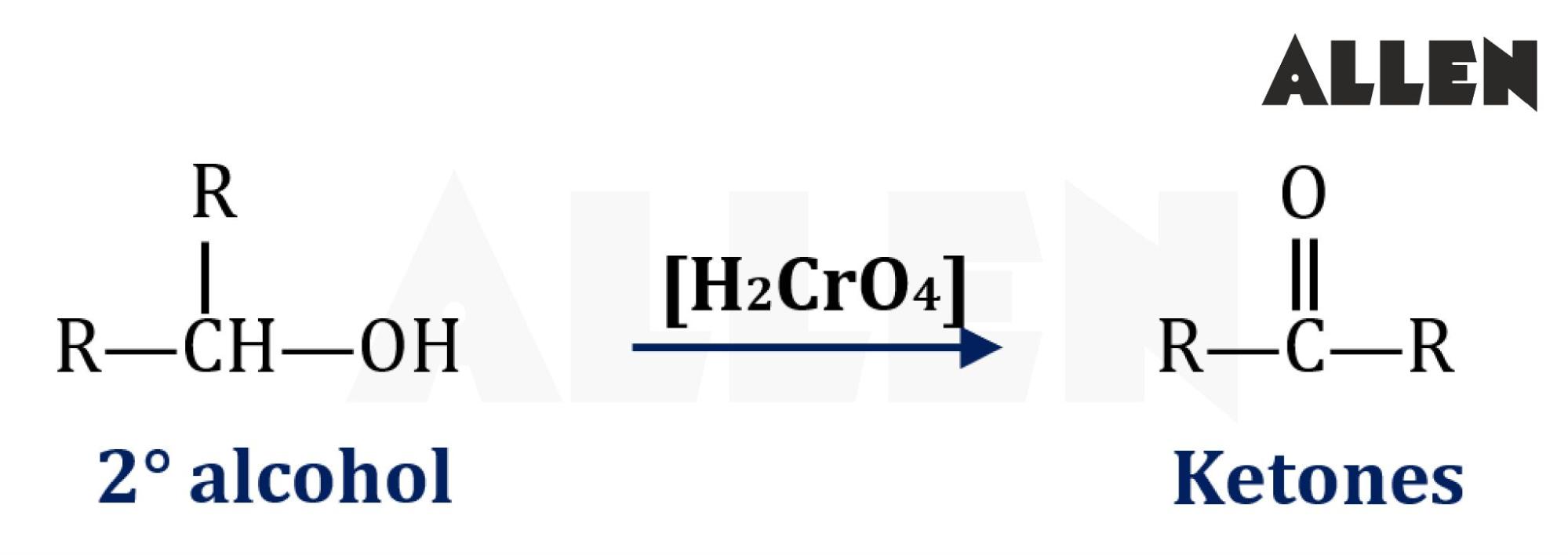Which Action Could Produce A Carbonyl Group
Ever wondered what makes some of your favorite scents, like vanilla or almond, so delightful? Or perhaps you've seen "carbonyl" pop up in cooking shows or science articles and felt a spark of curiosity? Well, you're in for a treat! Understanding how to create a carbonyl group is like unlocking a little secret to the world of flavors, fragrances, and even some everyday materials. It's a concept that's surprisingly accessible and genuinely fun to explore, making it a popular topic for anyone curious about how the world around them works.
For beginners in chemistry or even just curious minds, learning about carbonyl groups is a fantastic starting point. It’s a fundamental building block in organic chemistry, and understanding it can demystify many chemical reactions. For families, it can be a way to engage children in the science behind cooking or natural scents. Imagine explaining why a caramelized onion smells so good – it's all thanks to the magic of carbonyls! And for hobbyists, whether you're into making your own soaps, candles, or even experimenting with baking, knowing about carbonyl groups can open up a whole new world of creative possibilities. You might even be producing them without realizing it!
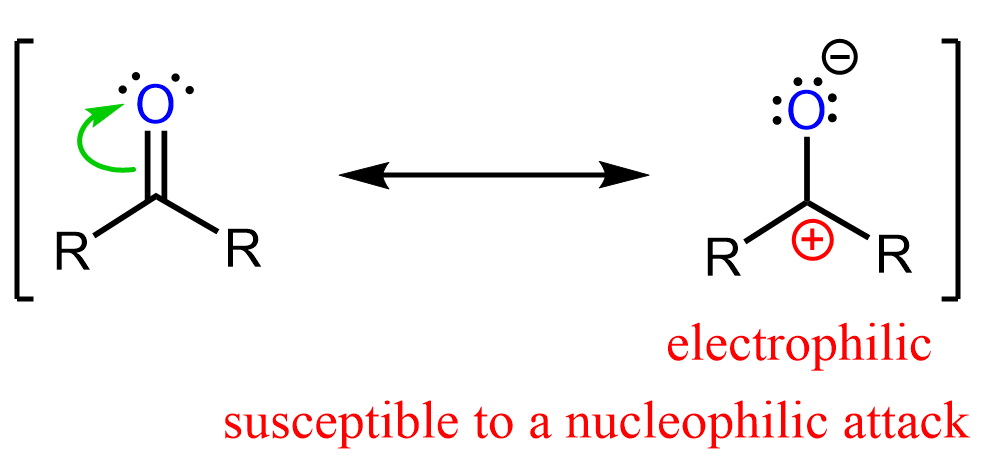
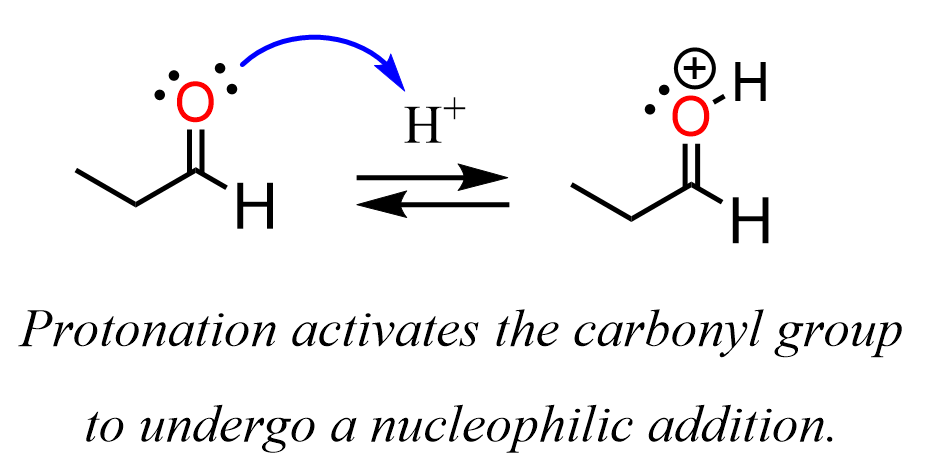
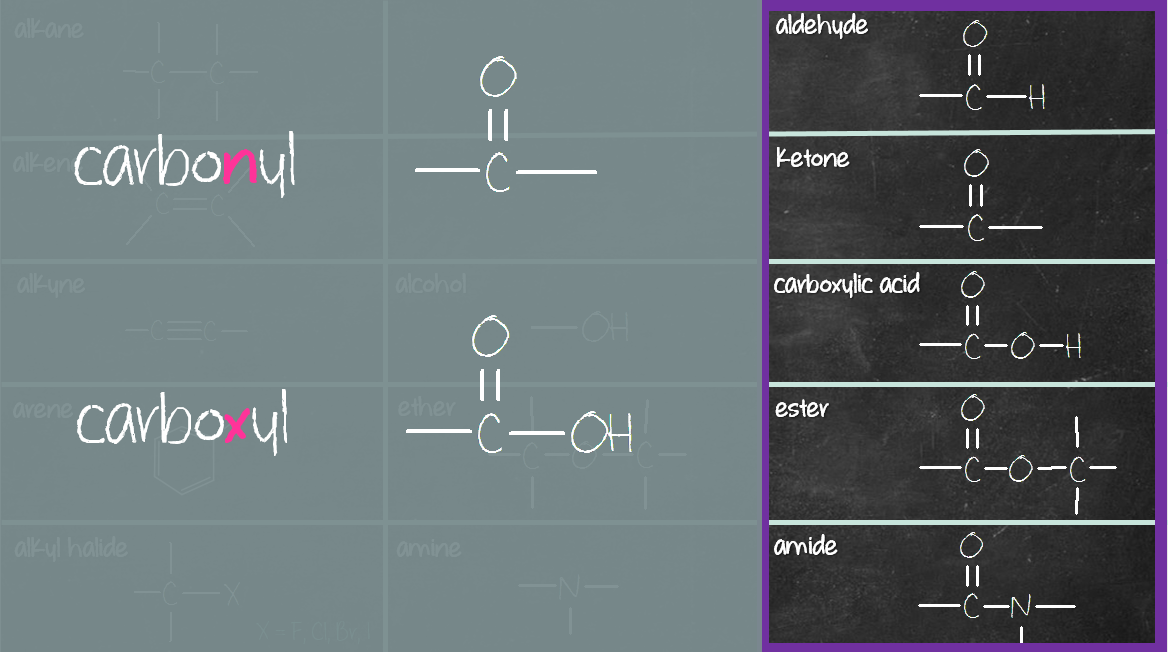
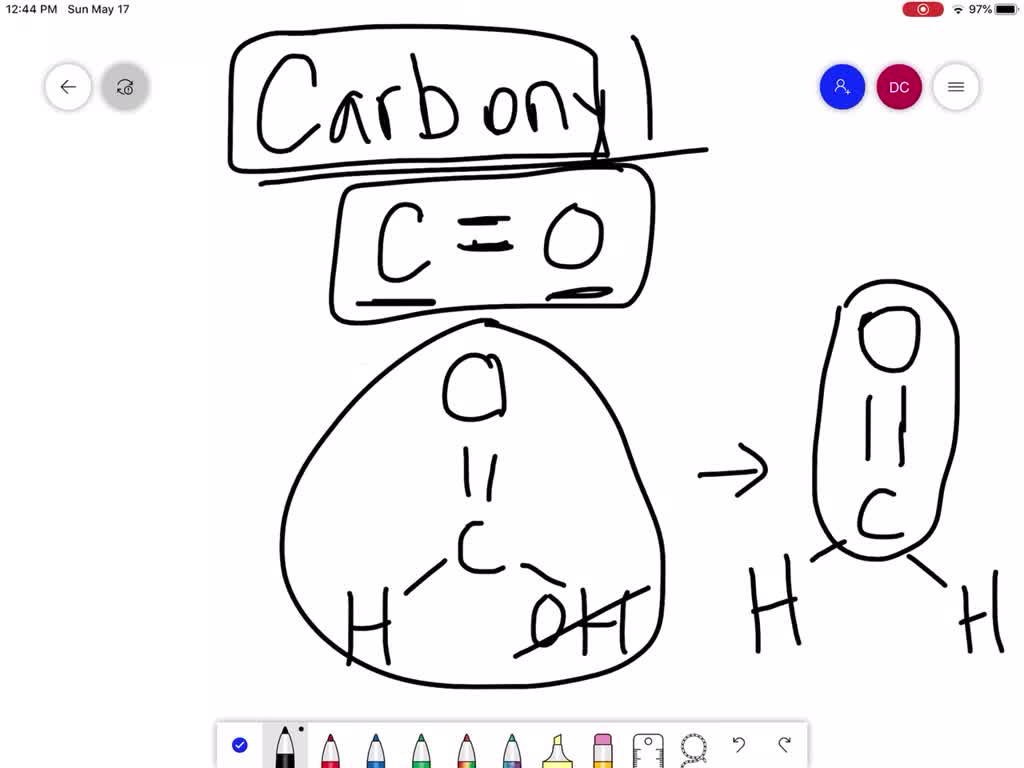
So, what exactly is a carbonyl group? At its core, it's a functional group in organic chemistry that consists of a carbon atom double-bonded to an oxygen atom (C=O). Think of it as a little power unit that influences the properties of a molecule. There are a few common ways to produce this versatile group. One of the most straightforward ways is through oxidation. For instance, when you take a simple alcohol, like ethanol (the kind found in beverages, but also in cleaning supplies), and carefully oxidize it, you can create a carbonyl group. A common example is the oxidation of a primary alcohol to form an aldehyde, which has the carbonyl group at the end of a carbon chain. Or, you can oxidize a secondary alcohol to create a ketone, where the carbonyl group is in the middle of a carbon chain.
Must Read
Another way is through reactions involving carboxylic acids. These molecules already contain a carbonyl group. You can then modify them through various reactions, like esterification, to create new compounds that still feature that essential C=O bond. Think about the sweet, fruity smell of an ester – that aroma is often attributed to the carbonyl group's influence. Variations abound! Depending on what you start with and the conditions you use, you can produce aldehydes, ketones, carboxylic acids, and esters, all of which are characterized by the presence of a carbonyl group.

Getting started with understanding this is easier than you might think. You don't need a full laboratory! Start by looking at common household items. Read ingredient lists for perfumes, flavorings, or even cleaning products. See if you can spot names that hint at these types of molecules. If you're feeling more adventurous, simple home experiments can illustrate these concepts. For instance, the process of making vinegar from apple cider involves the oxidation of alcohol – you're already seeing a carbonyl in action! Many introductory organic chemistry books or even online educational videos offer clear explanations and visual aids that make the concept of carbonyl formation incredibly engaging.
Exploring the creation of a carbonyl group is a journey into the heart of organic chemistry, revealing the molecular basis for many of the sensory experiences we cherish. It’s a concept that’s both educational and empowering, offering a deeper appreciation for the world of chemistry that surrounds us every day. So, dive in, be curious, and discover the fun of carbonyls!

+is+a+functional+group+consisting+of+a+carbon+double+bonded+to+an+oxygen.+This+bond+is+also+found+in+other+functional+groups%2C+such+as+carboxyl+group%2C+which+is+really+just+a+car…)