What Is The Mass In Grams Of 5.50 Moles Nh3

Hey there, fabulous humans! Ever find yourself staring at a science problem and thinking, "Ugh, is there a way to make this less… science-y?" Well, today, we're diving into a little bit of chemistry, but think of it more like a delightful culinary adventure, or perhaps a peek behind the curtain of your favorite skincare products. We’re talking about ammonia, that pungent, yet surprisingly useful, molecule you might have encountered in cleaning supplies or even as a natural byproduct of… well, let’s just say life. The question on everyone's mind (or at least, the one that popped into our heads on a particularly curious Tuesday) is: What is the mass in grams of 5.50 moles of NH3?
Now, before you picture yourself back in a dimly lit lab, struggling with Bunsen burners and scribbling furiously in a notebook, let’s take a deep breath. This isn't about acing a final exam; it's about understanding the building blocks of the world around us in a way that feels, dare I say, accessible. Think of moles as the chemists' favorite way to count things – a super-sized unit that’s way more convenient than trying to count individual atoms. It’s like when you’re baking and you need a dozen eggs; you don't count them one by one, you just grab the carton. A mole is essentially a carton of molecules, containing a whopping 6.022 x 1023 particles. Mind-boggling, right? We call this Avogadro's number, and it’s a cornerstone of chemistry, much like the perfect sourdough starter is to a baker.
So, we have 5.50 moles of NH3. That’s 5.50 cartons of ammonia molecules. Pretty sweet deal, considering how much work goes into making just one carton of eggs! But how do we translate this quantity into something we can actually weigh? This is where the concept of molar mass swoops in, like a perfectly timed plot twist. The molar mass is simply the weight of one mole of a substance, expressed in grams per mole (g/mol). It’s the key to unlocking the grams from our moles.
Must Read
To find the molar mass of ammonia (NH3), we need to look at its individual components: nitrogen (N) and hydrogen (H). Imagine building an ammonia molecule like you’re assembling a Lego set. You need one nitrogen brick and three hydrogen bricks. We’ll need to consult our trusty periodic table, which is basically the ultimate ingredient list for the universe. Nitrogen (N) has an atomic mass of approximately 14.01 g/mol. Hydrogen (H), bless its tiny, energetic heart, has an atomic mass of about 1.01 g/mol. Remember, we have three hydrogen atoms in each ammonia molecule, so we need to account for all of them.
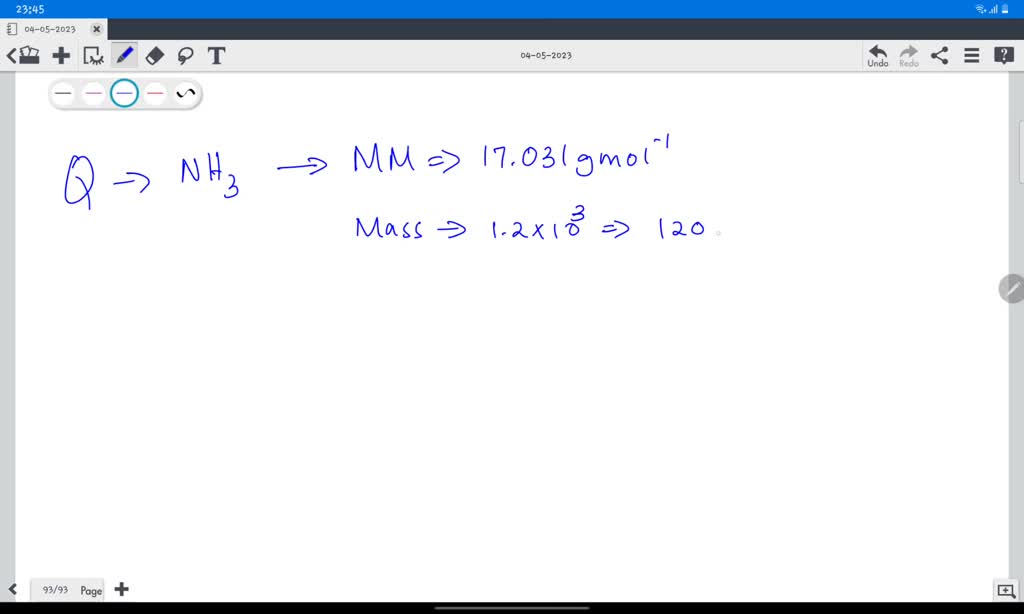
So, the molar mass of NH3 is calculated by adding up the atomic masses of its constituent atoms: Molar Mass of NH3 = (Atomic Mass of N) + 3 * (Atomic Mass of H) Molar Mass of NH3 = 14.01 g/mol + 3 * (1.01 g/mol) Molar Mass of NH3 = 14.01 g/mol + 3.03 g/mol Molar Mass of NH3 = 17.04 g/mol
There we have it! One mole of ammonia weighs 17.04 grams. This is like knowing that one dozen eggs weighs, say, 700 grams. Now we can figure out the weight of our 5.50 cartons!

To find the mass in grams of 5.50 moles of NH3, we simply multiply the number of moles by the molar mass: Mass (g) = Number of Moles (mol) * Molar Mass (g/mol) Mass (g) = 5.50 mol * 17.04 g/mol
Let’s do the math, and try not to get lost in the numbers! 5.50 * 17.04 = 93.72
So, the mass in grams of 5.50 moles of NH3 is approximately 93.72 grams. Isn’t that neat? We just took a quantity of molecules and turned it into a tangible weight. It’s like magic, but with slightly more reliable predictions!
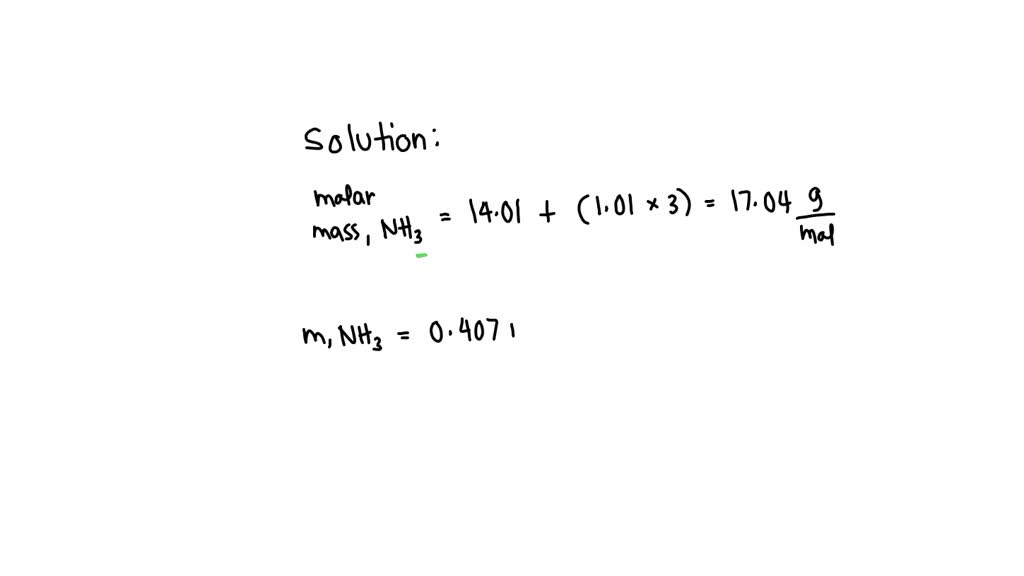
Now, why should this matter to your average, everyday awesome person? Well, ammonia, despite its sometimes-intimidating smell, plays a role in so many things we interact with. For instance, it's a crucial component in the production of fertilizers, which help grow the food that ends up on our dinner plates. Without ammonia, our agricultural yields would be significantly lower, and avocado toast might become a very, very rare commodity. Think about that next time you’re enjoying a fresh salad – you’re indirectly thanking ammonia!
Ammonia is also a key ingredient in the manufacturing of certain plastics, like nylon, which is used in everything from clothing to carpets. So, that comfy sweater you’re wearing? Yep, ammonia might have had a hand in its creation. It’s also used in the production of pharmaceuticals and even in some food additives (though its direct use in food is more regulated and less common than its role in production). It’s a true unsung hero of modern industry.
And let’s not forget its starring role in many common household cleaning products. That streak-free shine on your windows? Ammonia is often the secret weapon. Just remember to use it in a well-ventilated area – it’s potent stuff! It’s a bit like that friend who’s a little bit too loud at parties but ultimately brings the good vibes and gets the job done. Always remember to respect its power and handle it with care, just like you would any potent ingredient in your favorite recipe.

Think about the world of cosmetics and personal care too. While pure ammonia isn't typically found in your everyday moisturizer, its derivatives and related compounds are used in various formulations. For example, some hair dyes use ammonia to open up the hair cuticle, allowing the color to penetrate. So, that vibrant new hair color you might be sporting? Ammonia’s got a supporting role there, too.
Understanding these basic chemical concepts, like molar mass and moles, isn't just for scientists in lab coats. It helps us appreciate the intricate web of production and transformation that brings us the things we rely on every day. It’s about demystifying the world and realizing that even seemingly complex subjects can be broken down into understandable parts, much like deciphering a new recipe or understanding the ingredients in your favorite K-beauty serum.
It’s also fascinating to consider how different cultures have historically used or interacted with ammonia-producing substances. For example, in ancient Egypt, urine was collected and used for its ammonia content in tanning leather and cleaning textiles. While perhaps not the most glamorous application, it highlights the long-standing recognition of ammonia’s properties and its practical utility long before we had fancy periodic tables and mole calculations. It’s a testament to human ingenuity and observation, finding uses for materials in their environment.

Our little calculation, 5.50 moles of NH3 equaling 93.72 grams, might seem like a small piece of trivia, but it’s a gateway to understanding larger processes. It’s the difference between admiring a perfectly baked cake and understanding the role of yeast in making it rise. It’s the difference between enjoying a new outfit and appreciating the complex chemical processes that made the fabric. It’s about adding a layer of conscious appreciation to our daily experiences.
In a way, this whole exercise is a bit like learning a new language. At first, the terms and rules might seem daunting, but as you practice and apply them, they start to make sense, revealing a deeper understanding of the world. This simple conversion from moles to grams is a foundational phrase in the language of chemistry, and once you’ve got it, a whole new vocabulary of the material world opens up to you.
So, next time you encounter a number followed by "moles" or a chemical formula like NH3, don't feel intimidated. Take a moment, channel your inner curious explorer, and remember that with a little bit of molar mass magic, you can translate those abstract quantities into something tangible, something that connects you to the products, the food, and the very fabric of the world around you. It’s a small step, but it’s a step towards understanding, and that, my friends, is always a beautiful thing.
Think about it this way: your day is made up of countless tiny interactions and processes. From the water you drink to the electricity powering your devices, everything has a composition, a structure, and a history. Learning to quantify these things, even in a simple way like calculating the mass of a few moles of a molecule, is like gaining a new lens through which to view your reality. It adds a layer of appreciation for the unseen work that goes into making our lives comfortable and convenient. It’s a reminder that even the smallest components, when combined in the right way, can lead to something significant – a lesson that resonates far beyond the chemistry lab and into the grand experiment of our own lives.