When Metals React Do They Gain Or Lose Electrons

Hey there, science enthusiasts and curious minds! Ever wondered what’s really going on when you see a shiny piece of metal… well, not so shiny anymore? You know, like when your favorite silver necklace starts to tarnish, or when that trusty old wrench gets a bit of rust? It’s not magic, folks, it’s chemistry, and it’s way cooler than you might think. Today, we’re diving into the dazzling world of metal reactions, and specifically, we’re going to tackle a question that might sound a little intimidating but is actually quite fun: When metals react, do they gain or lose electrons? Buckle up, because this is where things get interesting!
Now, before your eyes glaze over and you think about hiding under your duvet, let’s keep it light. We’re talking about the tiny, invisible building blocks of everything around us – atoms. And inside every atom, there are even tinier things called electrons. Think of electrons as little energy packages that whiz around the center of an atom, kind of like tiny planets orbiting a sun. They’re super important for how atoms interact with each other.
So, when we talk about metals reacting, we’re essentially talking about these atoms of metal wanting to get a little more comfortable, a little more stable, you know? Atoms, like people, sometimes want to find a more relaxed state. And for many metals, this means playing a little game of electron-swapping.
Must Read
The Great Electron Giveaway!
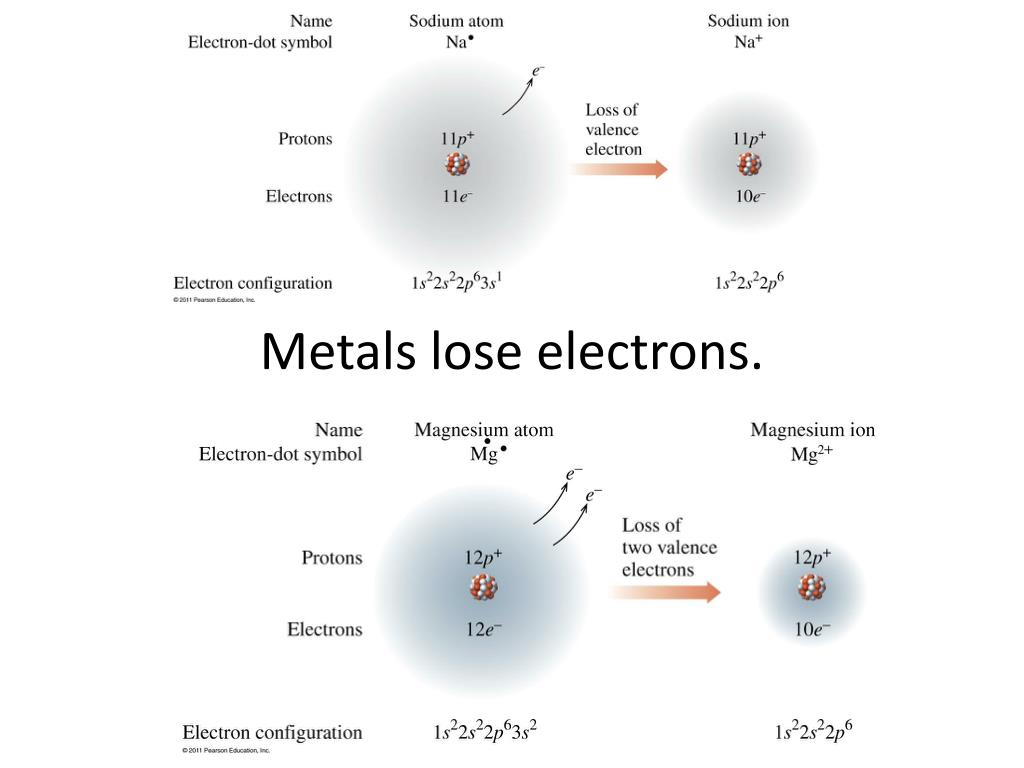
Here’s the big reveal, the moment of truth! When most metals decide to get social and start a reaction – whether it’s with oxygen in the air (hello, rust!) or something else entirely – they tend to be the generous givers. That’s right, metals usually LOSE electrons when they react. Ta-da!
Think about it this way: imagine a metal atom is like a person who has a few too many coins in their pocket. They feel a bit overloaded, a bit unstable. To feel better, to achieve that sweet spot of stability, they decide to hand some of those coins (our electrons!) over to someone else. And poof! They’ve just donated an electron.

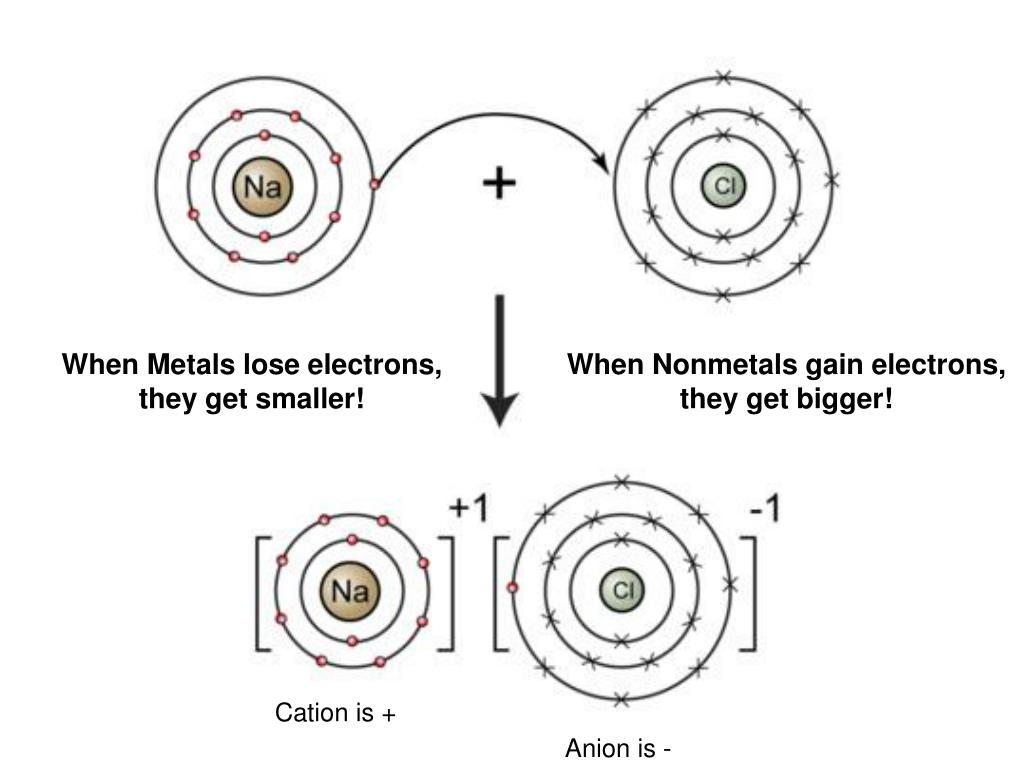
This electron-losing act is a big deal in the chemistry world. When a metal atom loses electrons, it becomes what we call a positive ion. Why positive? Because electrons have a negative charge. So, when you take away something negative, what’s left? Yep, a positive vibe! This positive ion is now feeling a lot more chill and stable.
Why So Generous, Metals?
You might be asking yourself, "Why are metals so keen on giving away their precious electrons?" It all comes down to achieving a more stable electron configuration. Think of it like having a perfectly balanced set of shelves. If an atom has an electron configuration that's a little wobbly, it's like having too many books on one shelf and not enough on another. It's just not ideal! By losing those extra electrons, metal atoms can often reach a more desirable, more stable arrangement, similar to how you'd rearrange your books for that satisfyingly even look.
This tendency for metals to lose electrons is why they’re often used in things that involve electricity. Electricity, at its core, is the flow of electrons! So, metals, being happy to let go of their electrons, are fantastic at conducting electricity. Think of the wires in your house, or the battery in your phone – they’re all powered by the amazing electron-donating abilities of metals. Pretty neat, right? It’s like they’re little electron highways!

Consider that shiny copper pot you love to cook with. Copper is a metal, and when it reacts (say, with air over time, though it’s a slow process for copper to tarnish noticeably), it’s giving up electrons. This electron transfer is what can lead to those subtle color changes we sometimes see. It's a silent, atomic party happening all the time!
Let's take iron, for example. That common metal is notorious for rusting. Rust is essentially iron oxide. What’s happening there? The iron atoms are reacting with oxygen. And in this reaction, guess what? The iron atoms are losing electrons to the oxygen atoms. The oxygen atoms are actually quite eager to snatch up those electrons! This electron exchange is the fundamental reason why iron transforms into that flaky, reddish-brown rust. It’s a bit sad for the iron, but it’s a beautiful demonstration of electron transfer.
It's All About Balance
Now, not every element is like this, of course. Some elements, like nonmetals, are often on the receiving end of these electron donations. They’re the ones who are happy to gain electrons to achieve their own stable configurations. It’s a cosmic give-and-take, a constant dance of electrons. Think of it like a potluck dinner: some people bring the main dish (metals, losing electrons), and others bring the dessert (nonmetals, gaining electrons). Everyone’s happy and well-fed, in an atomic sense!
This whole electron game is the foundation of so many chemical reactions. It’s how batteries work, how metals corrode, and even how your body uses certain elements. It’s a fundamental principle that governs the material world around us. And understanding it, even a little bit, can make you see everyday objects in a whole new light.
So, the next time you see a piece of metal, whether it's a glittering piece of jewelry or a sturdy bridge, remember the incredible microscopic ballet of electrons that’s happening within it. Metals are usually the generous ones, the electron-losers, the ones who become positive ions, all in the name of stability. It’s a simple concept with profound implications.
Making Life More Electrifying!
Doesn't that make you feel a little more… electrified? Knowing that these tiny, invisible electrons are constantly on the move, making things happen? It’s like having a secret superpower of observation. You can look at a rusty bike and understand the fundamental reason behind its transformation. You can appreciate the conductivity of a metal wire because you know about its electron-donating tendencies.
This understanding opens up a whole new world. It’s not just about memorizing facts; it’s about grasping the underlying principles that make our universe tick. It’s about realizing that even the most ordinary things are governed by extraordinary scientific laws.
So, don’t shy away from these questions! Embrace them! When metals react, they do indeed gain or lose electrons. And in the case of most metals, they are the generous souls who lose them. They become positive ions, eager to find that sweet spot of stability. Isn’t that just wonderfully optimistic?
Keep exploring, keep questioning, and keep marveling at the incredible science that surrounds us. The more you learn, the more you’ll see the magic in the mundane. Go forth, and let your curiosity be your guide!

.+%2B+1e-+lost.+Lithium.+Lithium+ion+(Li%2B).jpg)
