Osmosis And Tonicity Worksheet Answer Key

Ever found yourself staring at a pile of laundry, wondering why your favorite cotton t-shirt suddenly feels like cardboard? Or maybe you've tried to perk up a sad-looking salad, only to have it wilt even further? Well, get ready, because we're about to dive into the wonderfully weird world of osmosis and tonicity. And no, it's not as scary as it sounds! Think of it as the secret sauce behind why things plump up, shrivel, or stay just right. We'll even peek at the magical answers to a worksheet that helps make sense of it all.
So, what exactly is this osmosis thing? Imagine you have a special kind of screen, like a very, very tiny fence, that lets some things pass through but not others. This is called a semipermeable membrane. Now, picture two rooms separated by this fence. In one room, you have a lot of water. In the other, you have less water, but more of something else dissolved in it, like salt or sugar. What do you think happens?
Nature, being the clever thing it is, wants things to be balanced. So, the water molecules will start to move from the room with more water (the less concentrated side) to the room with less water (the more concentrated side) through that tiny fence. They're essentially trying to even out the playing field, diluting the concentrated side until both sides are equally happy. That, my friends, is osmosis in a nutshell! It's the natural flow of water across a membrane from an area of low solute concentration to an area of high solute concentration.
Must Read
Now, how does this connect to our everyday lives? Let's talk about plants. You know those gorgeous, plump tomatoes in your salad? That's osmosis at work! Plant cells have these semipermeable membranes, and when they have enough water, it moves into the cells, making them firm and turgid. Think of them as little water balloons that hold their shape beautifully. That's why watering your plants is so crucial – you're essentially giving their cells the water they need to stay upright and looking their best.
But what happens if a plant doesn't get enough water? The water inside the cells starts to move out, trying to find moisture elsewhere. This is when your leafy greens start to look a bit… droopy. They’re losing their internal water pressure, and it shows! It’s like your own body when you’re dehydrated – you feel a bit sluggish, right? Our cells are constantly engaged in this water-balancing act.

Tonicity: The 'Plumpness' Factor
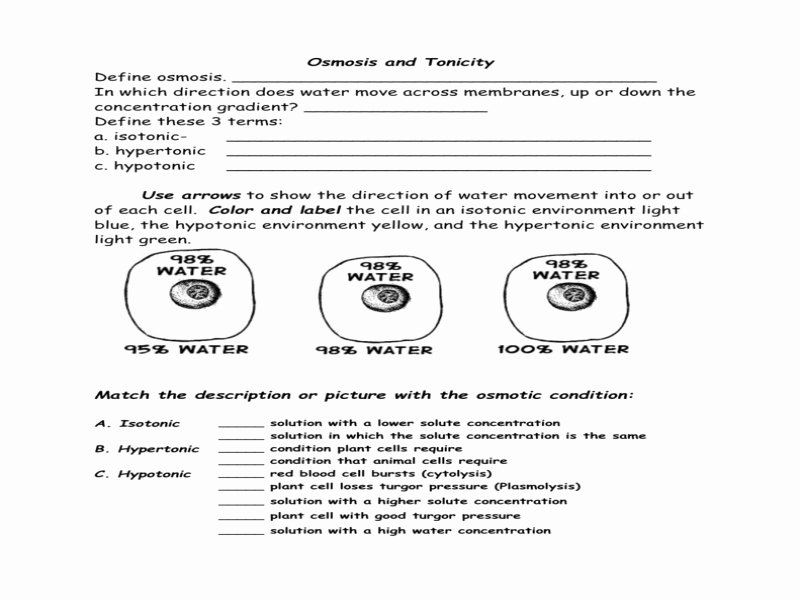
This is where tonicity swoops in. Tonicity describes the concentration of solutes in a solution compared to the concentration of solutes inside a cell. It’s like judging a party based on how many people are inside versus how much space there is. Three main scenarios come into play:
1. Isotonic Solutions: The Chill Vibes
Imagine you're at a party, and the number of people inside perfectly matches the available space. Everyone's comfortable, no one's feeling too crowded, and no one's feeling lonely. This is like an isotonic solution. When a cell is placed in an isotonic solution, the concentration of solutes outside the cell is the same as inside. Water moves in and out at an equal rate, so the cell stays its usual size. Nothing dramatic happens, and that's a good thing for cells in this situation!

2. Hypotonic Solutions: The Plump-Up Party!
Now, picture a party where there's way more space than people. Everyone's got room to spread out, maybe even do a little dance! This is akin to a hypotonic solution. In this case, the concentration of solutes outside the cell is lower than inside. Water, being the social butterfly it is, rushes into the cell to try and dilute the higher solute concentration inside. For most animal cells, this can be a bit too much of a good thing. They might swell up like overfilled water balloons and could even burst! Think of a raisin dropped into water – it plumps up beautifully. Plant cells, however, are built a bit tougher. They have a rigid cell wall that prevents them from bursting, so they just become nicely firm and turgid, which, as we discussed, is great for keeping them perky.
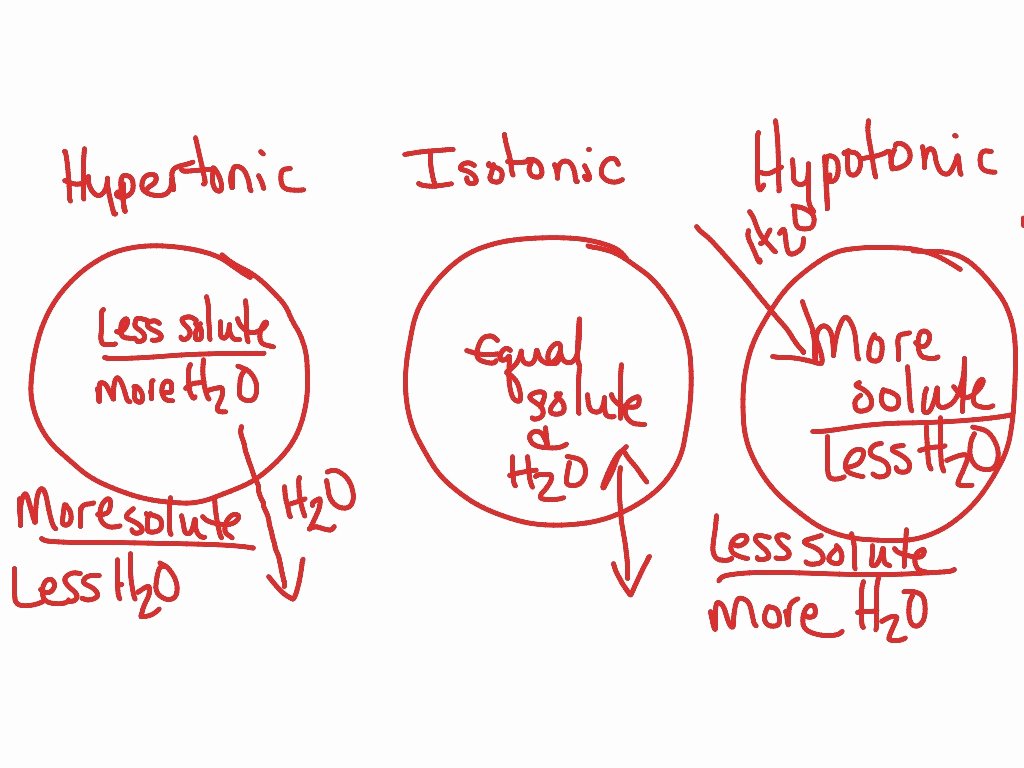
3. Hypertonic Solutions: The Shrink-Down Squeeze
On the flip side, imagine a party where the room is packed tighter than sardines! Everyone's feeling squeezed, and people might start to leave to find more breathing room. This is a hypertonic solution. Here, the concentration of solutes outside the cell is higher than inside. Water, seeking a less crowded situation, moves out of the cell and into the surrounding solution. This causes the cell to shrink and shrivel. Think of that sad, wilted lettuce again. If you put it in a highly concentrated salty or sugary solution, the water will leave the lettuce cells, making it even more limp. It’s like trying to take water out of something.
Why Should We Care About This Stuff?
Beyond keeping our salads crisp and our plants alive, understanding osmosis and tonicity is super important in fields like medicine and biology. For instance, when doctors give you an IV drip, the solution they use has to be carefully balanced to be isotonic with your blood. If it were too concentrated (hypertonic) or too dilute (hypotonic), it could cause serious problems for your red blood cells, making them shrink or swell uncomfortably.
And think about how we preserve food! Salting or sugaring meats and fruits creates a hypertonic environment. Any bacteria or mold cells that land on the food lose water to the sugary or salty surroundings and shrivel up, preventing spoilage. Pretty neat, right? It’s nature’s way of using water movement to keep things fresh!
So, when you see an osmosis and tonicity worksheet answer key, it’s not just a bunch of fancy terms and numbers. It's the explanation for why your grapes get wrinkly in a fruit salad or why those little cucumber slices on your eyes feel so refreshing (they're drawing out excess fluid!). It's about the fundamental movement of water, a process that keeps our bodies functioning, our plants growing, and our food from spoiling. It’s a tiny, invisible dance happening all around us, and now you're in on the secret!
The next time you see something plump up or shrink down, remember the amazing world of osmosis and tonicity. It's a reminder that even the simplest observations in our daily lives are connected to profound scientific principles. It’s all about balance, and water's role in achieving it. So, go forth and appreciate the subtle yet powerful influence of water on the world around you!





