Magnesium + Sulphuric Acid Balanced Equation

Ever wondered what happens when you mix everyday-ish stuff and get a little scientific fizz? Well, get ready to dive into the exciting world of chemical reactions, where even something as seemingly simple as Magnesium meeting Sulphuric Acid can spark some serious interest! It’s not just about bubbling beakers; understanding these reactions helps us appreciate the invisible forces that shape our world, from the materials we use to the processes that power industries. Think of it as a microscopic dance of atoms, and today, we’re going to get a front-row seat to one of the most engaging performances: the balanced equation of Magnesium and Sulphuric Acid.
The Dynamic Duo: Magnesium and Sulphuric Acid
So, what’s the big deal with Magnesium and Sulphuric Acid? Well, this particular reaction is a classic for a reason. It’s a great way to introduce the concept of chemical equations and the importance of balancing them. When these two substances meet, something quite dramatic happens: a vigorous reaction. You’ll often see a flurry of bubbles, a release of heat, and the formation of new substances. It’s a tangible demonstration of chemical change that’s both educational and a little bit thrilling.
The primary purpose of understanding this balanced equation is to illustrate the fundamental principle of the conservation of mass in chemistry. This principle states that matter cannot be created or destroyed in a chemical reaction; it can only change form. By balancing the equation, we ensure that the number of atoms of each element on the reactant side (what you start with) is exactly equal to the number of atoms of that same element on the product side (what you end up with). This isn’t just an academic exercise; it’s crucial for predicting the exact amounts of substances involved in any chemical process, from baking a cake to manufacturing life-saving medicines.
Must Read
The benefits of grasping this concept extend far beyond a single reaction. For students, it’s a foundational step in learning chemistry, opening the door to understanding more complex reactions and chemical processes. For those interested in science, it provides a satisfying sense of order and logic in the seemingly chaotic world of atoms and molecules. In practical terms, accurately predicting and controlling chemical reactions is vital for:
- Industrial Chemistry: Ensuring that chemical plants produce desired products efficiently and safely, minimizing waste and maximizing yield. Think of producing fertilizers, plastics, or even refining metals.
- Environmental Science: Understanding how pollutants react in the environment and developing methods to neutralize or remove them.
- Materials Science: Designing new materials with specific properties by carefully controlling their chemical composition and reactions.
- Everyday Life: Even seemingly simple things like understanding how batteries work or how certain cleaning products function rely on these fundamental chemical principles.
Let’s talk about the players involved. Magnesium (Mg) is a shiny, silvery metal that’s surprisingly lightweight and reactive. It’s found in many everyday items, from fireworks to antacids! Then there’s Sulphuric Acid (H₂SO₄), a strong, corrosive mineral acid. It’s a workhorse in industries, used in everything from fertilizer production to petroleum refining. When these two get together, they don’t just say hello; they have a chemical conversation that leads to some fascinating transformations.
The Balancing Act: From Messy Mix to Perfect Equation
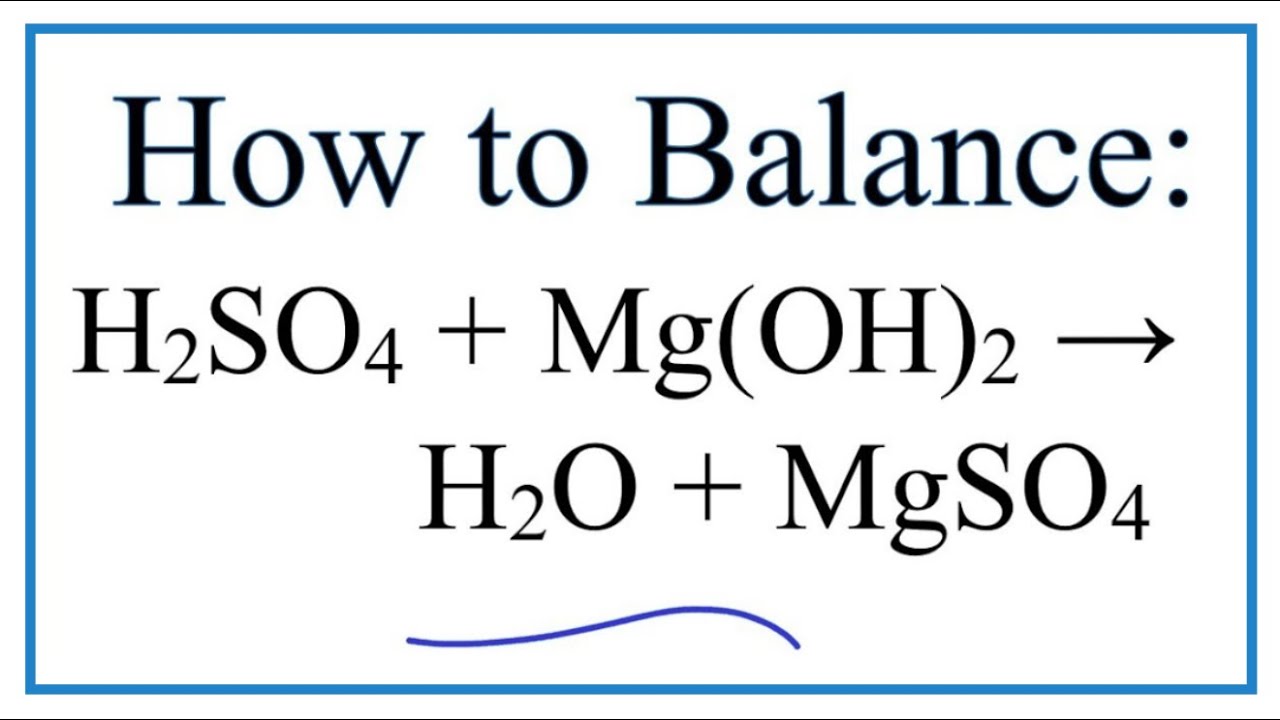
When you mix Magnesium metal with Sulphuric Acid, a chemical reaction takes place. You can observe it with your own eyes! The magnesium starts to fizz and dissolve, producing a gas and a new salt. The equation that describes this interaction, before we get our balancing act perfect, looks something like this:
Mg + H₂SO₄ → MgSO₄ + H₂
Now, this looks pretty neat, right? But remember the conservation of mass? We need to make sure we have the same number of atoms of each element on both sides of the arrow. Let’s take a peek:

- Magnesium (Mg): We have 1 Mg atom on the left and 1 Mg atom on the right. Looks good!
- Sulphur (S): We have 1 S atom on the left and 1 S atom on the right. Still good!
- Oxygen (O): We have 4 O atoms on the left and 4 O atoms on the right. Excellent!
- Hydrogen (H): We have 2 H atoms on the left and 2 H atoms on the right. Perfect!
In this particular case, the initial representation actually is the balanced equation! This is a wonderful example because it’s a straightforward one-to-one, one-to-one, one-to-one, one-to-one ratio of atoms reacting and forming. This means for every one atom of Magnesium that reacts, one molecule of Sulphuric Acid is needed, and in return, one molecule of Magnesium Sulphate (MgSO₄) and one molecule of Hydrogen gas (H₂) are produced.
The products of this reaction are Magnesium Sulphate (MgSO₄), which is a white crystalline salt, and Hydrogen gas (H₂). You’ll see the hydrogen gas escaping as bubbles – that’s the fizzing you observe! The formation of hydrogen gas is a key indicator that a reaction has occurred. The balanced equation isn’t just a set of symbols; it’s a precise recipe for a chemical change.
Understanding this balanced equation, Mg + H₂SO₄ → MgSO₄ + H₂, allows us to predict outcomes with certainty. If you had a certain amount of Magnesium, you’d know exactly how much Sulphuric Acid you’d need to react with it completely, and you’d know exactly how much Magnesium Sulphate and Hydrogen gas would be produced. This predictive power is what makes chemistry so incredibly useful and fascinating. So, the next time you see something bubble, remember the elegant dance of atoms and the crucial role of a balanced equation in bringing order to the chemical universe!