What Is The Mass Of Exactly 500 Sulfur Atoms

Ever wondered about the tiny, invisible building blocks that make up everything around us? It's a fascinating world, and today we're going to have some fun exploring it by figuring out the mass of a very specific number of sulfur atoms. Sounds a bit nerdy? Maybe! But understanding these fundamental concepts is like unlocking a secret code to how the universe works, and it can be surprisingly rewarding and useful for everyone.
Why is this kind of thing interesting? Well, for beginners, it’s a gentle introduction to the world of chemistry and the concept of atoms – the tiny particles that make up all matter. For families, it can be a great way to spark curiosity in kids about science. Imagine showing them how even something as small as a speck of dust is made of countless atoms, and how we can even calculate their weight! For hobbyists, whether you're into gardening (understanding soil nutrients), brewing (precision in ingredients), or even just appreciating the materials that make up your favorite gadgets, a grasp of these basic scientific principles can add a whole new layer of understanding and enjoyment.
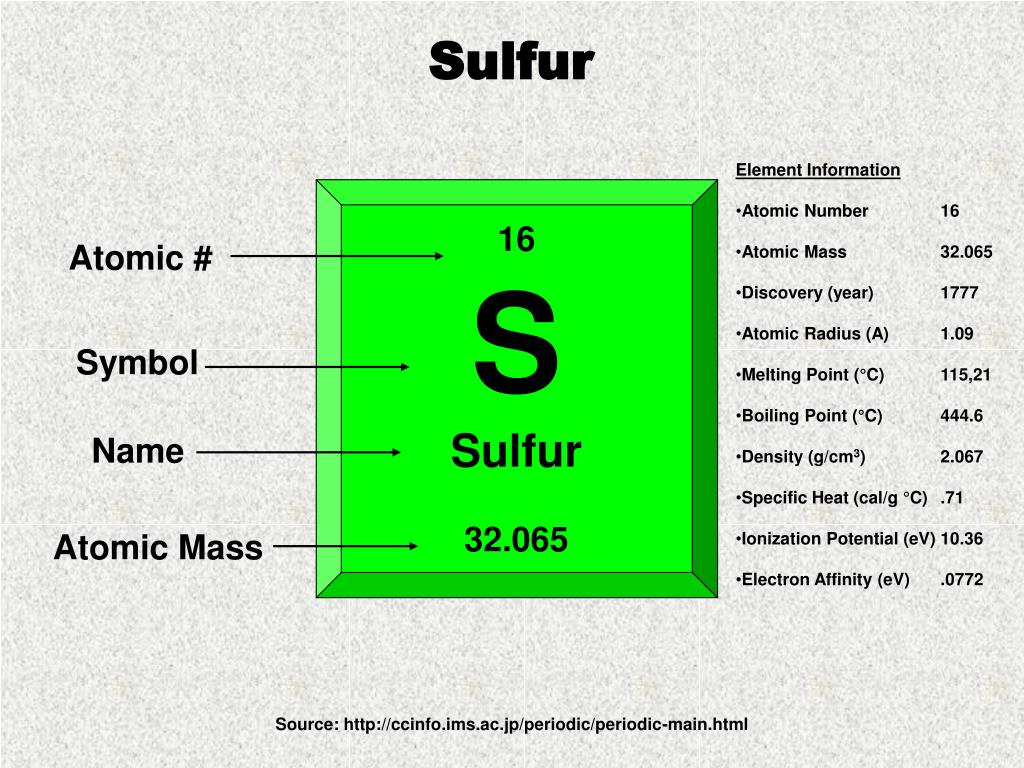
So, how do we find the mass of exactly 500 sulfur atoms? It all comes down to a concept called the atomic mass unit (amu) and Avogadro's number. Don't let the fancy names scare you! Think of it this way: each element, like sulfur, has a standard weight for one of its atoms. Sulfur's average atomic weight is about 32.06 amu. But we rarely deal with single atoms. We usually deal with moles, which are a huge collection of atoms. One mole of any substance contains about 6.022 x 1023 particles (atoms or molecules). This is Avogadro's number!
Must Read
To find the mass of 500 sulfur atoms, we can do a couple of simple calculations. First, we know the mass of one mole of sulfur is approximately 32.06 grams. Since 500 atoms is a tiny fraction of a mole, its mass will be equally tiny. We can calculate it by taking the mass of one atom (which is atomic mass in grams, or 32.06 grams / 6.022 x 1023 atoms) and multiplying it by 500.

Here's a simple tip for getting started with this kind of thinking: the next time you see something made of sulfur (like in matches or fertilizers, though be cautious with these!), think about the sheer number of atoms involved. You can also look up the atomic masses of other elements you find interesting, like oxygen or carbon, and see how their weights compare. For a fun variation, try figuring out the mass of 1000 atoms of a different element!
Understanding the mass of even a small number of atoms might seem abstract, but it connects us to the fundamental nature of matter. It’s a small step into a vast and wondrous scientific landscape, offering a glimpse into the power of precision and calculation that underpins so much of our modern world. It's a journey of discovery, and it all starts with asking "how much does this weigh?"




+are+in+1.8+x+atoms+of+sulfur+(Use+dimensional+analysis).jpg)