The Reduction Of 3-pentanone With Hydrogen
Ever wondered how chemists work their magic to transform one kind of molecule into another? It's a bit like being a culinary artist, but instead of ingredients, we're dealing with atoms and bonds! Today, we're diving into a particularly neat trick: turning a molecule called 3-pentanone into something a little different using good old hydrogen. Now, you might be thinking, "Chemistry? Fun?" But trust us, when you understand the "why" and "how," it gets surprisingly engaging, especially when you see how these simple transformations lead to materials and medicines we use every day. Think of it as unlocking new possibilities, molecule by molecule!
The Makeover: From Ketone to Alcohol
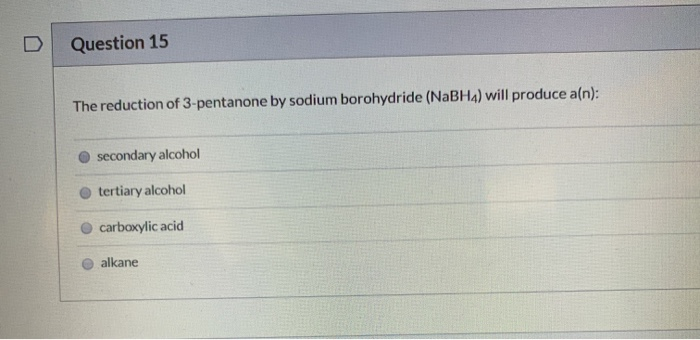
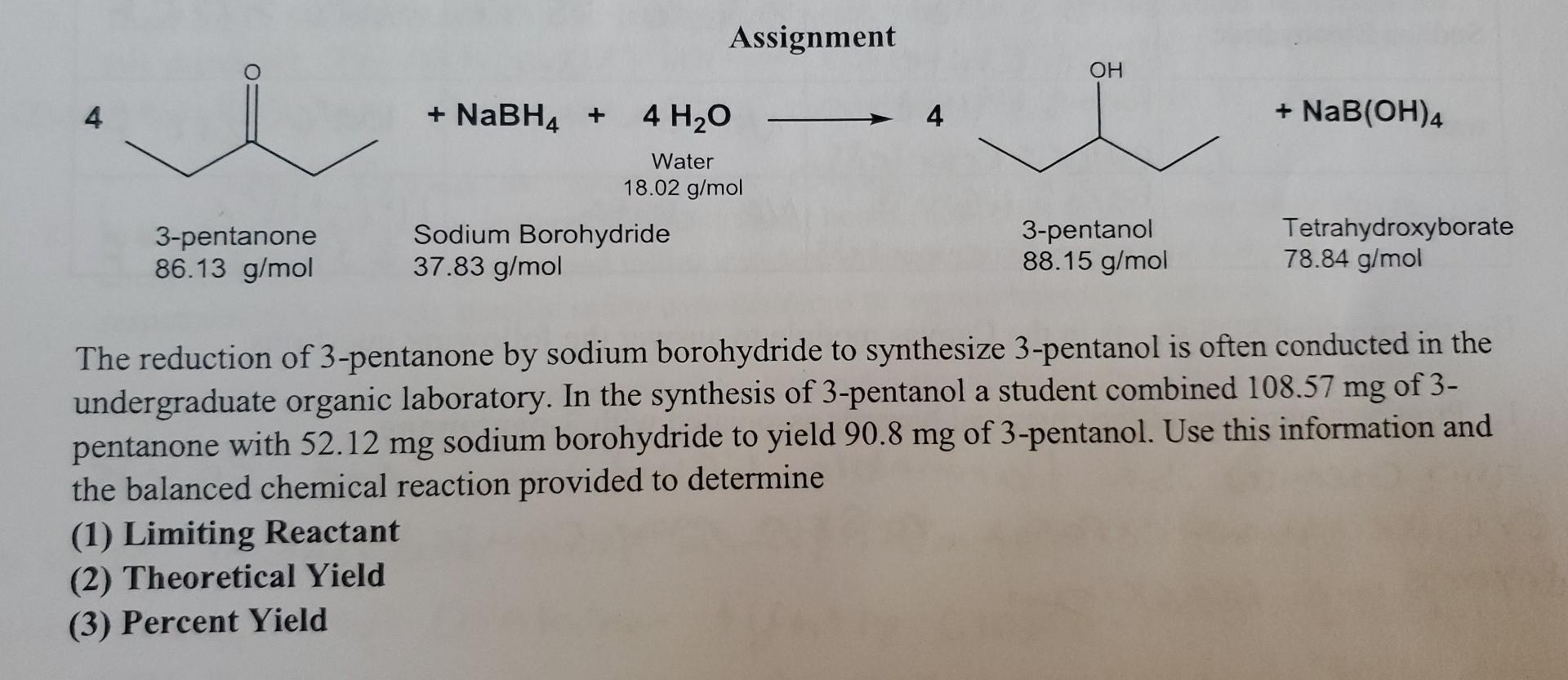
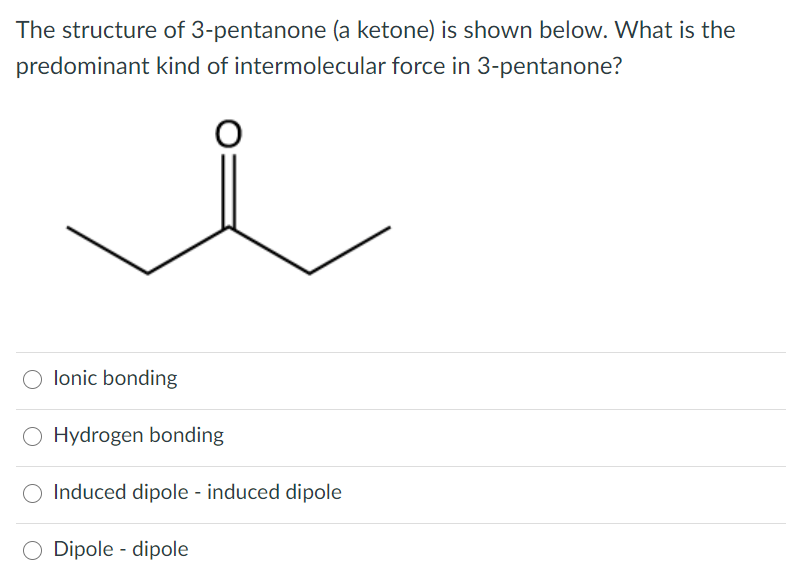
So, what exactly is this 3-pentanone we're talking about? Imagine a five-carbon chain with an oxygen atom double-bonded to the middle carbon. That "ketone" group (the C=O part) is what gives it its unique chemical personality. Now, hydrogen isn't just something you breathe; in chemistry, it's a fantastic little molecule, often represented as H₂. When we bring 3-pentanone and hydrogen together under the right conditions, something special happens: the double bond on the oxygen gets "reduced."
"This reduction process essentially adds hydrogen atoms across that double bond, transforming the oxygen from being double-bonded to carbon into being single-bonded and attached to two hydrogen atoms – making it a hydroxyl group (-OH). This means we've gone from a ketone (3-pentanone) to an alcohol!"
The specific alcohol we get from reducing 3-pentanone is called 3-pentanol. It's a simple, yet significant, chemical change. You can think of it like taking a slightly spicy ingredient and, with a bit of skillful preparation, turning it into something smoother and more versatile. This isn't just a theoretical exercise; this kind of transformation is fundamental to many industrial processes.
Must Read
Why Bother? The Perks of Reduction
Why go through the trouble of transforming 3-pentanone into 3-pentanol? Well, the benefits are numerous and quite impactful. Firstly, alcohols often have different properties than their corresponding ketones. 3-pentanol, for instance, is generally more soluble in water and can participate in different types of chemical reactions. This opens up a whole world of possibilities for creating new compounds.
One of the primary purposes of this kind of reaction is to synthesize specific compounds that are useful as building blocks for more complex molecules. In the pharmaceutical industry, for example, precisely controlled chemical reactions are crucial for creating life-saving drugs. Small changes, like converting a ketone to an alcohol, can alter a molecule's effectiveness, its interaction with the body, or even its safety profile.

Beyond medicine, this type of reduction is vital in the production of polymers and plastics. The ability to precisely tailor the functional groups on molecules allows chemists to design materials with specific strengths, flexibilities, and resistances. Imagine the intricate world of materials science – it's all built on these fundamental transformations!
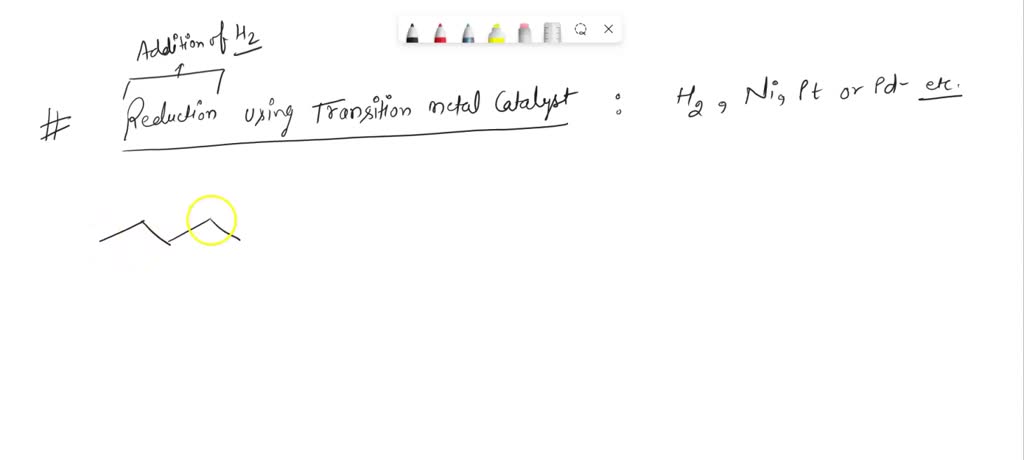
Furthermore, this reaction is a classic example of catalysis. Often, this reduction doesn't happen just by mixing 3-pentanone and hydrogen alone. Chemists use special substances called catalysts – think of them as highly efficient helpers – to speed up the reaction and make it more efficient. Common catalysts for this type of reaction include metals like palladium, platinum, or nickel. These catalysts are often supported on materials like activated carbon, giving them a large surface area to work on.

"The catalyst doesn't get used up in the reaction; it just facilitates the process, making it faster and requiring less energy. This is incredibly important for making chemical processes economically viable and environmentally friendly."
The use of hydrogen as a reducing agent is also quite appealing. Hydrogen is a clean reactant; when it reacts, the main byproduct is often just water, making it a more sustainable choice compared to some other reducing agents that might produce more waste. This focus on green chemistry is a huge driving force in modern chemical research.
In essence, the reduction of 3-pentanone with hydrogen is a demonstration of a fundamental chemical transformation that has far-reaching applications. It's a simple concept – adding hydrogen to change a molecule's structure – but the implications are vast, touching everything from the medicines we take to the materials we use every single day. It’s a testament to the power of chemistry to innovate and improve our world, one molecule at a time!