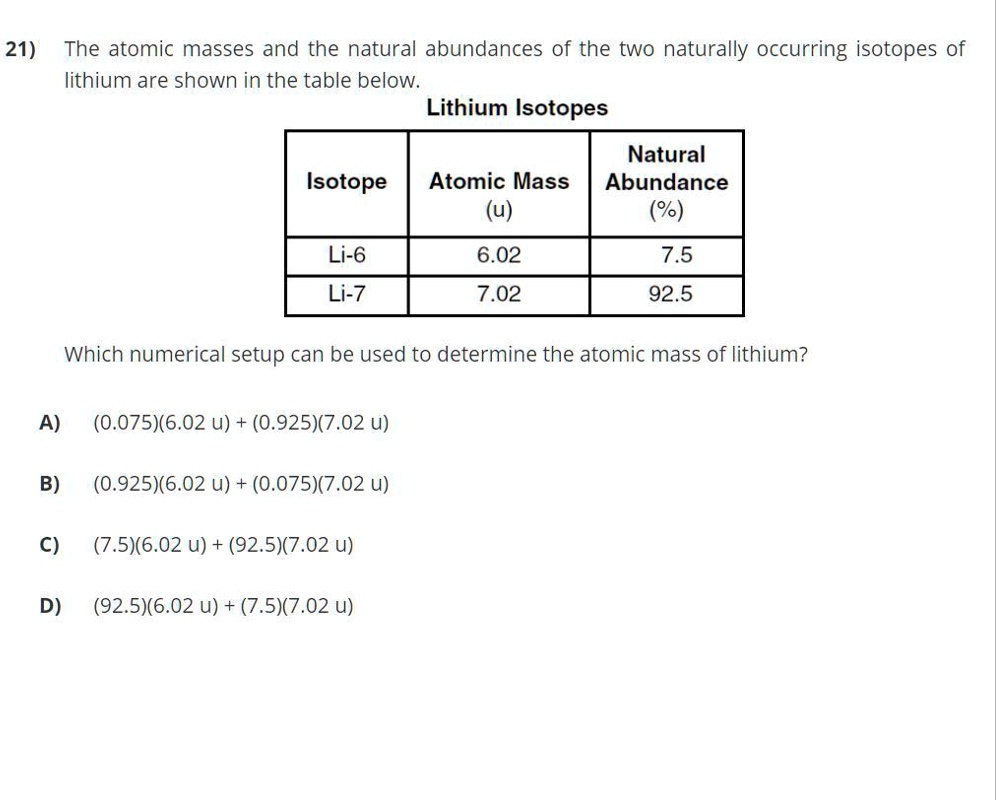
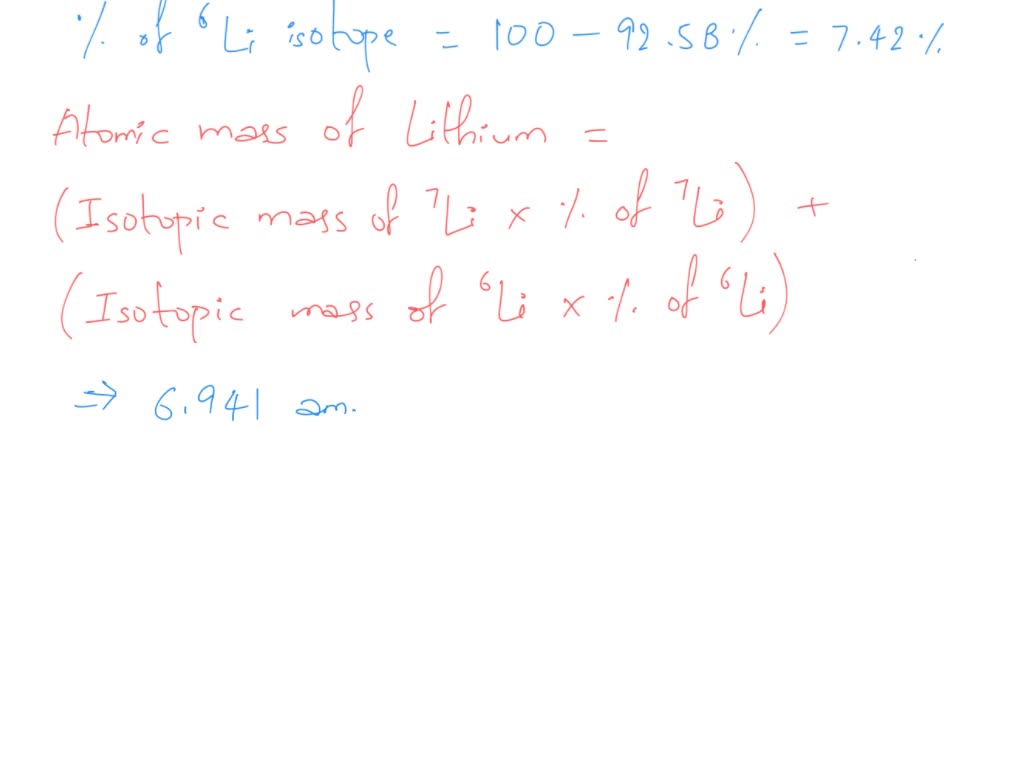Lithium Has Two Naturally Occurring Isotopes

So, let's talk about Lithium. You know, the stuff in your phone batteries? Turns out, this metal has a bit of a secret life. It's not just one, solitary element. Nope, Lithium is a bit of a twin. It's got siblings, naturally occurring ones!
We’re not talking about cousins or distant relatives here. These are its own flesh-and-blood, so to speak. These are its isotopes. Think of it like identical twins, but with a tiny, almost invisible difference.
The universe, in its infinite wisdom (or perhaps just a cosmic prank), decided that Lithium needed a bit of variety. So, it gave us Lithium-6 and Lithium-7. These are the two main players in the Lithium natural troupe.
Must Read
Now, you might be wondering, what’s the big deal? They’re both Lithium, right? Well, yes and no. They both act like Lithium. They both have that same charming Lithium personality. But they have slightly different weights.
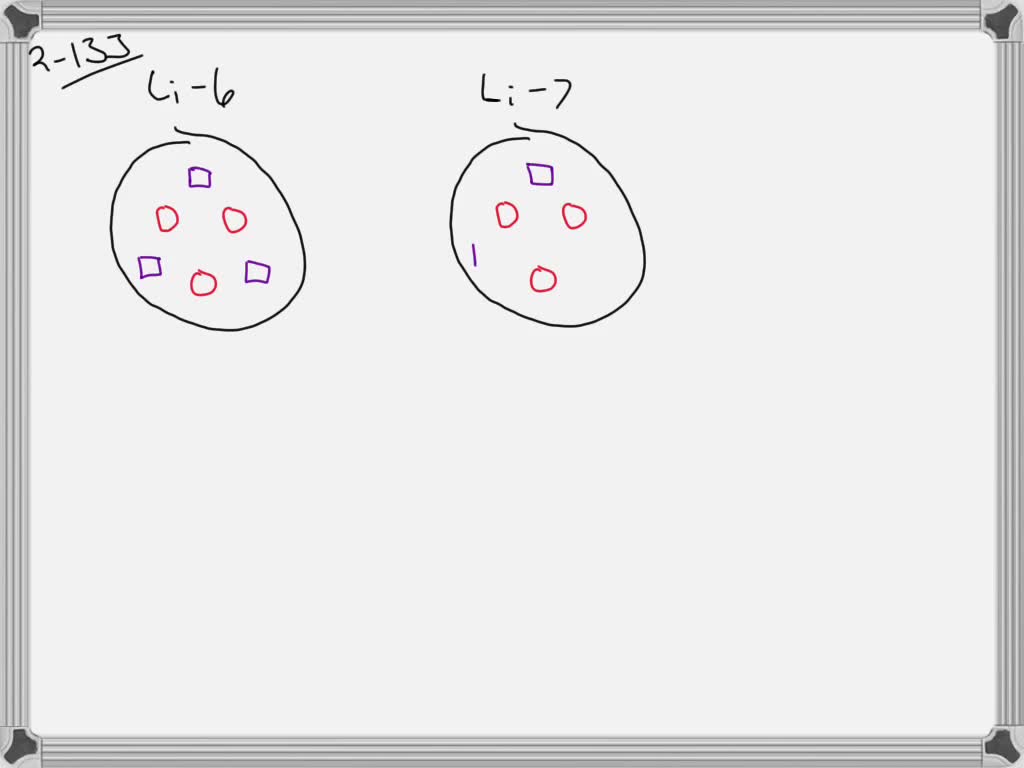
Imagine you have two brothers. They look almost exactly alike. They love the same video games. But one of them just happens to have a slightly heavier backpack. That's kind of like our Lithium isotopes. A slight difference in their "backpacks" – their neutrons!
The most common one, the one that really rocks the Lithium world, is Lithium-7. This guy is the popular kid. It makes up the vast majority of all the Lithium you'll find hanging out on Earth.
And then there’s Lithium-6. It’s a bit more rare. It’s like the quieter, more introverted sibling. Still important, still part of the family, but just doesn't grab the spotlight as much.
This difference in weight, it’s not something you can see with the naked eye. You’d need some serious scientific equipment to tell them apart. It's like trying to tell apart two sprinkles that are infinitesimally different in size. Still a sprinkle, but technically, not identical.
Why does this even matter? Well, for most everyday things, like charging your phone, it doesn't really. Your phone doesn't care if it's getting its energy from Lithium-6 or Lithium-7. It just wants to be powered up!
But in the world of science and some super-specific applications, these subtle differences can be a big deal. It's like how a chef knows that slightly different seasoning can change a whole dish. The same principle applies here.
Think about it like this: imagine you're a detective. You find two identical fingerprints. But one has a tiny smudge. That smudge, while small, might be the key to cracking the case. Our isotopes are kind of like those subtle clues.
Lithium-6, being lighter, can behave a little differently in certain chemical reactions. It’s like that lighter backpack – it might be easier for it to move around in some situations.
Scientists use this difference. They can actually separate these isotopes. It’s not easy, and it’s not cheap. It’s like trying to pick out one specific grain of sand from a beach. A very, very tiny and specific grain.
One of the coolest applications is in nuclear energy. Lithium-6 plays a special role in nuclear reactors. It can capture neutrons and then release its own. It's like a tiny energy exchange program happening at an atomic level.
It’s also used in making certain kinds of nuclear weapons. Yeah, not the most cheerful topic, but it's a reality of how these different isotopes can be used.
And then there's the whole field of geology and understanding the history of Earth. The ratio of Lithium-7 to Lithium-6 in rocks can tell scientists a lot about how those rocks formed and what processes they've been through.
It’s like reading ancient history written in stone, but the language is the subtle variation in Lithium's weight. Fascinating, right?
So, the next time you’re scrolling on your tablet or binge-watching a show, give a little nod to Lithium. And remember, it’s not just one element, it’s a dynamic duo, or rather, a dynamic pair, of naturally occurring isotopes!
It’s a reminder that even the simplest things around us can have hidden complexities. Things we don't think about, like the subtle differences in the weight of tiny atoms.
And honestly, isn't that a little bit amazing? That something so small, so fundamental, has this internal variation? It’s like discovering your favorite coffee mug has a secret twin sibling you never knew about.

The universe is full of these little surprises. And Lithium's isotopic nature is just one of them. It’s a testament to the intricate dance of matter and energy that’s always going on, whether we’re paying attention or not.
It's an "unpopular opinion" of mine, I guess. That the seemingly simple elements are actually quite fascinating in their subtle variations. That there's beauty in the almost-identical.
So, Lithium-7 and Lithium-6. They are the unsung heroes of the periodic table, working quietly behind the scenes, powering our devices and telling stories of Earth's past. They’re the dynamic duo that makes Lithium, well, Lithium, but with a bit of a twist.
And who knows what other tiny, hidden differences exist in the world around us, just waiting to be discovered or, at the very least, appreciated? It makes you wonder, doesn't it?
It’s these small details that make the grand picture so much more interesting. The subtle nuances that add depth and character. Just like our Lithium isotopes.
So, cheers to Lithium, and cheers to its two naturally occurring isotopes! May they continue to power our world and intrigue our minds with their subtle, yet significant, differences.

They are a perfect example of how even the most basic building blocks of our universe can have their own unique identities. Even if those identities are only distinguishable by the most discerning scientific eyes.
And that, my friends, is pretty darn cool. It’s the little things, you know? The almost-identical twins of the atomic world.
It's like having two flavors of the same ice cream, but you can only tell the difference if you're a super-taster. Still delicious, just... subtly different.
So, next time you see that little battery icon, remember Lithium. Remember its two natural isotopes. It’s a whole lot more than just a metal. It’s a family affair!
And that’s a thought that’s definitely worth smiling about. The universe is full of tiny marvels, and Lithium's isotopic twins are definitely among them.
They’re a subtle reminder that there's always more to discover, even in the elements we think we know.