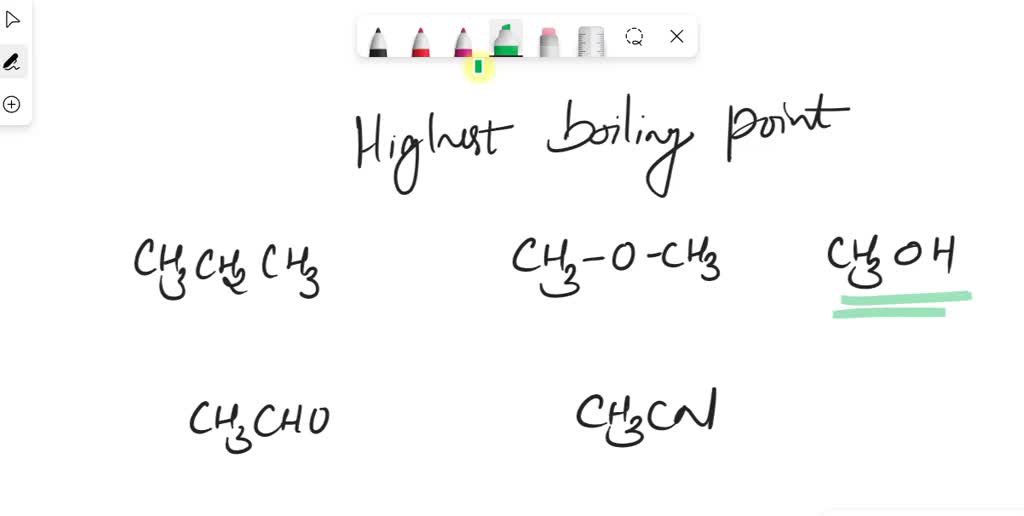
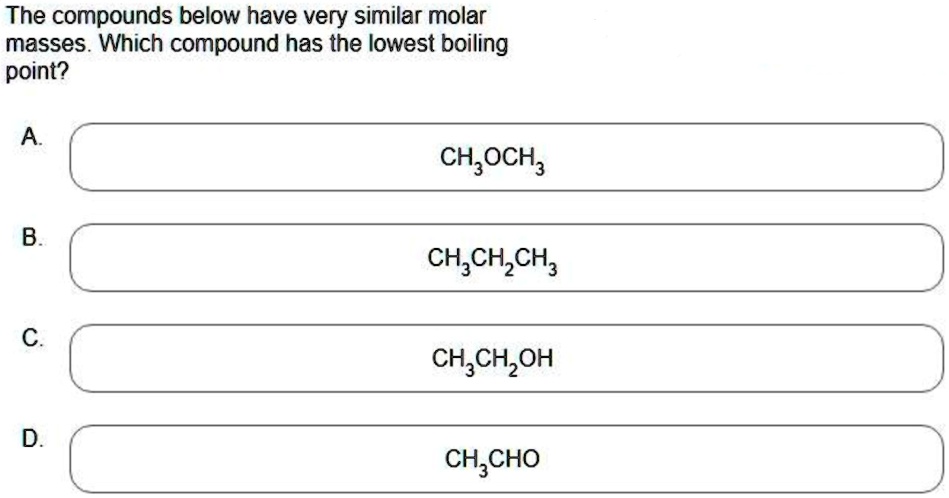
Which Compound Has The Lowest Boiling Point
So, have you ever, like, really thought about boiling points? I mean, beyond just knowing that water boils at 100 degrees Celsius and that’s your cue to chuck in the spaghetti? It sounds a bit nerdy, I know, but trust me, there's a whole universe of hilarious chemical shenanigans going on when things decide to ditch their liquid selves and become a gas. And today, my friends, we’re on a quest to find the ultimate, the absolute, the king of cold boils: the compound with the lowest boiling point!
Imagine a party. A very, very cold party. Most molecules are huddled together, like me at a Christmas party refusing to take off my scarf. They’re clinging on for dear life, nice and cozy in their liquid embrace. But then, there’s our champion. This dude is so anti-clinging, so over the whole ‘being a liquid’ thing, that it’s practically begging to float away at the slightest hint of warmth. It’s like the molecule that always leaves the party early, mumbling about an “early start tomorrow” even though we all know it’s just going to Netflix and chill.
Now, you might be thinking, “Isn’t it just, like, air?” And you’d be surprisingly close! Air is a mixture, and some of its components, like nitrogen and oxygen, are definitely on the chilly side of the boiling spectrum. Nitrogen boils at a frosty -196°C. That’s colder than a polar bear’s picnic! If you were to spill a bit of liquid nitrogen, it wouldn't just get your hands sticky, it would instantly vaporize, looking like a dramatic exit from a superhero movie. But is it the absolute lowest? Oh no, my friends, we're going deeper into the cryogenic abyss!
Must Read
We need to talk about what makes things boil. It’s all about the intermolecular forces. Think of these as the little, invisible hands that molecules use to hold onto each other. The weaker these hands are, the less heat you need to pry them apart and send them off on their gaseous adventures. So, we’re looking for molecules with incredibly weak intermolecular forces. We’re talking about a molecule that’s basically saying, “Nah, I’m good,” to every other molecule within a mile radius.
And the winner, the undisputed champion of not wanting to be liquid, is… drumroll please… Helium!

Yep, that stuff you inhale to make your voice sound like a chipmunk at Christmas is the reigning champ of low boiling points. Specifically, we’re talking about the most common isotope, Helium-4. And its boiling point? A mind-bogglingly frigid -268.9°C. To put that into perspective, that’s just a smidge above absolute zero, which is theoretically the coldest possible temperature, a place where all molecular motion supposedly stops. Helium is so close to not moving at all, it’s practically meditating!
Why is Helium so darn anti-social in the molecular world? It boils down to its electronic structure. Helium atoms are incredibly stable. They've got a full outer electron shell, which is like having the perfect, impenetrable shield. This makes them really bad at forming those attractive forces, the intermolecular forces, with other helium atoms. They’re like the ultimate introverts, preferring their own company and giving any attempt at a group hug a polite but firm “no thanks.”
This means that to even get Helium to consider being a liquid, you have to crank the pressure up and then cool it down to temperatures that would make a penguin shiver. Even at these extreme conditions, it’s a bit… weird. Liquid Helium is famously a superfluid. That means it can flow without any resistance, climbing up the sides of its container and squeezing through microscopic cracks like it’s got somewhere really important to be, despite having zero momentum. It’s the ultimate escape artist of the molecular world.
Imagine trying to pour liquid Helium. It wouldn't just pour; it would ooze, defying gravity and logic. It’s like trying to catch a ghost made of cold – utterly baffling and incredibly cool (pun intended!). Scientists have to use special equipment to even handle it, like a super-duper thermos that’s practically a black hole of insulation. Trying to keep liquid Helium liquid is a constant battle against the warmth of the entire universe, and the universe is a pretty big and warm place.
What about other noble gases, you ask? They’re also pretty chill, but not Helium-level chill. Neon, for instance, boils at a comparatively balmy -246°C. Argon is even warmer at -186°C. They’re like the cousins of Helium, still on the cooler side, but not quite the frosty royalty. They’re the ones who might show up to the party but will definitely be wearing a light jacket, whereas Helium is already halfway out the door in an Arctic parka.
So, why should you care about the boiling point of Helium? Well, besides the fact that it's a fantastic party trick for your chemistry-loving friends, it has some seriously important applications. Those super-strong magnets in MRI machines? They use superconducting coils, and those coils need to be kept incredibly cold. Liquid Helium is the go-to coolant for these things. Without it, your annual check-up might involve a lot more… manual diagnostics.
It's also used in scientific research to study quantum mechanics, those wonderfully bizarre rules that govern the universe at its smallest scales. Because liquid Helium is so close to absolute zero, it allows scientists to observe phenomena that are usually hidden by thermal noise. It’s like trying to hear a whisper in a crowded room – you need to turn down the background chatter. And Helium is the ultimate chatter-reducer.
So, the next time you see a balloon float away, remember the noble gas inside. It’s a tiny, invisible superhero of cold, a champion of low boiling points, and a testament to the fact that even the most seemingly simple elements can have the most mind-blowing properties. It’s a reminder that the universe is full of surprises, and sometimes, the most extraordinary things are the ones that are the hardest to get to stick around.