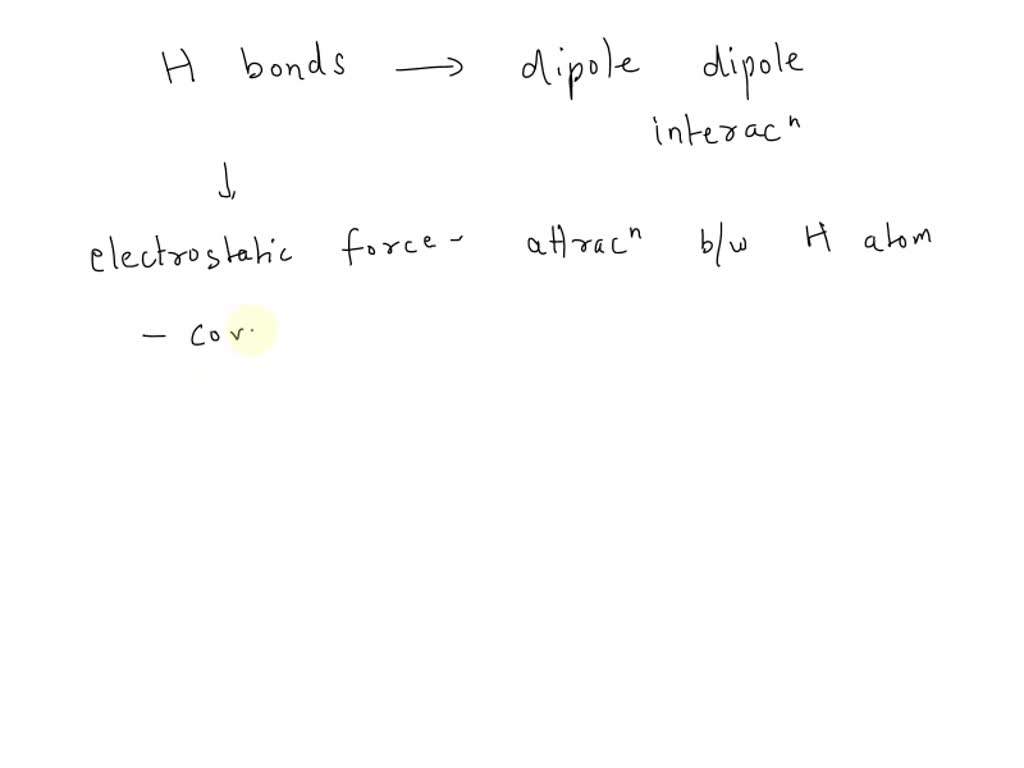
Weak Chemical Bonds Such As Hydrogen Bonds

In the vibrant world of creativity, some of the most captivating and delicate expressions often stem from the seemingly weakest of connections. Think of the gentle whisper of wind chimes, the ethereal shimmer of dew on a spiderweb, or the fleeting blush on a sunset sky. These beautiful moments, so easily overlooked, are often powered by forces that, in the grand scheme of chemistry, are quite subtle. Today, we're diving into the enchanting realm of weak chemical bonds, specifically focusing on the marvel that is the hydrogen bond, and exploring how its understated strength can inspire and empower your own creative endeavors.
For artists, crafters, and even the curious mind just looking for a new hobby, understanding and appreciating these gentle forces can unlock a treasure trove of inspiration. Imagine a painter capturing the subtle translucence of watercolor washes, where the water molecules' hydrogen bonds with pigment particles create those dreamy, blended effects. Or a sculptor working with clay, recognizing how the hydrogen bonds between water and clay particles give it that perfect, malleable consistency. Even a baker can appreciate the role of hydrogen bonds in the fluffy texture of a meringue or the elasticity of bread dough!
The beauty of hydrogen bonds lies in their flexibility and impermanence. Unlike the strong, rigid bonds that hold atoms together in a molecule, hydrogen bonds are like fleeting friendships – they can form, break, and reform with ease. This makes them incredibly versatile. Think of the intricate patterns seen in frost crystals, each unique yet all formed by the same fundamental water molecule interactions. Consider the way certain dyes adhere to fabrics, often facilitated by these weak attractions. Even in the natural world, the way leaves unfurl or petals open to the sun can be influenced by the water's cohesive properties, driven by hydrogen bonding.
Must Read
Ready to bring a little of this subtle science into your own creative space? You don't need a lab coat! Start with simple experiments that highlight these principles. Try making ice sculptures and observe how the water molecules arrange themselves. Experiment with different types of paper and how they absorb water – you’ll see variations influenced by the paper’s chemical makeup and its interaction with water’s hydrogen bonds. Even something as simple as observing condensation on a cold glass can be a lesson in molecular attraction. For crafters, consider working with materials that respond to moisture, like certain types of paper, natural fibers, or even edible gels, and see how you can manipulate them by controlling humidity – a direct nod to the power of hydrogen bonds.
What makes exploring these weak bonds so enjoyable is that it encourages us to look closer, to appreciate the understated elegance in everyday phenomena. It's about finding the profound in the seemingly simple, and recognizing that even the gentlest of forces can contribute to the most magnificent creations. So next time you see a shimmering drop of water or a perfectly formed snowflake, remember the quiet strength of the hydrogen bond, a reminder that sometimes, the most beautiful things are built on connections that are not only strong but also wonderfully fluid.