True Or False Diffusion Takes Place Only In Liquids

Ever wondered why that delightful aroma of freshly baked cookies eventually drifts across your entire house? Or how a drop of food coloring can magically spread through a glass of water until it's all one pretty shade? That, my friends, is the magic of diffusion in action! It’s one of those quiet, everyday phenomena that we often don’t even think about, but it’s actually a super important part of how the world works. And guess what? It’s not just about liquids doing their thing.
So, let’s tackle a common misconception head-on. A question that might pop into your head is: "True or False: Diffusion takes place only in liquids?" My answer to you, with a friendly wink, is a resounding FALSE! While liquids are definitely the rockstars of diffusion in our everyday lives, it’s far from the only place this amazing process happens. Get ready to have your mind gently stretched, like a piece of warm taffy.
Diffusion: The Great Spreader
At its core, diffusion is all about things moving from where there's a lot of them to where there's not so much. Think of it like a party. If you’ve got a massive bowl of popcorn in one corner of the room, and only a few stragglers in another, that popcorn isn’t going to stay put. It’ll start to spread out, bit by bit, until everyone has a chance to grab some. Naturally, without anyone having to force it.
Must Read
In the scientific world, we’re talking about molecules. These tiny, invisible particles are constantly jiggling and wiggling. When they’re all bunched up in one spot (a high concentration), they’ve got a lot of energy and nowhere to go but outwards, bumping into their neighbors and eventually spreading themselves thin. This happens until they’re evenly distributed, or until something stops them.
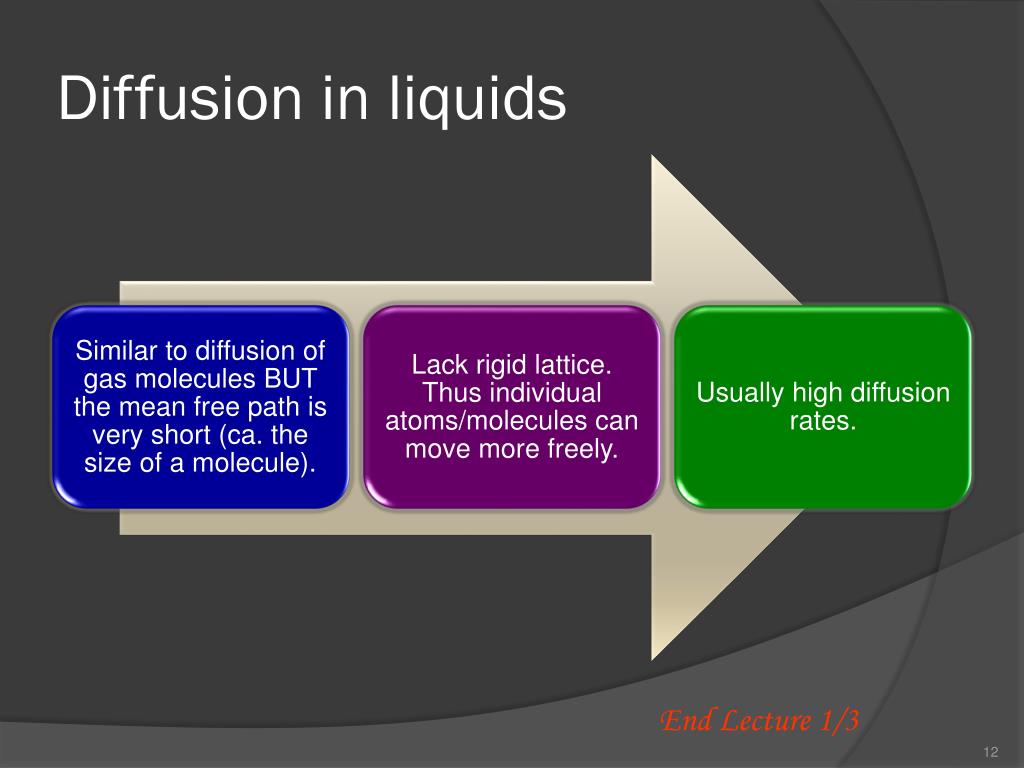
Diffusion in Liquids: Our Familiar Friend
You’ve seen this a million times. That drop of red food coloring in a glass of water? Initially, it’s a concentrated blob of redness. But give it a few minutes, and the red molecules start zipping around, weaving their way through the water molecules. Slowly but surely, the entire glass turns pink. No stirring required, though a little swirl can speed things up!

Or consider sugar dissolving in your tea. You plop in a spoonful of sugar, and at first, it’s just a little lump at the bottom. But as the sugar molecules break free and get to work, they spread throughout the warm liquid. Eventually, your entire cup is sweet. It’s a beautiful, delicious example of liquid diffusion.
Even something as simple as the smell of perfume or air freshener filling a room is diffusion. The scented molecules are initially concentrated in the spray or the potpourri, but they’re so eager to spread out that they travel through the air, reaching your nose from across the room.
But What About Gases?
Here’s where we bust the myth wide open! Diffusion happens with a vengeance in gases too. Think about a skunk. (Sorry, I know, not the most pleasant image, but it’s a very effective one!) When a skunk feels threatened, it lets loose its spray. Those smelly chemicals aren’t just going to hang around the skunk; they’re going to spread out into the air, and boy, do they spread!

The molecules in a gas are even more energetic and further apart than in a liquid. They’re like hyperactive toddlers let loose in a giant playground. They zip and zoom in all directions, colliding with air molecules and spreading out incredibly quickly. This is why you can smell something cooking from another room, or why that pungent durian fruit scent can be so… pervasive.
Another everyday example is when you open a can of tuna in an otherwise scent-free kitchen. Within minutes, the distinct aroma will likely be detectable in other parts of your home. That’s gas diffusion at work, with the volatile compounds from the tuna spreading through the air.
And Even in Solids? (Yes, Really!)
Now, this one might surprise you even more. Diffusion can, and does, happen in solids. It's much, much slower than in liquids or gases, because the molecules in a solid are packed very tightly together and don't move around as much. They’re more like people politely standing in a very crowded elevator, with only the tiniest amount of jostling room.
However, over long periods, diffusion can occur. Imagine two different metals pressed tightly together for years. Even though they’re solids, the atoms at the surface of one metal can slowly migrate into the other. This process is crucial in some manufacturing, like creating alloys or semiconductor components. It’s not something you’ll see happen in your lifetime with a typical household object, but it’s a fundamental aspect of how materials interact at a microscopic level.
A fun (though perhaps not entirely practical) example might be placing a gold coin on top of a silver coin and leaving them for a century. Eventually, the atoms of gold and silver would intermingle slightly at the interface, a testament to diffusion’s relentless, albeit slow, nature even in solids.

Why Should You Care?
Okay, so diffusion happens in liquids, gases, and solids. That’s neat, but why should it matter to you beyond understanding why your socks smell after a long day (that’s sweat molecules diffusing through the fabric!)?
Well, diffusion is the unsung hero behind so many essential processes, both in nature and in technology:
- Breathing: When you inhale, oxygen enters your lungs. Then, diffusion is what allows that oxygen to move from your lungs into your bloodstream, and for carbon dioxide to move out. Without it, we wouldn't be able to get the oxygen we need to live! It's literally life-sustaining diffusion.
- Plants Growing: Plants need nutrients from the soil and carbon dioxide from the air. Diffusion helps move these vital substances into the plant’s cells. It's also how they release oxygen back into the atmosphere.
- Medicine Delivery: Ever taken a pill? The active ingredients in that pill need to dissolve and then diffuse through your body's tissues to reach where they need to work. Controlled diffusion is key to making medicines effective.
- Cooking: That rich flavor in your stew? A lot of that comes from molecules of spices and ingredients diffusing into the broth as it cooks. Diffusion is a major player in making our food taste amazing.
- Technology: From the tiny components in your smartphone to the way batteries work, diffusion plays a critical role in many technological advancements. It's the quiet force enabling our modern world.
So, the next time you notice a smell drifting your way, or see something change color in water, take a moment to appreciate diffusion. It’s not just a science term; it’s the gentle, constant movement that shapes our world, making it a more connected, flavorful, and breathable place. And remember, it’s not just for liquids – it’s everywhere!