What Is The Lewis Dot Structure For Nitrogen

Ever wondered what those little dots around chemical symbols mean? Well, you're in for a treat, because understanding Lewis dot structures, especially for elements like nitrogen, is like unlocking a secret code in the world of chemistry! It's not just for scientists; it's a super accessible way to visualize how atoms interact, and honestly, it can be quite fun and surprisingly useful.
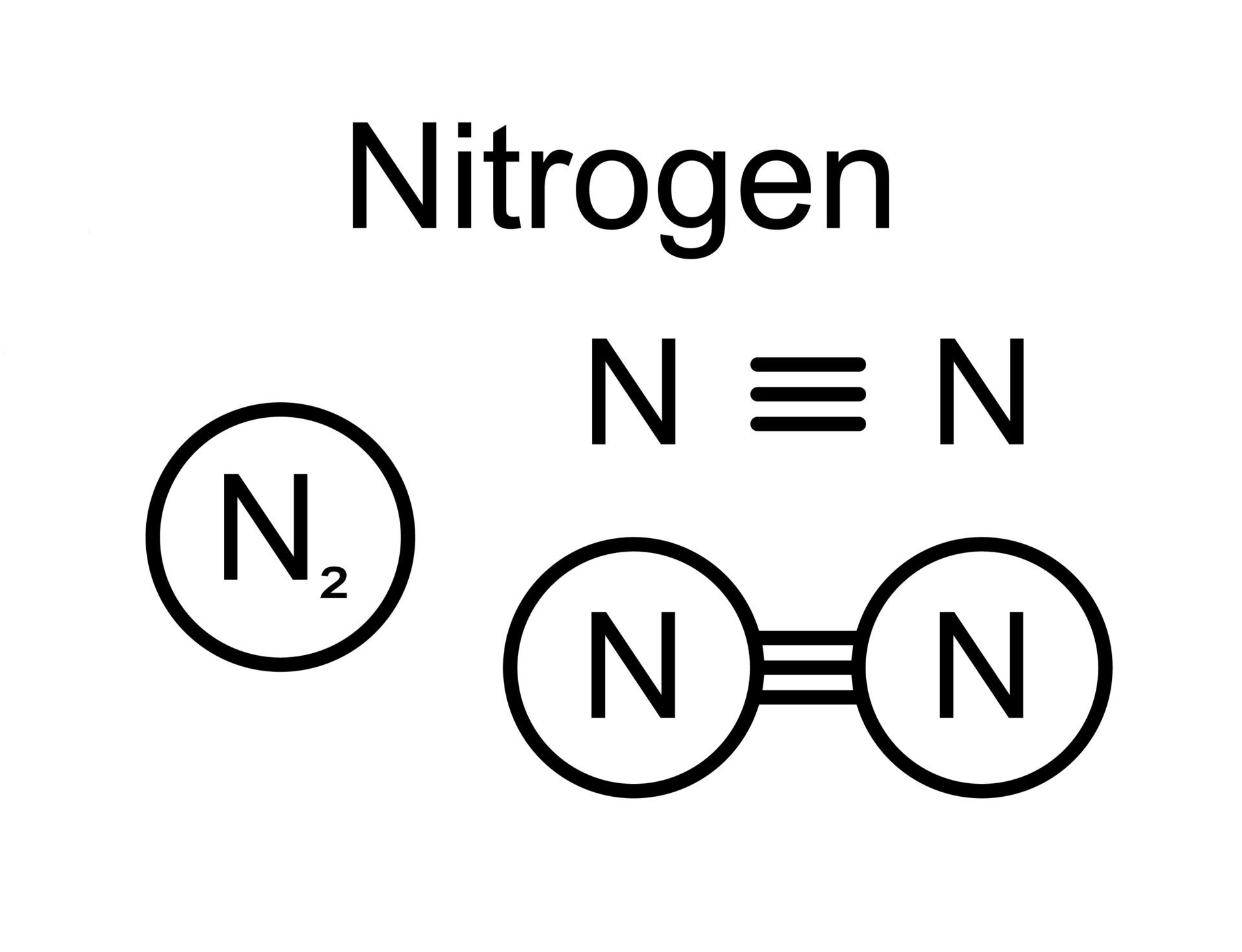
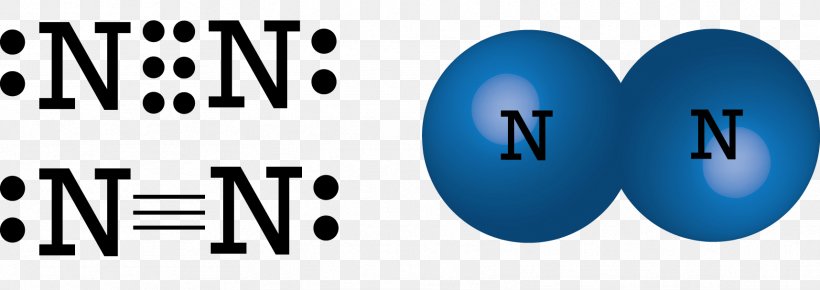
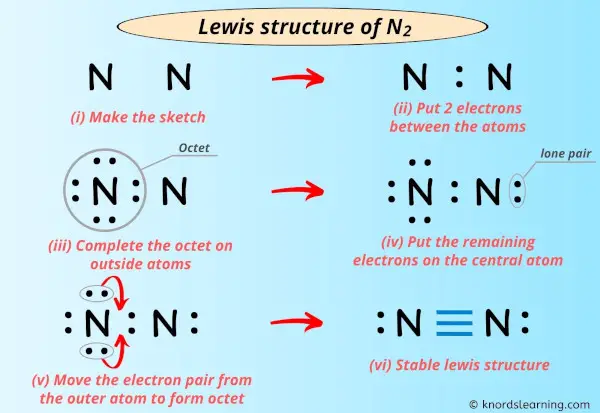
So, what exactly is a Lewis dot structure for nitrogen? Think of it as a simple drawing that shows an atom's valence electrons – those are the electrons on the outermost shell, the ones involved in bonding. For nitrogen, its symbol is 'N', and it has 5 valence electrons. In a Lewis dot structure, these 5 electrons are represented by dots placed around the symbol 'N'. We usually place one dot on each of the four sides first, then start pairing them up. So, you'd see 'N' with one pair of dots and three single dots, like a little nitrogen house with a sleeping area and three individual rooms!
Why is this useful? If you're a beginner just dipping your toes into chemistry, Lewis dot structures are your best friend. They make abstract concepts like bonding much more concrete and easy to grasp. For families exploring science together, it’s a fantastic visual aid for explaining how different elements can stick together to form molecules. Imagine showing your kids how nitrogen forms part of the air we breathe or even vital molecules in our bodies – it's a real-world connection! And for hobbyists, whether you're into gardening (nitrogen is key for plant growth!) or even certain crafts that involve chemical reactions, having this basic understanding can add a whole new dimension to your interests.
Must Read
Nitrogen is particularly interesting because it's so crucial. It's the 'N' in DNA, it's a major component of proteins, and it forms the diatomic molecule N₂, which makes up about 78% of the air around us. When nitrogen atoms bond with other atoms, like in ammonia (NH₃), you can see how the electrons are shared or transferred. In ammonia, the nitrogen atom shares its electrons with three hydrogen atoms, forming a stable molecule. Seeing this visually through Lewis dot structures makes the process of chemical bonding much clearer than just reading about it.
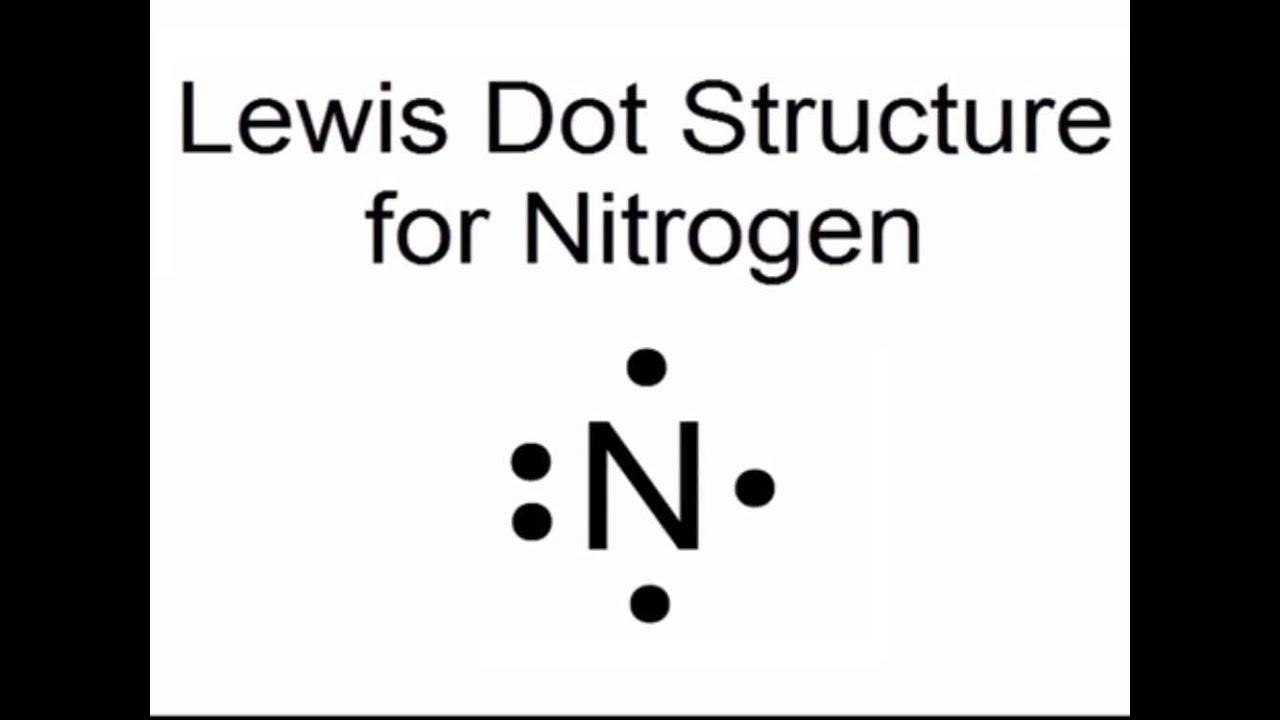
Getting started is a breeze! All you need is the chemical symbol for the element and a little knowledge about its valence electrons. For nitrogen, remember it's 'N' and it has 5 valence electrons. Grab a piece of paper and a pencil, write down 'N', and start dotting! There are plenty of online resources that show examples of Lewis dot structures for various elements and common molecules. You can even try drawing structures for other familiar elements like oxygen (O) or carbon (C) to practice.
So, the next time you hear about nitrogen, picture those little dots! Understanding the Lewis dot structure for nitrogen is a simple yet powerful way to demystify chemistry. It's an enjoyable skill that opens up a better appreciation for the building blocks of our universe, making science feel less intimidating and more like an accessible, fascinating puzzle.