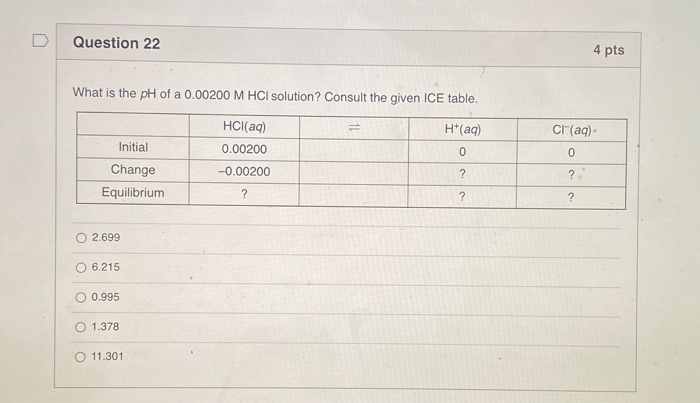
What Is The Ph Of A 0.00200 M Hclo4 Solution
Hey there, curious minds! Ever found yourself wondering about the nitty-gritty of chemistry, even if it's just a tiny bit? Like, what’s that whole "pH" thing really about? Today, we're diving into a specific, and frankly, pretty cool question: What is the pH of a 0.00200 M HClO4 solution? Sounds a bit technical, right? But stick with me, because it's actually a neat little puzzle with a satisfying answer.
First off, let's break down what those numbers mean. "M" stands for molarity, which is basically a fancy way of saying how concentrated a solution is. So, 0.00200 M HClO4 means we've got 0.00200 moles of perchloric acid (HClO4) dissolved in every liter of water. Think of it like how much sugar you dissolve in your iced tea – a little bit versus a whole lot. This is a pretty dilute solution, so it's like adding just a teaspoon of sugar to a giant pitcher of iced tea.
Now, the star of our show: HClO4. That’s perchloric acid. And when it comes to acids, perchloric acid is a real powerhouse. It's what chemists call a strong acid. What does that mean in practical terms? Well, imagine you have a bunch of tiny acid molecules. A strong acid like HClO4 is super eager to give away its hydrogen ions (which is what makes something acidic). In water, it pretty much breaks apart 100% into its constituent parts: a hydrogen ion (H+) and a perchlorate ion (ClO4-).
Must Read
This 100% dissociation is key! It’s like a perfectly ripe fruit that’s just begging to be picked. There are no half-hearted giveaways of hydrogen ions with HClO4. It's all or nothing. So, if we start with 0.00200 moles of HClO4, we end up with 0.00200 moles of H+ ions floating around in that liter of water. Easy peasy, right?
So, what's the pH deal?
Okay, so we know how many H+ ions we have. But how does that translate to pH? pH is our scale for measuring how acidic or basic something is. It's like a thermometer for acidity! The scale usually runs from 0 to 14. pH 7 is neutral, like pure water. Anything below 7 is acidic (think lemon juice or vinegar), and anything above 7 is basic or alkaline (think baking soda or soap). The lower the pH, the more acidic it is.

The cool thing is that pH is a logarithmic scale. This means that each whole number change on the pH scale represents a 10-fold change in acidity. So, a pH of 3 is 10 times more acidic than a pH of 4, and 100 times more acidic than a pH of 5! It's a bit like how decibels work for sound – a small increase in the number means a big jump in loudness.
The formula to calculate pH is pretty straightforward: pH = -log[H+]. Here, [H+] is the concentration of hydrogen ions we just figured out. In our case, [H+] is 0.00200 M. So, we need to calculate the negative logarithm of 0.00200.
Let's crunch some numbers!
Alright, time for a little math adventure. We need to find the negative logarithm of 0.00200. It might sound a bit intimidating, but it's really just plugging numbers into a calculator.

We can rewrite 0.00200 as 2.00 x 10-3. This scientific notation is super handy for dealing with very small or very large numbers. It’s like packing your ingredients into pre-measured bags before baking – makes the process smoother.
So, the equation becomes: pH = -log(2.00 x 10-3).
When you punch that into a calculator, you get something like 2.69897. Now, in chemistry, we usually round our pH values to a reasonable number of significant figures. Since our concentration (0.00200 M) has three significant figures (the 2 and the two zeros after it), our pH should also be reported with two decimal places.

So, rounding 2.69897 to two decimal places gives us pH = 2.70.
And there you have it! The pH of a 0.00200 M HClO4 solution is approximately 2.70.
Why is this cool?
Well, for starters, a pH of 2.70 is pretty acidic! Think about common household items. Lemon juice is typically around a pH of 2. Vinegar is usually around 2.5 to 3. So, our 0.00200 M HClO4 solution is in the ballpark of being as acidic as lemon juice. It's definitely more acidic than something like milk (pH around 6.5) or even plain water (pH 7).

This acidity means it has the potential to react with things. Strong acids can corrode metals, and they taste very sour (though you should NEVER taste lab chemicals!). Understanding the pH helps chemists predict how substances will behave and interact with their surroundings. It’s like knowing the personality of your ingredients before you start cooking – it tells you what you can expect!
Also, the fact that we can calculate this precisely from a simple concentration is pretty neat. It shows how mathematical relationships underpin the physical world around us. We take a simple measurement (how much acid is there) and use a bit of math magic to understand a fundamental property (its acidity). It’s a little glimpse into the order and logic of the universe, wouldn't you say?
So, next time you hear about pH or strong acids, you'll have a better appreciation for what it all means. It's not just numbers; it's a way of describing the chemical character of a solution, and in the case of HClO4, a rather potent character indeed! Keep those curious questions coming; the world is full of fascinating little puzzles just waiting to be solved!