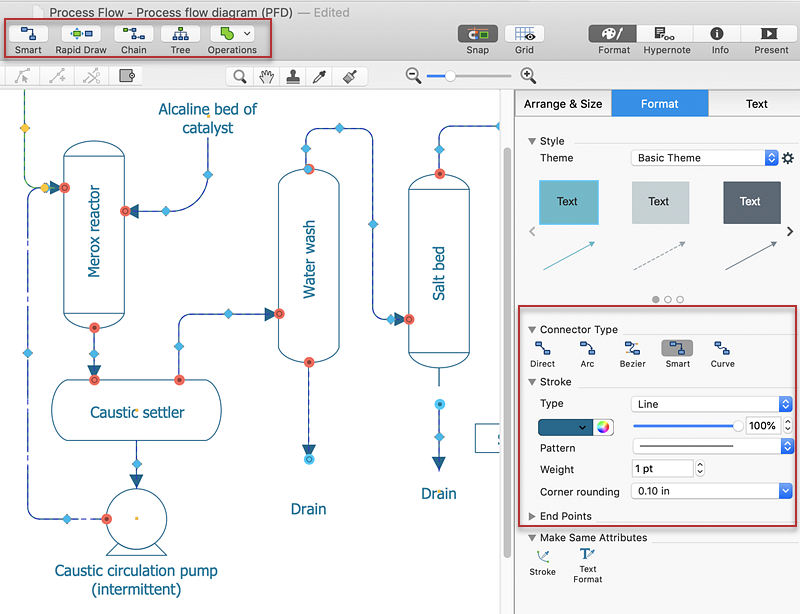
Process To Find Basic Chemicals In A Sample

Ever wondered what makes up that cool rock you found? Or what’s in that weird-smelling liquid your uncle brewed in the garage? It's all about the basic chemicals, my friend! And guess what? Finding them isn't some super-secret mad scientist operation. It's actually pretty neat, like a detective game for your sample.
Think of it like this: your sample is a mystery box. And we want to know what little building blocks are rattling around inside. These building blocks are the elements. They’re like the alphabet of the universe! You’ve got your hydrogen, your oxygen, your carbon – the usual suspects.
So, how do we peek inside this mystery box? Well, scientists have some super cool tricks up their sleeves. It’s not like cracking a safe, but it’s close! We’re basically asking the sample to spill the beans about what it’s made of.
Must Read
The Sneaky Spectacles: Spectroscopy
One of the most popular ways is called spectroscopy. Sounds fancy, right? But it’s all about light. Think of light as a secret message carrier. Different chemicals react to light in different ways. It’s like each chemical has its own unique fingerprint when it comes to light.
Imagine shining a beam of light through your sample. Some of that light gets absorbed. Some of it gets bounced back. And depending on which colors of light are absorbed or bounced, we can tell what’s inside! It’s like saying, "Aha! This sample absorbed a lot of blue and green light, so it must have this chemical in it!"
It’s kind of like how a prism splits white light into a rainbow. Each chemical has its own special rainbow, or spectrum, that it shows when you hit it with the right kind of light. Pretty wild, huh?
There are different types of spectroscopy, too. You’ve got techniques that look at how chemicals absorb light (absorption spectroscopy) and others that look at the light they give off when they’re excited (emission spectroscopy). It's like having a whole toolbox of light-reading gadgets.
A fun fact? Stars are analyzed using spectroscopy! We can tell what elements are in stars millions of light-years away just by looking at the light they send us. So, if it’s good enough for outer space, it's pretty darn good for your backyard sample.

The Fiery Flame Test: A Classic for a Reason
Now, for a really old-school, visually exciting method: the flame test! This is where things get a little more hands-on, and potentially a lot more colorful.
Basically, you take a tiny bit of your sample and dip it into a flame. And bam! Different elements make the flame different colors. It’s like a tiny fireworks show for science!
Lithium? It makes the flame a gorgeous red. Sodium? It turns the flame a bright, unmistakable yellow, like a little sunburst. Potassium? A lovely lilac. Copper? Oh, that’s a stunner – it can give you brilliant blues and greens.
It’s not super precise for identifying complex mixtures, but for spotting the presence of certain common elements? It’s a blast! And honestly, who doesn't love watching flames change color?
Imagine you’re in a science class, and the teacher is doing flame tests. The room is a little dim, and suddenly, whoosh, a flame turns bright orange. Everyone oohs and aahs. That's the magic of elemental colors!
This method relies on the fact that when atoms get heated up, their electrons jump to higher energy levels. When they fall back down, they release energy in the form of light, and the color of that light depends on the element. It's like the electrons are little performers doing a light show!

The Magnetic Tricksters: Magnetic Properties
Some chemicals have a secret superpower: they’re magnetic! Not all of them, of course, but if your sample is attracted to a magnet, you’ve got a clue.
Think iron. That's a classic magnetic material. So, if you wave a magnet over your sample and a bit of it sticks? You’re likely dealing with something containing iron. It's a pretty straightforward way to narrow down possibilities.
This property comes from the arrangement of electrons within the atoms. Some substances have unpaired electrons that line up, making them magnetic. It's like the tiny electrons are holding hands and creating a force field!
It’s not just about iron, either. There are other elements and compounds that exhibit magnetic behavior, though it can be more subtle. But for a quick, fun test? A magnet is your best friend.
Imagine you're a pirate searching for treasure, and your compass (a magnet!) starts spinning wildly over a specific spot. That's kind of what we're doing, but instead of gold, we're looking for the magnetic bits in our sample.

The Chemical Detectives: Reactions and Reagents
Then there are the chemical reactions. This is where things can get a bit more complex, but also super revealing. Scientists use specific chemicals, called reagents, that are known to react in predictable ways with certain other chemicals.
It’s like having a special key that only opens a specific lock. You add a reagent to your sample, and if it fizzes, changes color, or forms a precipitate (that’s fancy talk for a solid chunk forming in a liquid), you’ve just gotten a big hint about what’s in there.
For example, if you add a certain reagent and your sample turns a deep blue, you might be looking at the presence of starch. If it turns a milky white, it could be an indication of certain types of ions.
This is where the fun of chemistry really shines. It’s all about cause and effect. You do one thing, and the sample reacts. It’s a conversation, and the chemical reaction is the answer!
Some reagents are super specific. They’re like the ultimate lie detectors for chemicals. If the reaction happens, you’re 99.9% sure that chemical is present. Pretty neat, right?
Think of it as a scavenger hunt. Each reagent is a clue, and the reaction is finding the treasure. And the treasure is knowing exactly what’s in your sample!
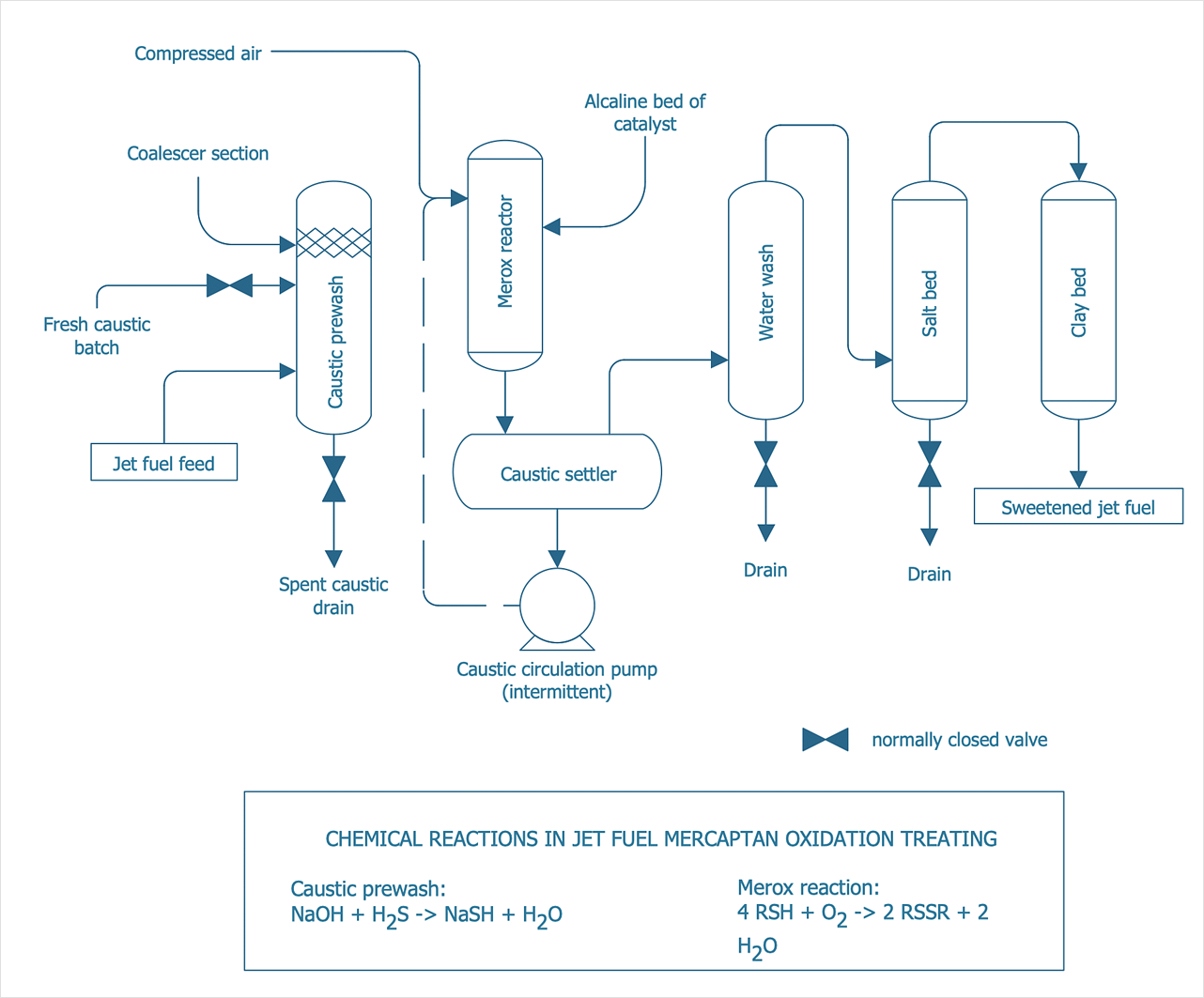
Putting It All Together: The Detective’s Toolkit
So, to find basic chemicals in a sample, scientists often use a combination of these methods. They’re not usually just relying on one trick. They’re using their detective toolkit.
They might start with a broad technique like spectroscopy to get a general idea of the elements present. Then, they might use a flame test to confirm the presence of certain metals. If they suspect a specific compound, they’ll use targeted chemical reagents to confirm it.
It’s all about building a case. Each test provides a piece of evidence. And when all the evidence points to the same conclusion, you’ve cracked the case!
It's like putting together a puzzle. You have all these different pieces – the light fingerprints, the colorful flames, the magnetic attractions, the dramatic reactions – and when you fit them all together, you see the whole picture.
And the best part? This isn’t just for super-advanced labs. Many of these principles are the foundation for experiments you can do at home (with adult supervision, of course!). Learning about the basic chemicals around us is a fantastic way to understand the world a little bit better, and to appreciate all the hidden wonders in even the most ordinary-looking samples.
So next time you see a rock, a plant, or even a questionable puddle, remember that it’s a treasure trove of basic chemicals, just waiting to be discovered. And the methods to find them are as fascinating as the chemicals themselves!