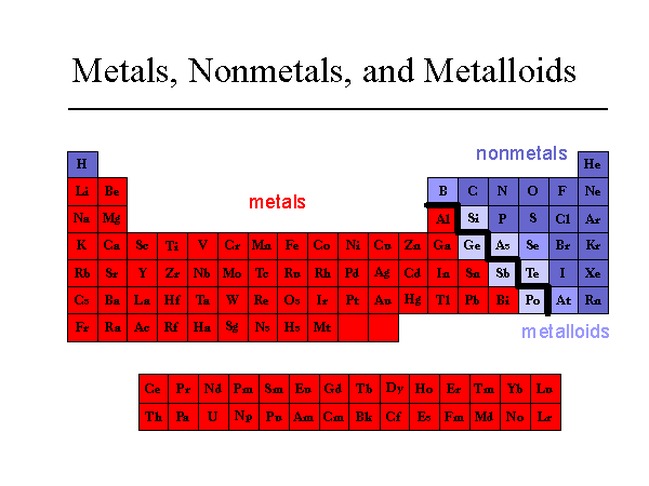
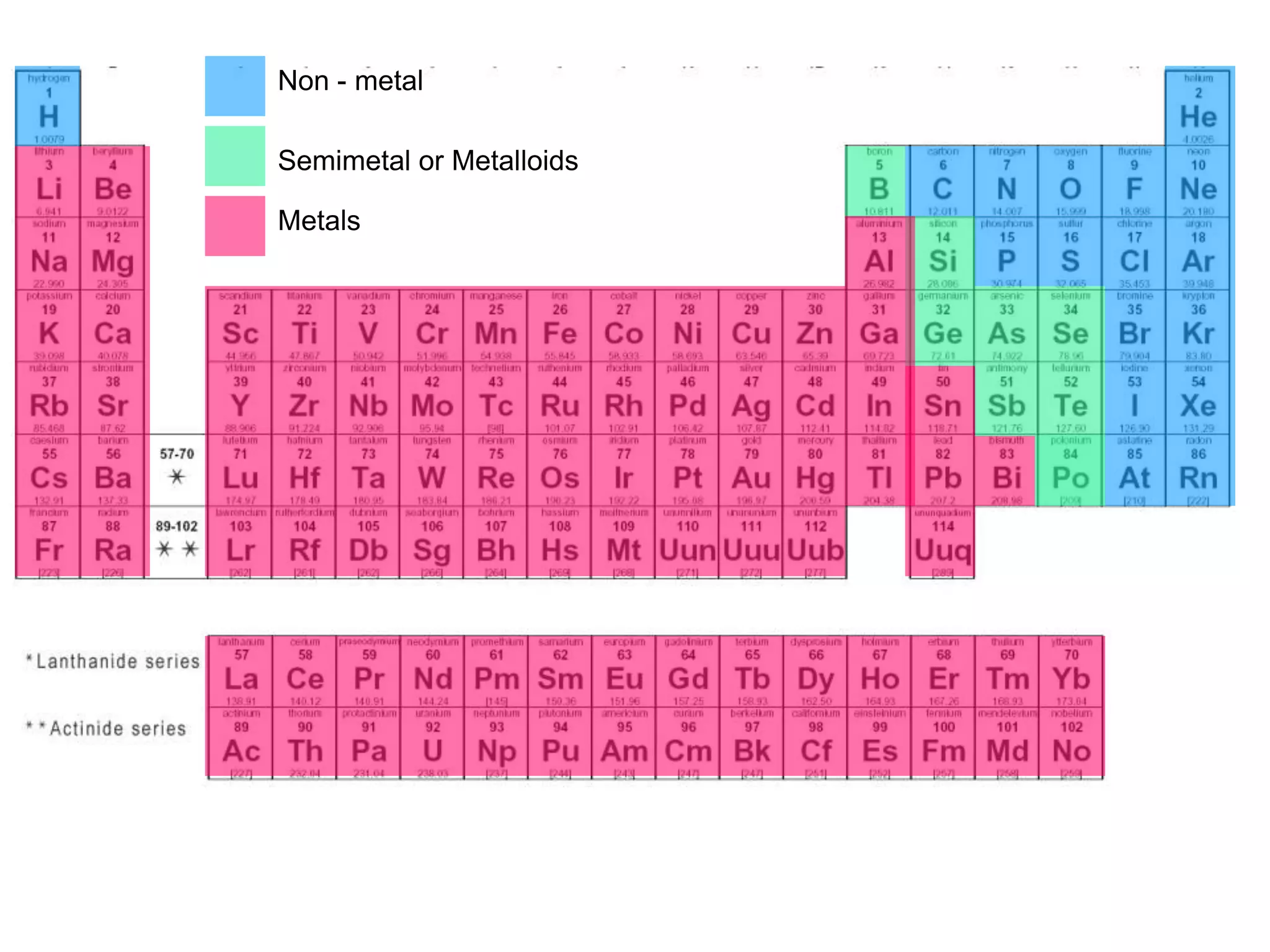
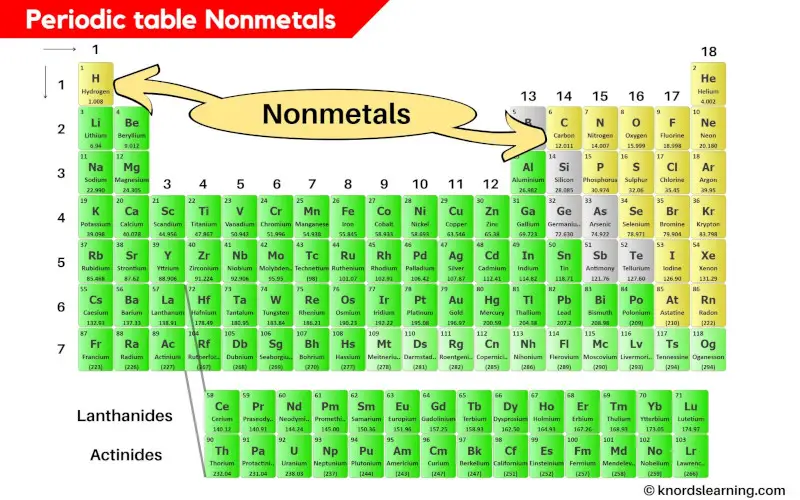
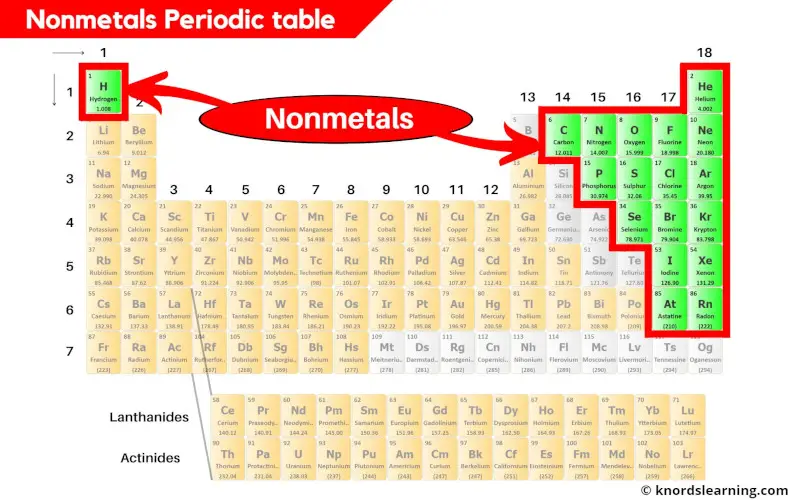
Where Are The Most Active Nonmetals Located

Hey there, ever stopped to think about the invisible stuff that makes up our world? You know, the stuff that’s not quite metal, but plays a starring role in everything from the air we breathe to the way our bodies work? Yep, we're talking about the nonmetals! And today, we’re going on a little adventure to find out where these energetic little guys like to hang out.
Now, when I say "active," I don't mean they're running marathons or joining a choir (though some might secretly love karaoke!). In the world of chemistry, "active" means they're super eager to mingle and form bonds with other elements. They're the life of the party, always looking for a dance partner.
The Usual Suspects: Where the Nonmetals Shine
So, where do these social butterflies of the periodic table tend to gather? Well, they're actually all around us, often in places you might not expect. Think of them as the quiet achievers of the universe.
Must Read
Let's start with a biggie: oxygen. This is probably our most famous nonmetal friend. Without it, our morning coffee would be… well, just hot water, and forget about breathing! Oxygen is in the air we share, making up about 21% of it. It’s also a key ingredient in water (H₂O – that’s two hydrogen atoms for every one oxygen atom, another nonmetal duo!), which is pretty handy for, you know, living.
Imagine you’re out for a brisk walk, breathing deeply. That refreshing feeling? That's mostly thanks to our friend oxygen, diligently doing its job. It’s like the friendly neighbor who always leaves their porch light on, making the whole street feel a little brighter and more welcoming.
In the Air We Breathe
Most of the oxygen we encounter is floating freely in the atmosphere. It's not really "located" in one specific spot, but rather dispersed everywhere we can take a breath. It’s like the invisible blanket that covers our planet, keeping us all warm and well-oxygenated. Pretty neat, right?
But oxygen isn't just content with the air. It’s also found in rocks and minerals, like rust on an old bike or the shiny mica in some countertops. It’s a real team player, always ready to combine with other elements to create stable compounds.

The Mighty Carbon: The Backbone of Life
Next up, let's talk about carbon. This little guy is the undisputed champion of forming complex structures. Think of it as the master architect of the universe, capable of building everything from the tiniest single-celled organisms to, well, us!
Carbon is the foundation of all organic life. Everything that’s alive, from a giant redwood tree to a microscopic bacterium, is built on a framework of carbon atoms. Your very own body, your pet dog, the grass in your backyard – all are carbon-based marvels.
It's in the food we eat. That delicious slice of pizza? Carbon. That juicy apple? Carbon. Even the graphite in your pencil is a form of carbon. It’s practically everywhere you look, even if you can’t see it directly.
Carbon's superpower is its ability to link up with itself and other elements in an almost endless variety of ways. It’s like having a box of LEGOs that can build literally anything you can imagine. This versatility is what makes it so crucial for life as we know it.
Where Does Carbon Live? Everywhere!
Carbon is found in the atmosphere as carbon dioxide (CO₂), a greenhouse gas that helps keep our planet warm (though too much can be a problem, but that's a story for another day!). It’s in the oceans, dissolved in the water. It’s in the soil, a vital part of healthy ecosystems.

And, of course, it’s in all living things. When plants grow, they’re taking carbon dioxide from the air and turning it into their own tissues. When we eat plants (or animals that ate plants), we’re essentially consuming that stored carbon. It’s a grand cosmic recycling program!
The Halogens: A Zesty Bunch
Now let's meet a family of particularly feisty nonmetals: the halogens. This group includes elements like fluorine, chlorine, bromine, and iodine. They're known for being extremely reactive, meaning they really love to grab an electron from other atoms. Think of them as the enthusiastic puppy of the nonmetal world, always ready for a game of tug-of-war.
Chlorine is probably the most familiar halogen. You've encountered it in your swimming pool, keeping the water clean. It’s also used in disinfectants and to make PVC plastic, which is used in everything from pipes to window frames.
Then there's fluorine. This one is super reactive and found in toothpaste to help strengthen your teeth. It’s that little ingredient that gives you that extra layer of protection against cavities. Pretty cool for something so small!
Iodine is another important one. It's essential for your thyroid gland to function properly, and a lack of it can lead to health problems. That's why some table salt is "iodized" – a little sprinkle of this nonmetal to keep us healthy.
Found in Salt and the Sea
Halogens are often found in salts. In fact, the word "halogen" literally means "salt-former." They readily combine with metals to create ionic compounds that we know as salts. Table salt, or sodium chloride (NaCl), is a perfect example – a friendly partnership between a metal (sodium) and a nonmetal (chlorine).
You'll also find them in seawater. Bromine, for instance, is present in the ocean in significant amounts. So, next time you're by the sea, remember you're looking at a vast reservoir of these reactive nonmetals!
Sulfur and Phosphorus: The Smell and the Spark
Let's not forget about sulfur and phosphorus. Sulfur is famous for its distinctive smell – think of rotten eggs! While that smell might not be your favorite, sulfur is crucial for many biological processes and is a key component of proteins.
Phosphorus is another essential element for life. It’s a vital part of DNA, the blueprint of life, and is also found in our bones and teeth. It’s also famously used in matches – the head of a match contains phosphorus compounds that ignite when struck, giving you that satisfying spark.

Imagine striking a match on a dark evening. That little flare of light? That's phosphorus saying "hello!" It's a small but powerful reminder of the essential nonmetals at work.
In Rocks, Foods, and Fireworks
Sulfur can be found in various minerals and is released into the atmosphere through volcanic activity. It's also present in some foods like eggs and garlic. Phosphorus, on the other hand, is abundant in rocks and is essential for plant growth, making it a key ingredient in fertilizers.
Why Should You Care?
So, why all this fuss about nonmetals? Because they are the absolute bedrock of our existence! Without these active elements, life as we know it simply wouldn't be possible. They’re in the air we breathe, the water we drink, the food we eat, and even the technology we use.
Understanding where these nonmetals are and how they behave helps us understand our world better. It helps us develop new medicines, create cleaner energy sources, and even appreciate the intricate workings of our own bodies.
They are the unsung heroes, the quiet engines that drive so much of what we take for granted. So, the next time you take a deep breath, or enjoy a glass of water, or even just look at a plant, give a little nod to the amazing, active nonmetals that are making it all happen. They’re everywhere, and we’re all the better for it!