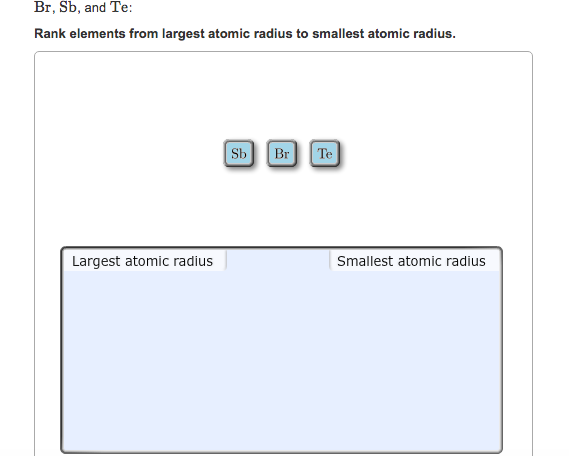
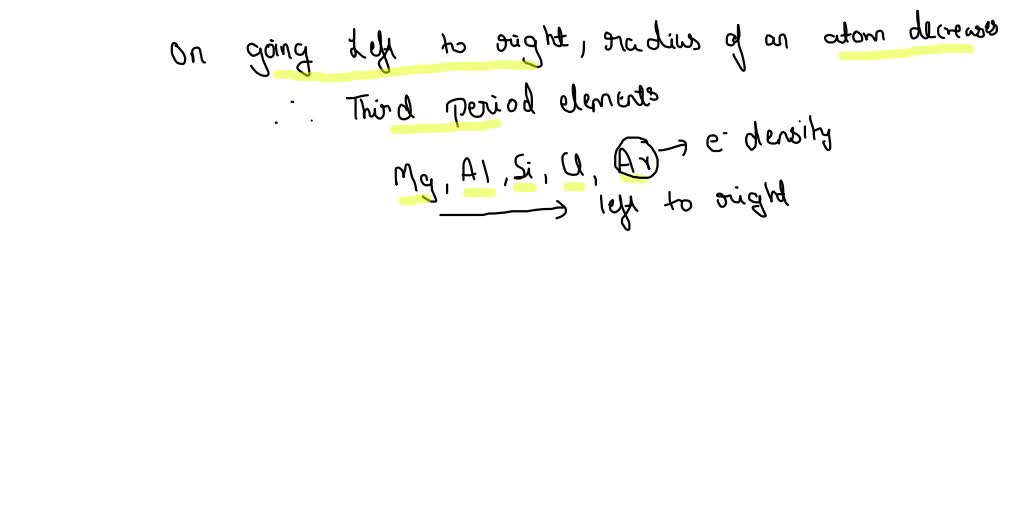Rank From Smallest To Largest Atomic Radius

Ever wondered about the tiny, invisible building blocks that make up everything around us? Well, get ready to have some fun exploring the fascinating world of atoms! Today, we're diving into a cool concept called atomic radius, which is basically a way to measure how big an atom is. Think of it like trying to figure out the size of different marbles – some are tiny, some are a bit bigger. Understanding how these atomic sizes change is surprisingly neat and can even help us understand why different materials behave the way they do. It’s a little bit like a cosmic puzzle, and figuring out the order from smallest to largest atomic radius is a great way to start piecing it together!
Why is this useful, you ask? For beginners just dipping their toes into science, it's a fantastic way to grasp a fundamental concept without getting bogged down in complex jargon. Imagine a family science night: kids can become mini-chemists, guessing which elements might be the smallest and which are the largest based on their position on the periodic table. For hobbyists, like those interested in geology or even crafting with metals, understanding atomic radius can offer insights into how elements bond, why certain metals are malleable, or how different minerals form. It’s a little piece of knowledge that can unlock bigger understandings.
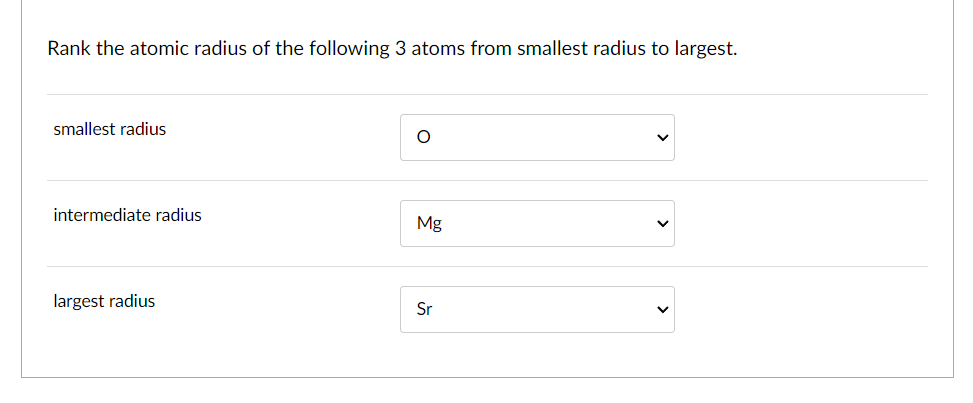
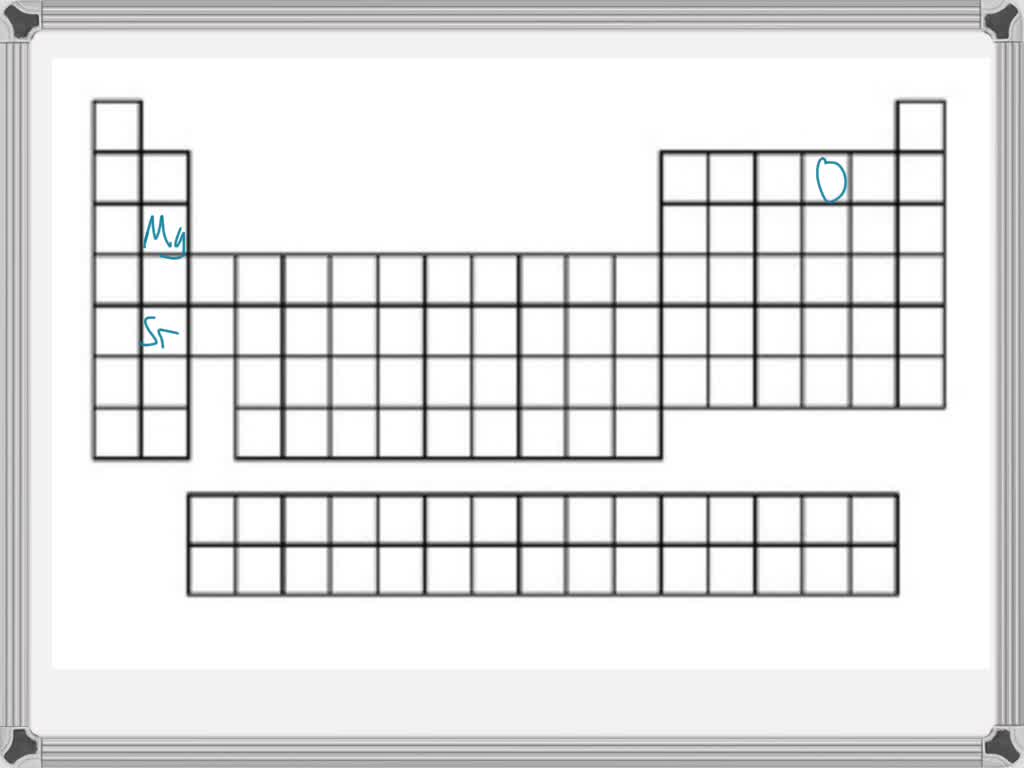
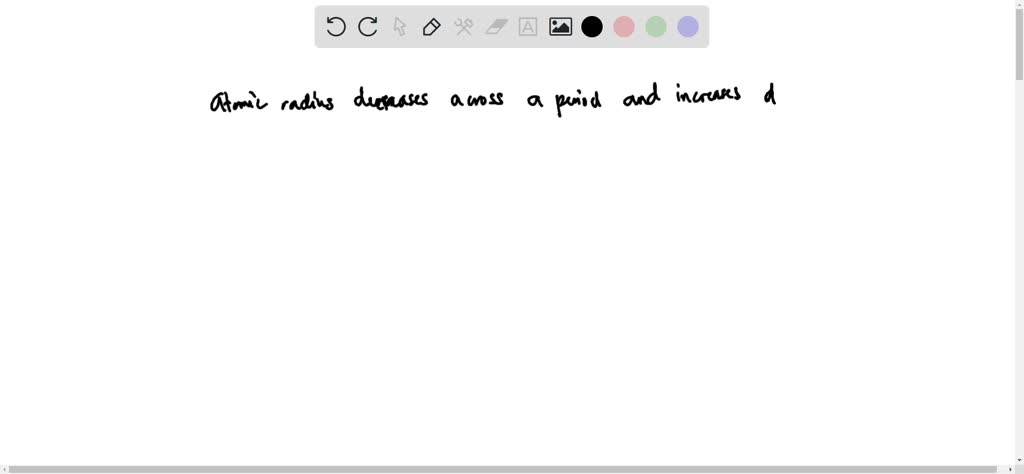
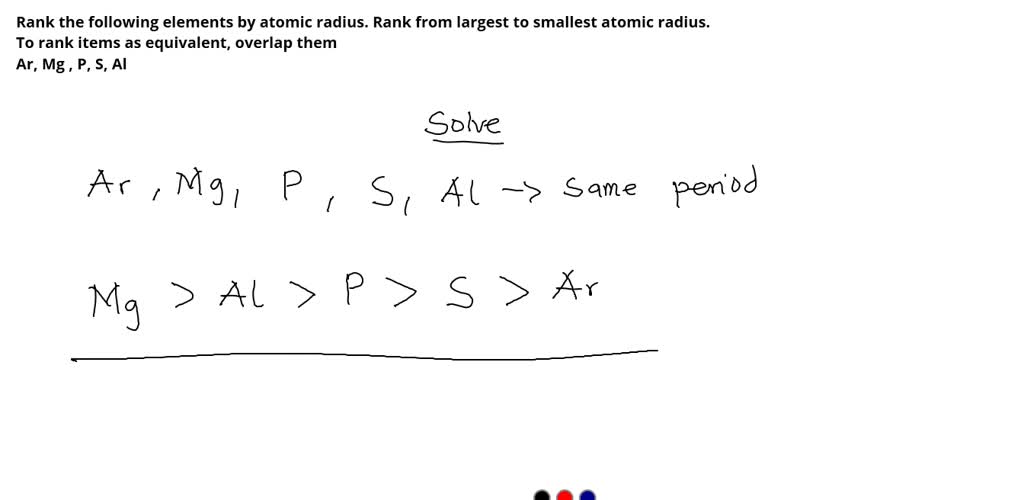
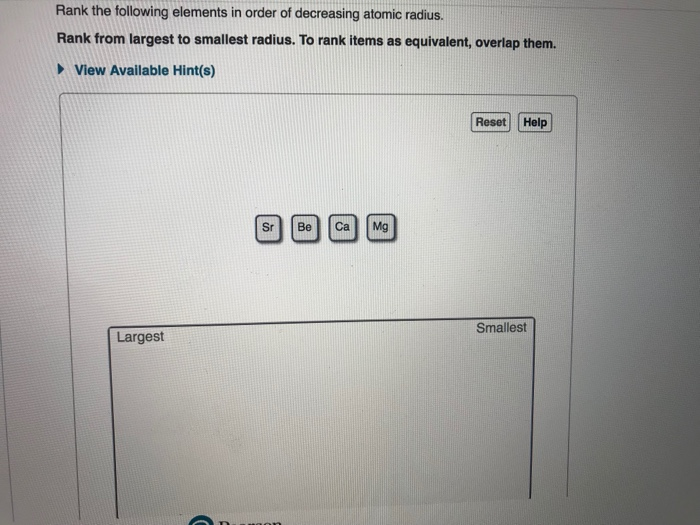
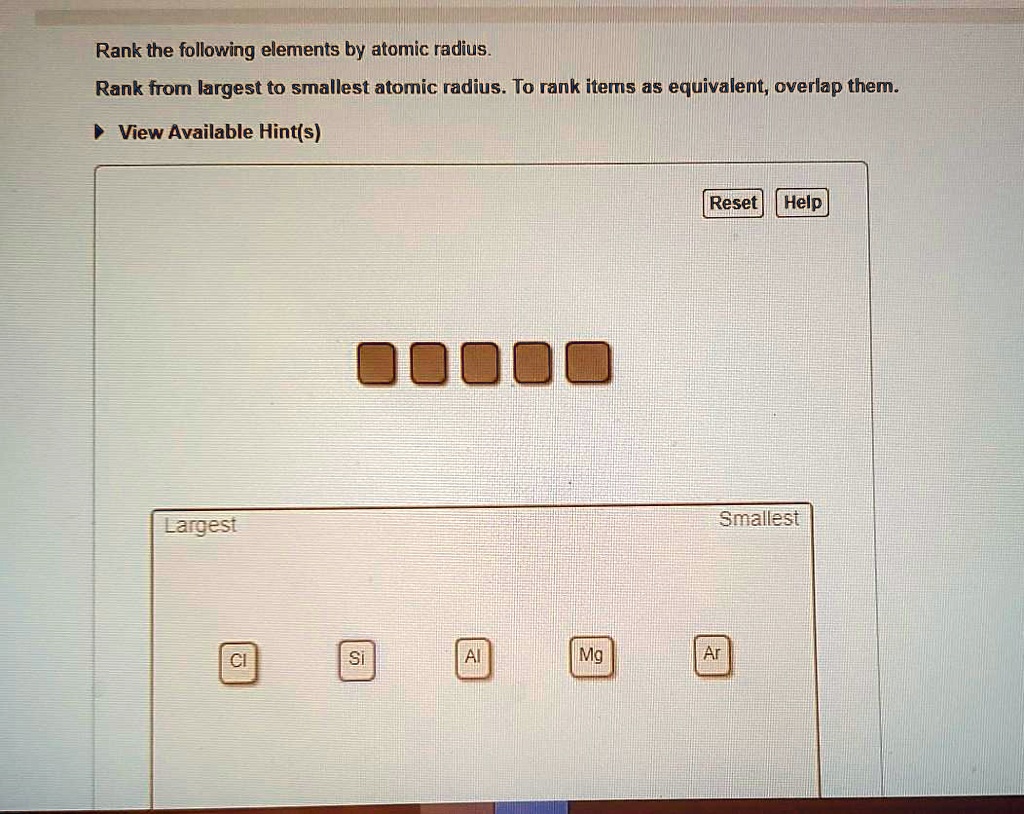
So, how do we actually rank them from smallest to largest? It's not as random as you might think! Generally, as you move across a row on the periodic table from left to right, the atomic radius decreases. This is because the number of protons in the nucleus increases, pulling the electrons closer. Conversely, as you move down a column, the atomic radius increases because there are more electron shells being added, pushing the outer electrons further away from the nucleus. So, elements on the far left and bottom of the periodic table tend to be the largest, while those on the far right and top are the smallest. For instance, Francium (Fr) is one of the largest, while Helium (He) is one of the smallest!
Must Read
Ready to give it a try? The best way to get started is to grab a periodic table – you can find plenty online or in science textbooks. Look at the trends we just discussed. Pick two elements and try to predict which one has a larger atomic radius. For example, compare Sodium (Na) and Chlorine (Cl). Sodium is to the left of Chlorine in the same row. Based on our rule, which one do you think is bigger? (Spoiler alert: Sodium is larger!). You can even try comparing elements in different rows and columns. It’s like a fun game of elemental size comparison!
Exploring atomic radius from smallest to largest is more than just memorizing facts; it’s about appreciating the elegant patterns that govern the universe at its most fundamental level. It’s a simple concept that opens up a whole world of scientific discovery, making learning about chemistry an enjoyable and rewarding experience for everyone. So next time you see a periodic table, remember the little marbles and have fun ranking them!