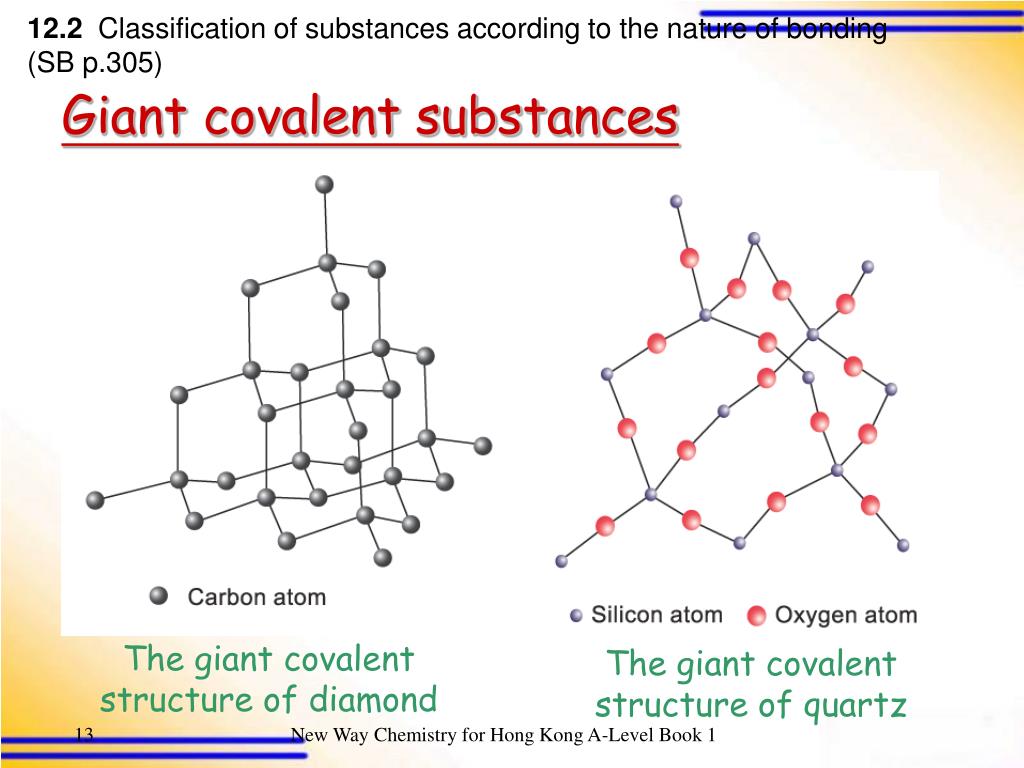
What Is An Example Of A Giant Covalent Structure

Ever looked at something incredibly strong, something that seems to defy the usual rules of how things break or bend, and wondered what makes it tick? We’re not talking about your favorite superhero’s invincibility here, but the fascinating world of chemistry and the sheer power of tiny atoms linked together in truly epic ways. Today, we’re diving into the realm of giant covalent structures, and trust us, it's less of a dry science lesson and more of an exploration of the titans of the material world.
Think about it. We interact with these structures every single day, often without even realizing the incredible engineering happening at the molecular level. From the gleam of your phone screen to the hardness of a diamond ring, these are the unsung heroes of our modern lives. They’re the quiet giants, the bedrock of so much that we take for granted. So, grab a cozy beverage, settle in, and let’s unlock the secrets of these awesome molecular architectures.
The Unbreakable Bonds: What Exactly Are We Talking About?
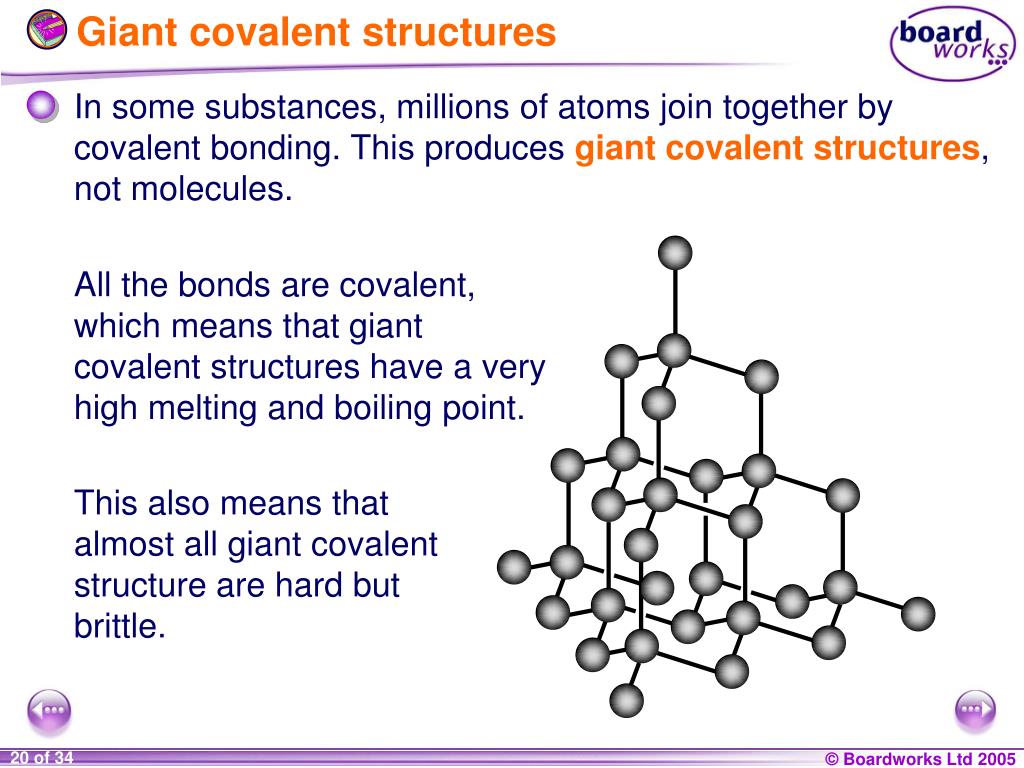
Alright, let’s break down the jargon, shall we? A covalent bond is basically a handshake between atoms. They decide to share electrons, creating a super-stable connection. Now, imagine this handshake happening not just once, but over and over again, creating a massive, interconnected network. That’s a giant covalent structure in a nutshell. It's like a huge, intricate web where every atom is tightly bound to its neighbors, forming a single, colossal molecule.
Must Read
Unlike simple molecules, like water (H₂O) where you have a few atoms grouped together, giant covalent structures are… well, giant! They don't form discrete little units. Instead, they extend in all three dimensions, creating a solid lattice where the covalent bonds are the glue holding the whole thing together. It’s this continuous network of strong bonds that gives these materials their extraordinary properties. They're the ultimate team players of the atomic world.
Think of it like a perfectly knitted sweater. Each stitch is connected to the ones around it, forming a single, cohesive piece. Now imagine that sweater stretched out to cover an entire building – that’s kind of the scale we’re talking about with these structures, albeit with atoms instead of yarn!
Diamond: The King of Hardness and Sparkle
When you hear "giant covalent structure," the first thing that probably pops into your head is diamond. And for good reason! This precious gem is the poster child for this type of bonding, and its properties are nothing short of legendary. A diamond is essentially a colossal molecule where each carbon atom is covalently bonded to four other carbon atoms in a tetrahedral arrangement. This creates an incredibly strong, three-dimensional network.
This arrangement means that every single carbon atom is involved in strong, shared bonds. There are no weak points, no easy places to break. This is why diamonds are renowned for their incredible hardness. They rank a perfect 10 on the Mohs scale of mineral hardness, meaning they can scratch virtually any other substance. It’s why diamond-tipped tools are used for cutting and grinding.
But it's not just about toughness. The specific arrangement of carbon atoms in a diamond also gives it its dazzling sparkle. Light enters the diamond, bounces around internally due to its refractive properties, and then exits, creating that iconic brilliance. It's a testament to how the microscopic structure dictates macroscopic beauty.
Fun Fact: Did you know that the industrial production of synthetic diamonds has come a long way? They’re now used in everything from drill bits to specialized coatings, proving that these ‘gems’ are more than just pretty faces!
Practical Tip: Thinking of buying a diamond? Understand the 4 Cs: Carat, Cut, Color, and Clarity. While its natural hardness is a given, the way it's cut significantly impacts its sparkle and value. And remember, for everyday wear, a slightly less perfect stone can still be incredibly beautiful and more budget-friendly.
Silicon Dioxide: The Ubiquitous Foundation
If diamond is the king, then silicon dioxide (SiO₂) is the hardworking queen, quietly supporting much of our modern world. You probably know it better as silica. This compound is found everywhere – in sand, quartz, glass, and even in many of the electronic components that power our devices.
In silicon dioxide, each silicon atom is bonded to four oxygen atoms, and each oxygen atom is bonded to two silicon atoms. This forms a vast, repeating network. Unlike diamond’s perfect tetrahedral structure, silicon dioxide often forms a more varied, interconnected framework. This leads to a material that is also very hard and has a high melting point, but is less rigid than diamond.
Think about the glass in your windows. That’s essentially fused silicon dioxide. Its transparency, durability, and resistance to heat make it an indispensable material for countless applications. Sand itself is largely composed of silica grains, a testament to its abundance.

Cultural Reference: The iconic glass pyramids of the Louvre in Paris are a beautiful example of how silicon dioxide, in its glass form, can be integrated into stunning architecture. It’s a material that balances strength with aesthetic appeal.
Fun Fact: Some varieties of quartz, a form of silicon dioxide, are used in watches to keep incredibly accurate time. The quartz crystal vibrates at a precise frequency when an electric current is applied, making it a highly reliable timekeeping mechanism.
Practical Tip: When handling glassware, especially older pieces, remember that while strong, they can still be brittle. Treat them with a little extra care, and consider using placemats and coasters to protect surfaces and prevent accidental chips.
Graphite: The Slippery Surprise
Now, here’s where things get really interesting. Graphite is another giant covalent structure made entirely of carbon atoms, just like diamond. But its properties are vastly different! This is where the magic of arrangement really shines.
In graphite, the carbon atoms are arranged in flat layers, with each carbon atom bonded to three other carbon atoms in a hexagonal pattern. These layers are stacked on top of each other, but the bonds between the layers are much weaker than the strong covalent bonds within each layer. This difference in bonding is key to graphite’s unique characteristics.
This layered structure explains why graphite is so slippery. The layers can easily slide past each other, making it an excellent lubricant. Think about the 'lead' in your pencil – it's actually graphite, and the reason it leaves a mark is because layers of graphite rub off onto the paper as you write.

Furthermore, graphite is a surprisingly good conductor of electricity. This is because within each layer, there are delocalized electrons that are free to move. This conductivity, combined with its high melting point, makes graphite useful in applications like electrodes for batteries and in the manufacture of certain high-temperature materials.
Cultural Reference: The classic pencil is an everyday marvel powered by graphite’s unique structure. Imagine all the art, notes, and ideas that have flowed through a simple graphite pencil!
Fun Fact: The black, sooty appearance of graphite is a direct result of its layered structure and the way light interacts with those flat surfaces.
Practical Tip: If you’re using a graphite lubricant, remember a little goes a long way. Too much can make things excessively messy. And when working with graphite, be mindful that it can transfer easily, so protect your surfaces and clothing.
Beyond the Big Three: Other Titans of the Material World
While diamond, silicon dioxide, and graphite are the most well-known examples, the world of giant covalent structures extends further. Many silicon compounds and boron compounds also exhibit this type of bonding, leading to materials with exceptional strength and high melting points. For instance, silicon carbide (SiC) is another incredibly hard and durable material, often used in abrasives and high-performance ceramics.
These materials are often engineered for specific, demanding applications. They are the unsung heroes in industries that require extreme durability, heat resistance, or electrical properties. They might not be as glamorous as a diamond, but their contribution to technological advancement is undeniable.
Fun Fact: Some allotropes (different structural forms) of silicon, like specific crystalline arrangements, can also be considered giant covalent structures with unique electronic properties, making them relevant in semiconductor technology.
Practical Tip: When you encounter products made from these advanced materials (like high-performance brake pads or certain types of specialized tools), you’re often interacting with the result of giant covalent bonding. Appreciating their origin can add a layer of wonder to everyday objects.
The Unseen Strength: Connecting to Our Lives
So, what’s the takeaway from all this talk of atoms and bonds? It’s a reminder of the incredible power of structure. The way atoms connect dictates everything from the sparkle of a gem to the utility of a pencil. These giant covalent structures are the silent architects of our world, providing the strength, resilience, and functionality that we rely on.
Think about it the next time you pick up a glass, admire a piece of jewelry, or even jot down a quick note. You’re holding something that, at its core, is an extraordinary example of nature’s engineering prowess. It’s a testament to how even the smallest building blocks, when arranged just right, can create something truly magnificent and enduring.
These aren't just abstract scientific concepts; they are the very fabric of our material reality. They inspire us to think about innovation, about building things that last, and about the beauty that can be found in the most fundamental of connections. It's a little bit of everyday magic, waiting to be noticed.