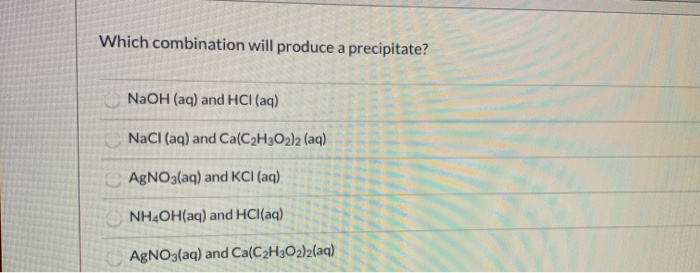
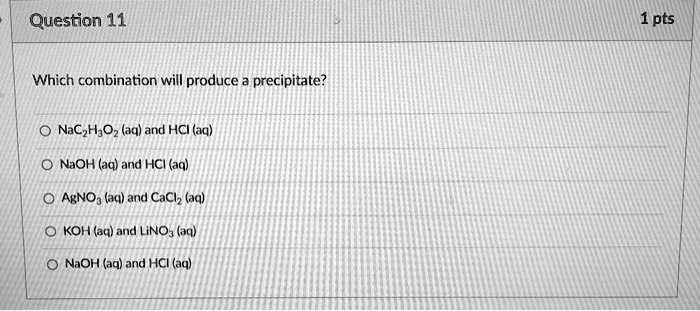
Which Combination Will Produce A Precipitate

Ever found yourself mesmerized by those vibrant, almost magical transformations that happen when two seemingly ordinary liquids meet? We're talking about the captivating world of precipitation reactions, and surprisingly, they're not just confined to sterile laboratory beakers. This fascinating phenomenon has a creative sparkle all its own, inspiring artists, delighting hobbyists, and even offering a touch of wonder for the casually curious. It’s like watching tiny, colorful crystals bloom out of thin air – a secret recipe for visual delight!
The benefits of exploring precipitation are wonderfully diverse. For artists, it's a unique medium for creating unexpected textures and patterns. Imagine a painter using a carefully controlled precipitation to add shimmering, crystalline elements to their work, or a textile artist dyeing fabrics with results that are unpredictable and breathtaking. Hobbyists can enjoy the satisfaction of scientific exploration without the need for complex equipment. Think of it as a fun, hands-on science experiment that yields beautiful results. And for the casual learner, it’s an accessible way to demystify basic chemistry, proving that science can be both educational and aesthetically pleasing. It’s about embracing the unexpected and learning through doing.
The applications and variations are as limitless as imagination. Artists might explore creating "crystal gardens" where intricate structures grow from a solution, or perhaps use precipitation to achieve unique marbled effects on paper or canvas. Think of the subtle beauty of a silver iodide precipitate, often a cloudy white, appearing as if by magic. Or the vibrant blues and greens that can emerge from certain copper sulfate reactions. You might see it used to create iridescent effects in crafts, or even in the creation of special pigments. The subject matter can range from abstract bursts of color to the more controlled growth of crystalline forms, mimicking natural geological processes or completely fantastical creations.
Must Read
Ready to try your hand at some precipitation magic at home? It’s easier than you might think! Start with readily available, safe materials. For instance, mixing a solution of Epsom salts (magnesium sulfate) with a bit of dish soap can create a cloudy, frothy precipitate. Another simple experiment involves dissolving baking soda in water and then adding a bit of vinegar – watch the fizzy reaction, which involves the formation of carbon dioxide gas and a temporary precipitate. Always work in a well-ventilated area and wear old clothes, as some reactions can be a bit messy. Start small, observe carefully, and don't be afraid to experiment with different concentrations and combinations. You might be surprised by what you discover!
Ultimately, the joy of precipitation lies in its inherent element of surprise and discovery. It’s a tangible representation of chemical forces at play, transforming the invisible into the visible. There's a profound satisfaction in orchestrating these reactions and witnessing the beautiful, often unexpected, outcomes. It's a reminder that even in the most fundamental of natural processes, there's an incredible capacity for wonder and artistry. So, go ahead, mix a little science with a lot of imagination – you might just create something truly extraordinary!