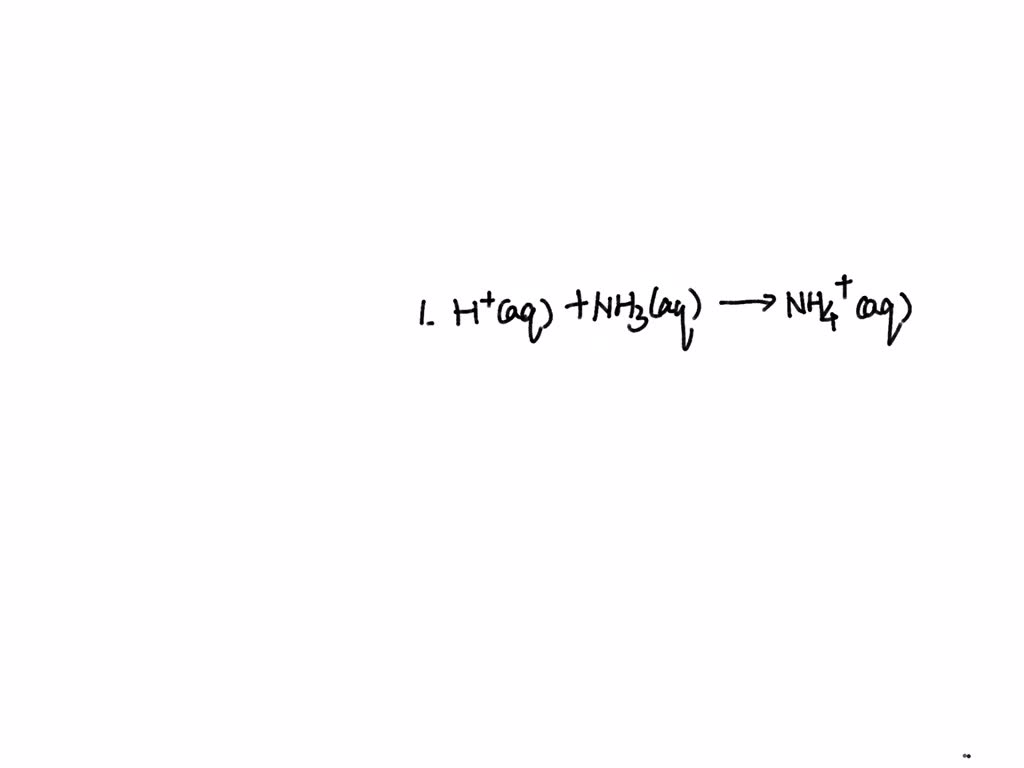Nitric Acid And Ammonia Net Ionic Equation

Hey there! Grab your favorite mug, settle in. We're about to dive into something that sounds super science-y, but trust me, it's actually kinda cool. We're talking about nitric acid and ammonia, and their epic net ionic equation. Don't freak out, okay? It's not like a pop quiz. Think of it more like a behind-the-scenes peek at how these guys interact. You know, like when your nosy neighbor spills the tea? This is kind of like that, but with chemicals. Fun, right?
So, first things first, what even are nitric acid and ammonia? Imagine you've got nitric acid – it’s that powerful stuff, seriously no joke. It’s like the boss of acids, always ready to donate a proton. You know, a little hydrogen ion that’s just itching to escape. And then there's ammonia. Ammonia is super interesting. It's a base, a total opposite of acid, but not in the mean way. More like in the "I'll gladly take that proton off your hands" kind of way. It’s like the welcoming committee for lost protons.
When these two get together, it's a bit of a drama. A chemical drama, of course! Because, you know, opposites attract? Or maybe it's just that they're both a bit… reactive. Nitric acid, HNO₃, is all like, "Here, take this H⁺, I'm done with it!" And ammonia, NH₃, is like, "Ooh, a proton! I'll swoop in and grab that!" It’s a classic acid-base reaction, a real crowd-pleaser in the chemistry world. They essentially neutralize each other, which is a fancy way of saying they calm down and form something new. No more wild protons running around causing trouble.
Must Read
Now, let’s talk about equations. You’ve probably seen those things that look like a secret code, right? Letters, numbers, arrows… it’s enough to make your brain do a little jig. We’re going to break down the overall equation first, the big picture, the whole shebang. So, nitric acid, that’s our HNO₃. And ammonia, that’s our NH₃. They get together, and what do they make? Drumroll, please…ammonium nitrate! Yeah, I know, fancy name. It’s basically NH₄NO₃. It’s a salt, formed from the cation of the base (ammonium, NH₄⁺) and the anion of the acid (nitrate, NO₃⁻). See? They’re like a chemical power couple, perfectly paired.
So, the molecular equation looks like this: HNO₃(aq) + NH₃(aq) → NH₄NO₃(aq). Pretty straightforward, right? The (aq) just means they're dissolved in water. Because, let's be real, most chemical reactions happen in water. It’s like the ultimate hangout spot for molecules. They’re floating around, bumping into each other, doing their thing. Water is the ultimate wingman for chemistry. Without it, a lot of these reactions would be, well, a lot less exciting. Imagine trying to have a party without music, or a pizza without cheese. That's water for a lot of chemistry.
But here’s where it gets really interesting. We're going to take that overall equation and break it down even further. We’re going to look at the full ionic equation. This is where we show everything that's dissolved in the water. Every ion, every molecule that’s doing its own thing. It’s like getting the gossip from everyone at the party, not just the main couple. We want the juicy details, people!
So, when nitric acid, HNO₃, dissolves in water, it’s a strong acid. That means it completely dissociates. It breaks apart into its ions. It’s not shy about it, not like some people. It’s like, "Yup, I’m H⁺ and NO₃⁻, and I’m proud of it!" So, HNO₃(aq) becomes H⁺(aq) + NO₃⁻(aq). Easy peasy. Think of it as nitric acid shedding its coat, revealing its true ionic self. It's practically naked in solution, just ions chilling.
Now, what about ammonia, NH₃? This one’s a little different. Ammonia is a weak base. This means it doesn't completely break apart in water. It’s more like, it partially reacts with water to form ammonium ions (NH₄⁺) and hydroxide ions (OH⁻). So, NH₃(aq) + H₂O(l) ⇌ NH₄⁺(aq) + OH⁻(aq). See that double arrow? That means it’s reversible. It’s not a done deal. Some ammonia stays as NH₃, some becomes NH₄⁺. It’s a bit indecisive, I guess you could say. It's like that friend who can't decide what to order at a restaurant. They're hovering between options, never fully committing. But in our specific reaction with a strong acid like nitric acid, the ammonia gets fully protonated. It’s like the nitric acid is so persuasive, it convinces all the ammonia to take a proton and become ammonium. So, for the purpose of this reaction with a strong acid, we often treat the ammonia as if it’s fully reacting to form NH₄⁺. It’s like a forceful conversion.
And the ammonium nitrate, NH₄NO₃? That's a soluble salt. So, when it forms, it also breaks apart into its ions in water. It’s like, "Okay, we’ve formed, now let's chill separately!" So, NH₄NO₃(aq) becomes NH₄⁺(aq) + NO₃⁻(aq). So, in the water, we've got these little guys swimming around: H⁺, NO₃⁻, NH₃ (though most of it has reacted), and then when the NH₄NO₃ forms, we get NH₄⁺ and NO₃⁻. It’s a full-blown ionic party in there!
So, the full ionic equation looks like this: H⁺(aq) + NO₃⁻(aq) + NH₃(aq) → NH₄⁺(aq) + NO₃⁻(aq). Wait, did I mess up? Let me re-check. Ah, yes! Ammonia is NH₃, and it accepts the proton. So, it's HNO₃ (which gives H⁺) and NH₃ (which takes H⁺). The NH₃ itself doesn't have ions initially, but it forms the ammonium ion. So, when HNO₃ reacts with NH₃, the H⁺ from HNO₃ goes to the NH₃ to form NH₄⁺. And the NO₃⁻ from HNO₃ is just… there. So the product is NH₄NO₃, which then dissociates into NH₄⁺ and NO₃⁻.
Let's try that full ionic equation again, being super careful. We have the reactants: H⁺(aq) + NO₃⁻(aq) from the dissociation of HNO₃, and NH₃(aq) which is a molecule. On the product side, we have NH₄⁺(aq) and NO₃⁻(aq) from the dissociation of NH₄NO₃. So, the full ionic equation is: H⁺(aq) + NO₃⁻(aq) + NH₃(aq) → NH₄⁺(aq) + NO₃⁻(aq). Hmm, still feels a bit off. Let's rethink the reaction itself. Nitric acid (a strong acid) reacts with ammonia (a base). The acid donates a proton (H⁺) to the base. So HNO₃ loses H⁺, becoming NO₃⁻. NH₃ gains H⁺, becoming NH₄⁺. These then combine to form NH₄NO₃, which dissociates into NH₄⁺ and NO₃⁻ in water.
Okay, let’s be super precise. HNO₃ dissociates into H⁺ and NO₃⁻. NH₃ is a molecule that reacts. It doesn’t dissociate in the same way. So the reaction is really between H⁺ and NH₃ to form NH₄⁺. The NO₃⁻ is just a spectator. So, the actual reactants are H⁺ and NH₃. And the products are NH₄⁺ and NO₃⁻. Let’s write the correct full ionic equation:
HNO₃(aq) + NH₃(aq) → NH₄⁺(aq) + NO₃⁻(aq)
Okay, so this is the reaction we’re starting with. Now, let’s expand everything that’s an ion in solution. HNO₃ is a strong acid, so it dissociates: H⁺(aq) + NO₃⁻(aq). Ammonia (NH₃) is a molecular compound, it doesn’t dissociate into ions before reacting. It’s a molecule. The product, ammonium nitrate (NH₄NO₃), is a soluble salt and dissociates into ions: NH₄⁺(aq) + NO₃⁻(aq). So, our full ionic equation is:
H⁺(aq) + NO₃⁻(aq) + NH₃(aq) → NH₄⁺(aq) + NO₃⁻(aq)
See that? It’s showing all the ions and molecules that are actually present in the solution before and after the reaction. It’s like the ultimate character list for our chemical drama. We've got our protons running around, our nitrate ions, our ammonia molecules, and then after the show, our ammonium ions and more nitrate ions.
Now, the fun part! The net ionic equation. This is where we strip away all the stuff that doesn't change. We're looking for the real stars of the show, the ones who are actively participating. Think of it as kicking out all the background characters, the ones who just showed up and then left without doing anything significant. In chemistry terms, these are called spectator ions. They’re just watching the main event unfold, totally unbothered.
So, let's look at our full ionic equation again: H⁺(aq) + NO₃⁻(aq) + NH₃(aq) → NH₄⁺(aq) + NO₃⁻(aq). Can you spot any ions that are on both sides of the arrow, exactly the same? Yup! The NO₃⁻(aq) is chilling on the left and chilling on the right. It went into the reaction as a nitrate ion and came out as a nitrate ion. It didn't participate in the actual chemical transformation. So, it’s a spectator ion. Bye-bye, NO₃⁻!
What's left after we remove the spectator ions? Let’s see…

We have H⁺(aq) on the left. It’s definitely involved because it's an acid. And it forms the NH₄⁺ ion on the right. So, H⁺ is a key player. Good!
We have NH₃(aq) on the left. This is our base, and it accepts the proton. So, NH₃ is also a key player. Excellent!
On the right, we have NH₄⁺(aq). This is the product formed from the reaction of H⁺ and NH₃. So, NH₄⁺ is also a product of the actual reaction. Fantastic!
So, after kicking out the spectator NO₃⁻, what are we left with? We're left with:
H⁺(aq) + NH₃(aq) → NH₄⁺(aq)
And there you have it! The net ionic equation for the reaction between nitric acid and ammonia. It’s like the cliff notes of the reaction. It shows you the essential chemical change that’s happening. It’s the core of the interaction, the real reason these two chemicals decided to have a meet-up.
This net ionic equation is super important because it focuses on the species that are actually reacting. It tells us that in any reaction between a strong acid (that provides H⁺) and ammonia, the fundamental process is the protonation of ammonia to form the ammonium ion. It doesn't matter what other ions are hanging around, as long as they're just spectating. This is why nitric acid and hydrochloric acid (HCl) reacting with ammonia will have the same net ionic equation, because both HNO₃ and HCl are strong acids that readily provide H⁺ ions.
Think about it this way: if you’re watching a movie, and there are a bunch of extras milling around in the background, they’re important for setting the scene, but they’re not driving the plot. The plot is all about the main characters and their interactions. The net ionic equation is like focusing solely on those main characters and their dramatic dialogue. It cuts out all the fluff, all the unnecessary bits, and gets straight to the heart of the matter.
So, what does H⁺(aq) + NH₃(aq) → NH₄⁺(aq) really tell us? It tells us that a free hydrogen ion (from a strong acid) readily combines with an ammonia molecule to form an ammonium ion. It's a direct transfer of a proton. It's a very fundamental acid-base reaction. It's like a handshake, but with atoms and molecules. A very enthusiastic handshake.
It’s fascinating, isn’t it? How we can take a complex chemical reaction and boil it down to its absolute essence. It’s like stripping away all the fancy packaging to get to the real product. And in this case, the real product of the interaction between nitric acid and ammonia is the formation of the ammonium ion from a proton and an ammonia molecule. The nitrate ion just tags along for the ride. It’s like the sensible friend who’s always there, but doesn't get caught up in the main event.
So, next time you hear about nitric acid and ammonia, you can be like, "Oh yeah, I know what’s going on there. It’s that H⁺ + NH₃ → NH₄⁺ thing!" You'll be the coolest person at the chemistry party. You’ll be dropping knowledge bombs like it’s your job. And all thanks to a little bit of understanding about net ionic equations. Who knew chemistry could be so… conversational? It’s like we just had a little chat about it over coffee. Hope it wasn't too strong for you!