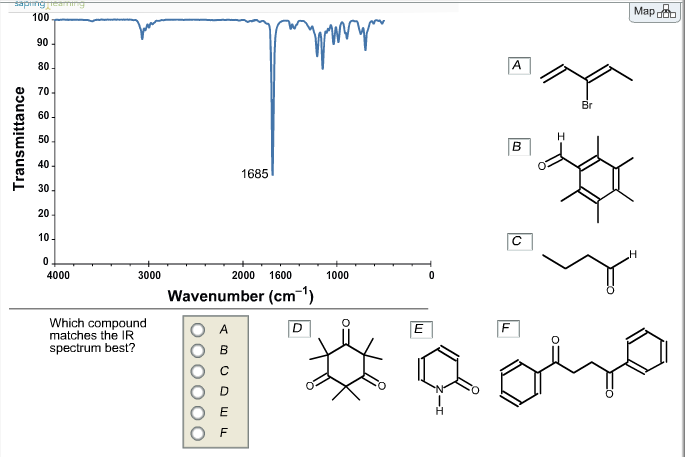
You know, I was staring at this pile of scientific papers the other day, a whole stack of them, and one in particular caught my eye. It had a ridiculously complex chemical structure on the cover, looking like a tiny, spiky hedgehog made of atoms. And next to it? A squiggly line graph that looked suspiciously like a seismograph reading after a particularly dramatic earthquake. My first thought, I'll be honest, was: "Okay, what's this all about?" It felt like trying to solve a mystery where the clues were… well, squiggles. Sound familiar to anyone else who's ever dabbled in the wonderful world of chemistry?
This entire saga, and trust me, it felt like a saga, revolved around a single, albeit crucial, question: which of these potential compounds actually is the one we're after? You see, in science, especially in the lab, sometimes you make stuff. You mix this with that, heat it up, cool it down, add a pinch of catalyst (or a whole heaping spoonful if you're feeling brave), and you hope you've created the molecule you intended. But how do you know? You can't just take its word for it. You need proof. And that's where our friend, the Infrared (IR) spectrum, comes in.
Think of the IR spectrum as a molecule's unique fingerprint. No two molecules, unless they're identical twins (which in chemistry means being the exact same compound, obviously!), have the same fingerprint. It's a graph that tells us about the vibrations happening within the molecule. Yep, molecules are constantly jiggling, stretching, bending, and wiggling like nobody's business. And these movements are specific to the types of bonds and atoms present in the molecule.
Must Read
So, the mystery we were trying to solve was essentially a molecular identification game. We had a synthesized sample, let's call it "Mystery Compound X," and a bunch of reference compounds that we thought it might be. Our job was to compare the IR spectrum of Mystery Compound X with the IR spectra of the reference compounds and see which one was the closest match. It’s like trying to find your friend in a crowded room by comparing their silhouette to a lineup of other people. A bit of a challenge, right?
The Power of the Squiggle
Let's dive a little deeper into what makes these squiggles so darn powerful. Infrared spectroscopy works by shining infrared light onto a sample. Different bonds within the molecule absorb different frequencies of this infrared light. When a bond absorbs a specific frequency, it starts to vibrate more intensely. The instrument then measures which frequencies were absorbed and at what intensity, and voilà! We get our spectrum. It’s like playing a molecular musical instrument and then recording the notes that come out.
Each type of bond has a characteristic absorption range. For instance, a carbon-hydrogen (C-H) bond might absorb in one region, while a carbon-oxygen (C=O) bond (like in a ketone or carboxylic acid) will have a very distinct and strong absorption in a different region. A hydroxyl (O-H) group, the kind you find in alcohols and carboxylic acids, has its own signature wobble. The number of peaks, their positions (wavenumbers, to be precise – think of it as the 'pitch' of the vibration), and their relative strengths all contribute to the overall fingerprint.
This is where things get interesting. Imagine you're expecting to make ethanol, which has an O-H group. If your spectrum doesn't show that characteristic broad O-H stretch, you might be in trouble. Or worse, if you see a very strong peak where you'd expect a C=O bond, and you were definitely not trying to make a ketone, then something has gone hilariously wrong. This is the kind of moment where you start questioning all your life choices leading up to that point. We've all been there, haven't we?
The Case of the Contested Compound
So, back to our specific case. We had synthesized a compound, and based on the reaction we ran, we believed it should be [Let's just call it 'Compound A' for now, to keep things simple, though in reality it was far more complicated and probably had a much scarier name]. We had three other candidate compounds, [Compound B, Compound C, and Compound D], which were byproducts that could have formed or were related starting materials. Our task was to be the molecular detective and figure out which one was our true prize.
We took our synthesized sample and ran its IR spectrum. Then, we pulled up the pre-recorded IR spectra of Compounds B, C, and D from our lab’s spectral database. Now, the real work began: the visual comparison. It sounds simple, but let me tell you, when you’re looking at a bunch of squiggly lines, it can feel like deciphering hieroglyphics. You’re looking for similarities, for matching peaks in roughly the same positions, for comparable relative intensities. It’s a bit like a game of "spot the difference," but with potentially millions of dollars (or at least a good grade) on the line.
Our synthesized sample had a really strong, sharp peak around 1715 cm⁻¹ (wavenumber). This is a classic indicator of a carbonyl group (C=O). Okay, so we know we likely have something with a double-bonded oxygen. Now, let's look at our suspects.
Compound B, our first suspect, was supposed to be a simple alkane. No carbonyls there, thank goodness. Its spectrum was pretty flat and unremarkable, with only weak C-H stretching bands. Definitely not our compound. That was an easy one to rule out. Phew, one down! Time for a virtual high-five?
Compound C, on the other hand, was an alcohol. It had a nice, broad O-H stretch in the 3300 cm⁻¹ region, which is its hallmark. It also had some C-H stretches, but crucially, it lacked that sharp, strong peak at 1715 cm⁻¹. So, Compound C was out too. Another one bites the dust! Our mystery is getting closer to being solved.
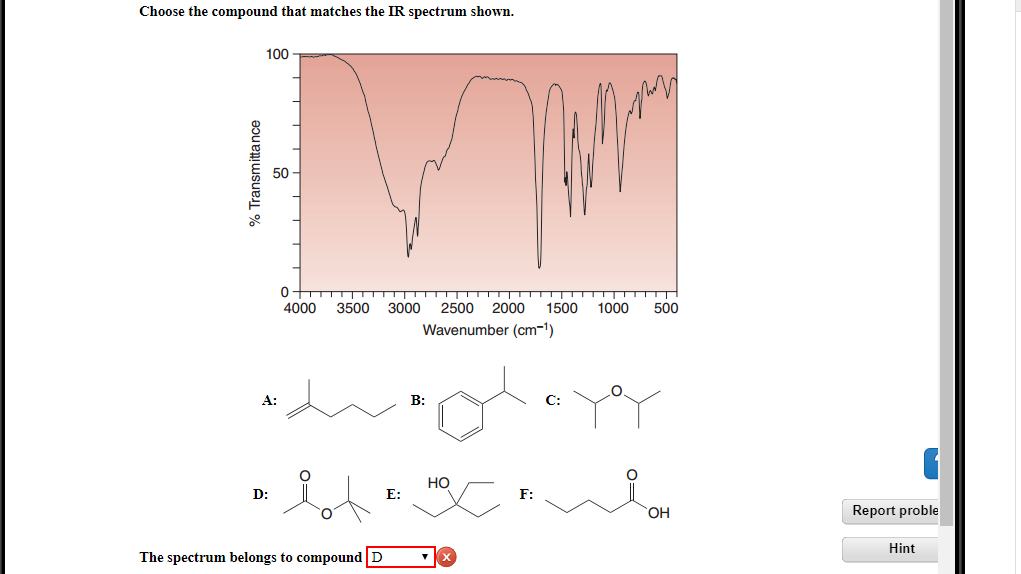
Now we were left with Compound D. Compound D was a carboxylic acid. Carboxylic acids also have a C=O bond, but it’s usually found a little further upfield, around 1700-1725 cm⁻¹, and often accompanied by a very broad O-H stretch that can sometimes overlap with other signals. This was where things got tricky. Our synthesized compound did have that peak around 1715 cm⁻¹. So, potentially it could be Compound D?
However, our synthesized sample had a very clean spectrum. Apart from the C=O stretch, we had a few other distinct peaks, including some sharp C-H stretches in the typical alkyl region (around 2800-3000 cm⁻¹). What we didn't have was that characteristic broad, fuzzy O-H stretch that screams "carboxylic acid." It was absent. Completely.
This is where the power of exclusion in spectroscopy really shines. By not seeing the O-H stretch that should be there if it were Compound D, we could confidently say it wasn't Compound D either. This felt like a detective finding a crucial piece of evidence that disproves the main suspect's alibi. You can almost hear the dramatic music swell, can't you?
When the Spectrum Doesn't Quite Add Up
So, if it wasn't B, C, or D, what was it? This is where the plot thickens, as they say. Sometimes, the spectrum of your synthesized compound doesn't perfectly match any of your reference compounds. This can happen for a few reasons.

Firstly, experimental error. Maybe the instrument wasn't perfectly calibrated. Maybe the sample wasn't pure. Maybe the reference spectra are from a slightly different experimental setup. We always try to minimize these, of course, but it’s a factor.
Secondly, unexpected byproducts. Our intended reaction might have produced a different compound than we thought. This is a common occurrence in organic synthesis. Sometimes, you get a whole cocktail of molecules, and you have to figure out which one is the main event and which are just crashing the party. This is where truly understanding the nuances of IR spectroscopy becomes essential. You have to be able to recognize the fingerprint of what should be there, and also the fingerprints of things that shouldn't be there but might have formed.
Thirdly, and perhaps most excitingly, you might have synthesized a novel compound! This is the dream scenario for many chemists. You've created something completely new, and its IR spectrum is, by definition, going to be different from anything you have in your database. In this situation, you'd compare your spectrum to known functional groups and try to piece together the structure, perhaps using other spectroscopic techniques like NMR (Nuclear Magnetic Resonance) for confirmation.
In our case, the spectrum of our synthesized compound had that strong C=O peak, and clear C-H stretches. It was missing the O-H stretch of the alcohol (Compound C) and the carboxylic acid (Compound D). It also lacked the characteristic signals of the alkane (Compound B).
What was left? We had another synthesized compound in mind, let's call it Compound E. Compound E was a ketone. Ketones have a C=O bond, and typically a strong peak in the 1700-1725 cm⁻¹ region. Crucially, they don't have an O-H group. They do have C-H bonds, which would show up in the fingerprint region, just like we observed.

So, the IR spectrum of our synthesized compound was remarkably similar to the reference spectrum of Compound E. The position and intensity of the C=O stretch were spot on. The C-H stretching and bending vibrations in the fingerprint region also showed excellent correlation. It wasn't a 100% perfect match down to the last tiny wiggle – perhaps due to slight differences in sample preparation or concentration – but it was overwhelmingly the closest. It was the one that made the most sense.
The Verdict is In (and it's a Squiggle!)
When you're trying to match IR spectra, it's not just about finding one or two matching peaks. You're looking at the entire spectrum as a whole. You’re considering the presence or absence of key functional group absorptions. You’re examining the "fingerprint region" (typically below 1500 cm⁻¹), which is packed with complex bending and stretching vibrations that are highly specific to the overall molecular structure.
In our case, the combination of a strong carbonyl peak at the correct position and the absence of an O-H peak, coupled with the overall pattern of other absorptions, strongly pointed towards our synthesized compound being Compound E. It was like putting together a jigsaw puzzle where most of the pieces click perfectly into place. The pieces that didn't quite fit with B, C, and D suddenly made perfect sense when we compared them to E.
So, the compound that matched the IR spectrum best was, in fact, Compound E, the ketone. It means our reaction had proceeded as intended, producing the desired ketone, rather than one of the other potential products. This was a moment of quiet triumph in the lab. No confetti cannons, sadly, but a good cup of tea and the satisfaction of a problem solved. All thanks to the humble, yet incredibly powerful, IR spectrum.
It’s a constant reminder that science, at its core, is about observation, comparison, and deduction. And sometimes, the most complex answers come from interpreting the seemingly simple language of vibrating molecules. Pretty cool, huh? Next time you see a squiggly line graph, remember it’s more than just lines on paper; it’s a story waiting to be told.